Unit 8 Stoichiometry Notes Balancing Equations Stoichiometry Limiting

Unit 8 Stoichiometry Notes Balancing Equations, Stoichiometry, Limiting Reactants, % Yield
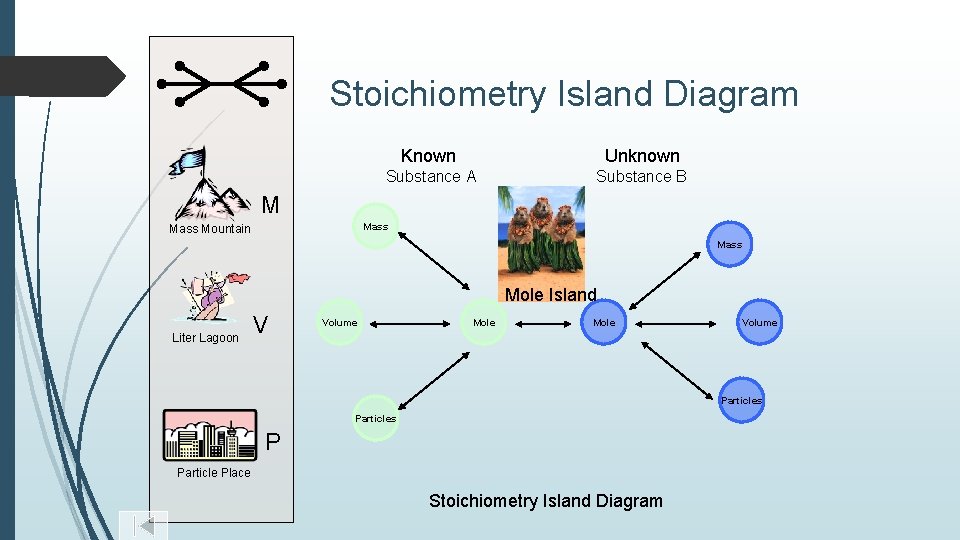
Stoichiometry Island Diagram Known Unknown Substance A Substance B M Mass Mountain Mass Mole Island Liter Lagoon V Volume Mole Volume Particles P Particle Place Stoichiometry Island Diagram
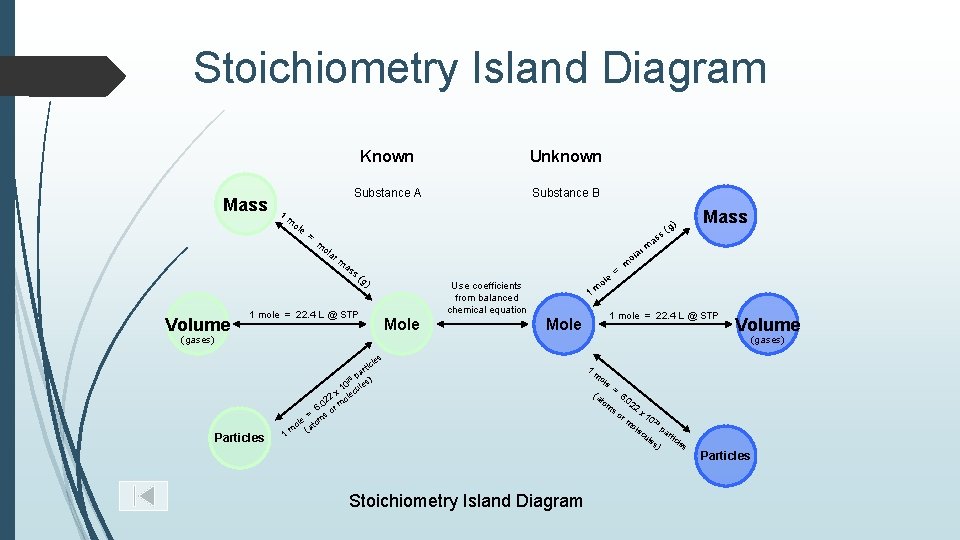
Stoichiometry Island Diagram Mass 1 m ole = Known Unknown Substance A Substance B g s( rm as s( g) Use coefficients from balanced chemical equation 1 mole = 22. 4 L @ STP Mole 1 m ole = m m lar o 1 mole = 22. 4 L @ STP Mole (gases) Volume (gases) les Particles Mass as m ola Volume ) 1 c rti pa ) 23 0 les x 1 lecu 2 o 02 6. or m = s m ole (ato m Stoichiometry Island Diagram 1 m ole (a to m = 6. 0 22 x 1 rm 0 23 ole pa cu r les ticle ) s so Particles
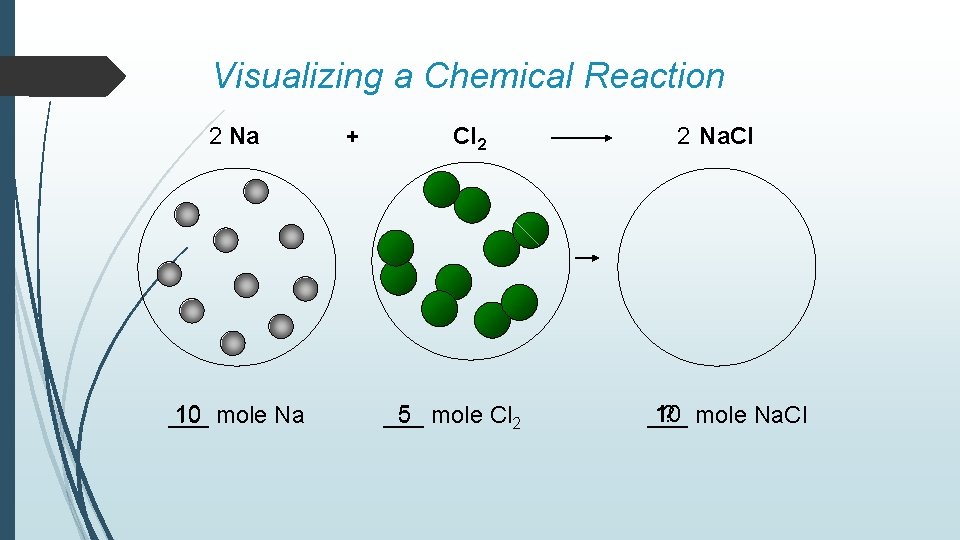
Visualizing a Chemical Reaction 2 Na 10 mole Na ___ + Cl 2 5 mole Cl 2 ___ 2 Na. Cl 10 ? mole Na. Cl ___
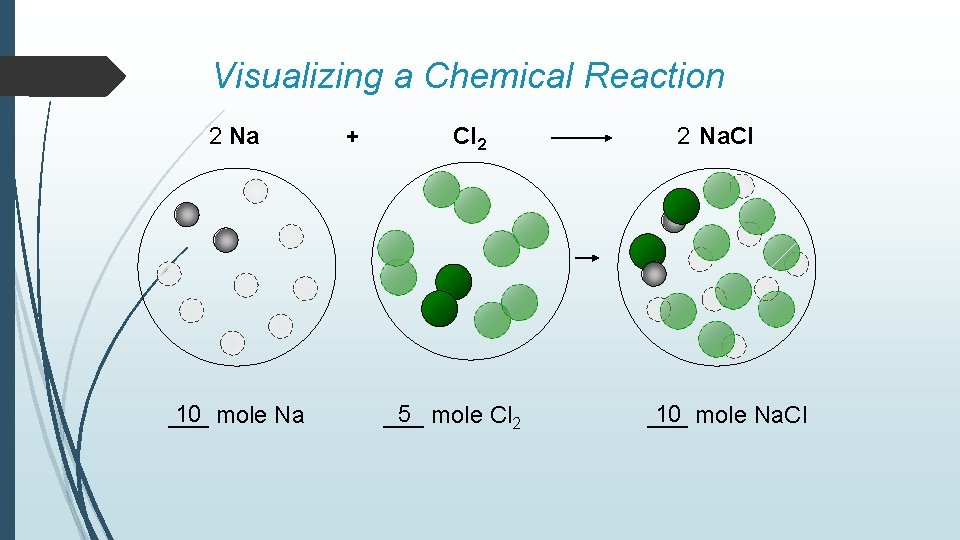
Visualizing a Chemical Reaction 2 Na 10 mole Na ___ + Cl 2 5 mole Cl 2 ___ 2 Na. Cl 10 mole Na. Cl ___

Formation of Ammonia N 2 (g) 3 H 2 (g) + 2 NH 3 (g) + 2 atoms N + 6 atoms H 2 atoms N and 6 atoms H 1 molecule N 2 + + 3 molecules H 2 2 molecules NH 3 30 molecules H 2 20 molecules NH 3 10 molecule N 2 1 x 6. 02 x 1023 molecules N 2 1 mol N 2 28 g N 2 + 3 x + + 6. 02 x 1023 molecules H 2 22. 4 L N 2 + 22. 4 L 6. 02 x 1023 molecules NH 3 3 mol H 2 2 mol NH 3 3 x 2 g H 2 2 x 17 g NH 3 34 g reactants Assume STP 2 x 22. 4 L 67. 2 L H 2 22. 4 L 34 g products 22. 4 L 44. 8 L NH 3

Molar Volume at STP 1 mol of a gas=22. 4 L at STP Standard Temperature & 0°C and 1 atm Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem Pressure
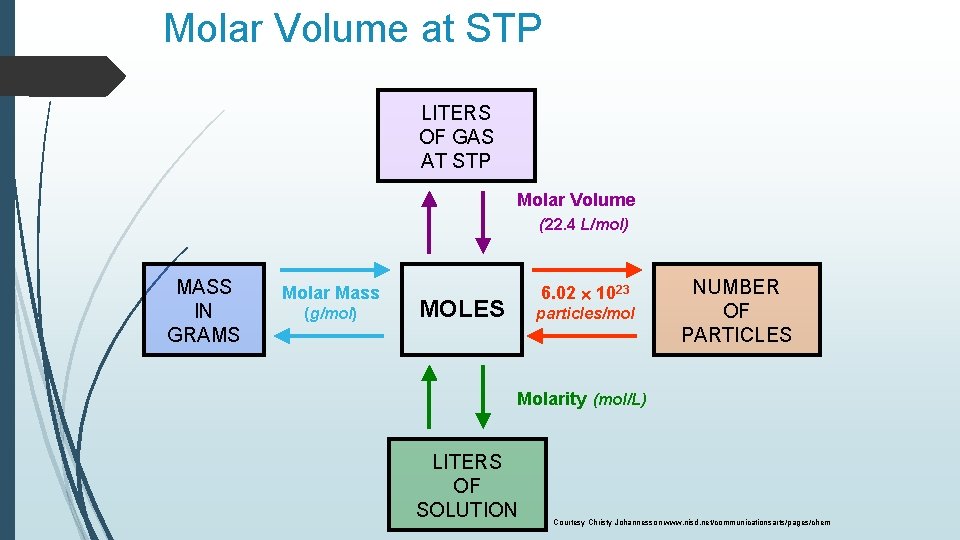
Molar Volume at STP LITERS OF GAS AT STP Molar Volume (22. 4 L/mol) MASS IN GRAMS Molar Mass (g/mol) 6. 02 1023 MOLES particles/mol NUMBER OF PARTICLES Molarity (mol/L) LITERS OF SOLUTION Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
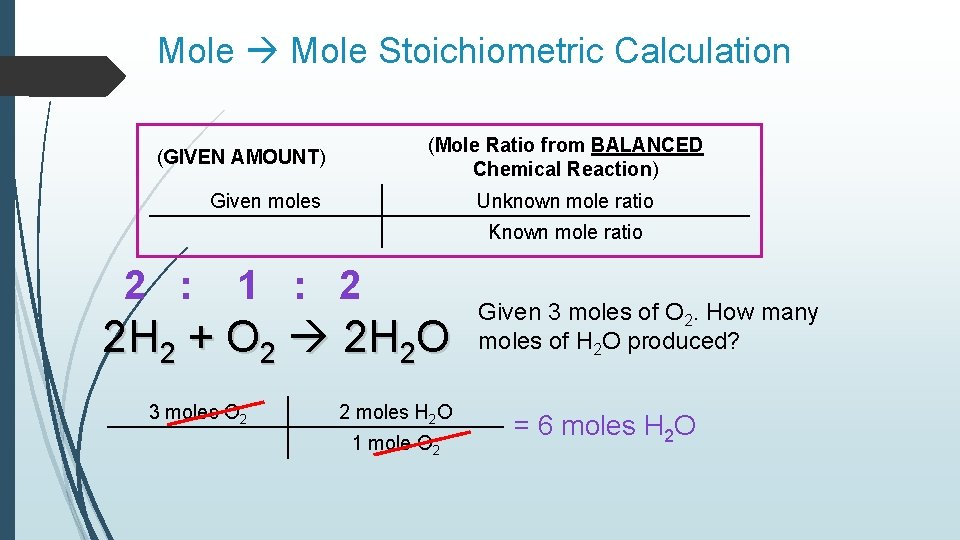
Mole Stoichiometric Calculation (Mole Ratio from BALANCED Chemical Reaction) (GIVEN AMOUNT) Given moles Unknown mole ratio Known mole ratio 2 : 1 : 2 2 H 2 + O 2 2 H 2 O 3 moles O 2 2 moles H 2 O 1 mole O 2 Given 3 moles of O 2. How many moles of H 2 O produced? = 6 moles H 2 O

Mole Mass Stoichiometric Calculation (GIVEN AMOUNT) (Molar Mass) (Mole Ratio from BALANCED Chemical Reaction) Given mass 1 mol known Unknown mole ratio mass known Known mole ratio 2 : 1 : 2 2 H 2 + O 2 2 H 2 O 4 moles H 2 Given 4 moles of H 2. How many grams of H 2 O produced? 2 moles H 2 O 18. 02 g H 2 O 2 mole H 2 1 mol H 2 O = 72. 08 grams H 2 O

Mass Stoichiometric Calculation (GIVEN AMOUNT) (Molar Mass Known) (Mole Ratio from BALANCED Chemical Reaction) (Molar Mass Unknown) Given mass 1 mol known Unknown mole ratio mass unknown mass known Known mole ratio 1 mol unknown 2 : 1 : 2 2 H 2 + O 2 2 H 2 O 64 g O 2 How many grams of H 2 are needed to react with 64 grams of O 2? 1 mole O 2 2 mol H 2 2. 02 g H 2 32 g O 2 1 mol H 2 = 8. 08 grams H 2

Mole Liters Stoichiometric Calculation (GIVEN AMOUNT) (Mole Ratio from BALANCED Chemical Reaction) Liter/mol conversion Given moles Unknown mole ratio 22. 4 L unknown Known mole ratio 1 mol unkown 2 : 1 : 2 2 H 2 + O 2 2 H 2 O 3 moles O 2 Given 3 moles of O 2. How many liters of H 2 needed? 2 moles H 2 22. 4 L H 2 1 mole O 2 1 mol H 2 = 134. 4 Liters H 2

Stoichiometry Practice Problems How many moles of KCl. O 3 must decompose in order to produce 9 moles of oxygen gas? 2 KCl. O 3 2 KCl + 3 O 2 ? mol 9 mol O 2 9 mol 2 mol KCl. O 3 3 mol O 2 = 6 mol KCl. O 3 Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

How many moles of KCl. O 3 must decompose in order to produce 9 moles of oxygen gas? 2 KCl. O 3 2 KCl + 3 O 2 ? mol 9 mol O 2 9 mol 2 mol KCl. O 3 3 mol O 2 x mol KCl. O 3 = 9 mol O 2 2 mol KCl. O 3 3 mol O 2 = 6 mol KCl. O 3 Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem 6 mol
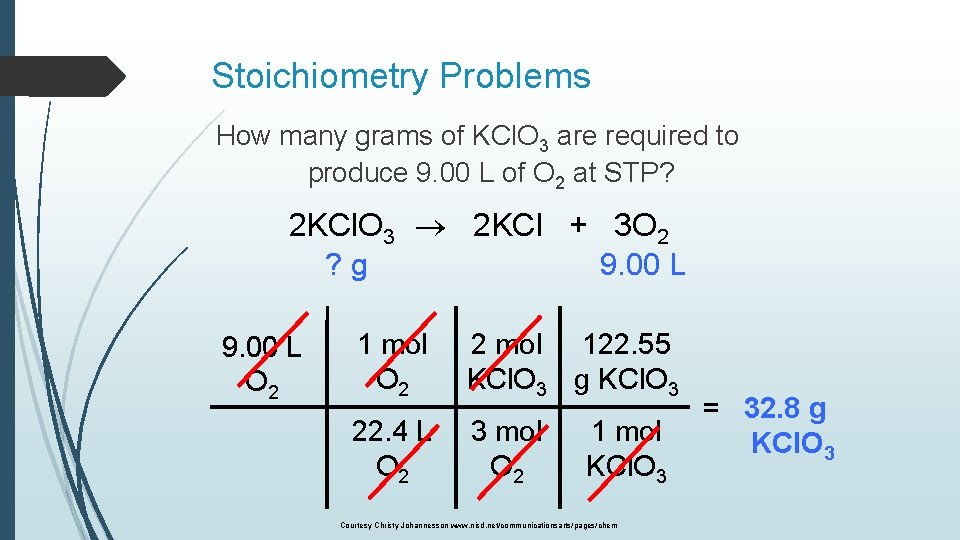
Stoichiometry Problems How many grams of KCl. O 3 are required to produce 9. 00 L of O 2 at STP? 2 KCl. O 3 2 KCl + 3 O 2 ? g 9. 00 L O 2 1 mol O 2 2 mol 122. 55 KCl. O 3 g KCl. O 3 22. 4 L O 2 3 mol O 2 1 mol KCl. O 3 Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem = 32. 8 g KCl. O 3

Stoichiometry Problems How many grams of KCl. O 3 are required to produce 9. 00 L of O 2 at STP? 2 KCl. O 3 2 KCl + 3 O 2 ? g 9. 00 L x g KCl. O 3 = 9. 00 L O 2 1 mol O 2 22. 4 L O 2 2 mol KCl. O 3 122. 55 g KCl. O 3 = 32. 8 g KCl. O 3 3 mol O 2 1 mol KCl. O 3 Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem 32. 8 g

How many grams of KCl. O 3 are required to produce 9. 00 L of O 2 at STP? 2 KCl. O 3 2 KCl + 3 O 2 ? g 9. 00 L O 2 1 mol O 2 2 mol 122. 55 KCl. O 3 g KCl. O 3 22. 4 L O 2 3 mol O 2 x g KCl. O 3 = 9. 00 L O 2 1 mol O 2 22. 4 L O 2 1 mol KCl. O 3 = 32. 8 g KCl. O 3 32. 8 g 2 mol KCl. O 3 122. 55 g KCl. O 3 = 32. 8 g KCl. O 3 3 mol O 2 1 mol KCl. O 3 Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
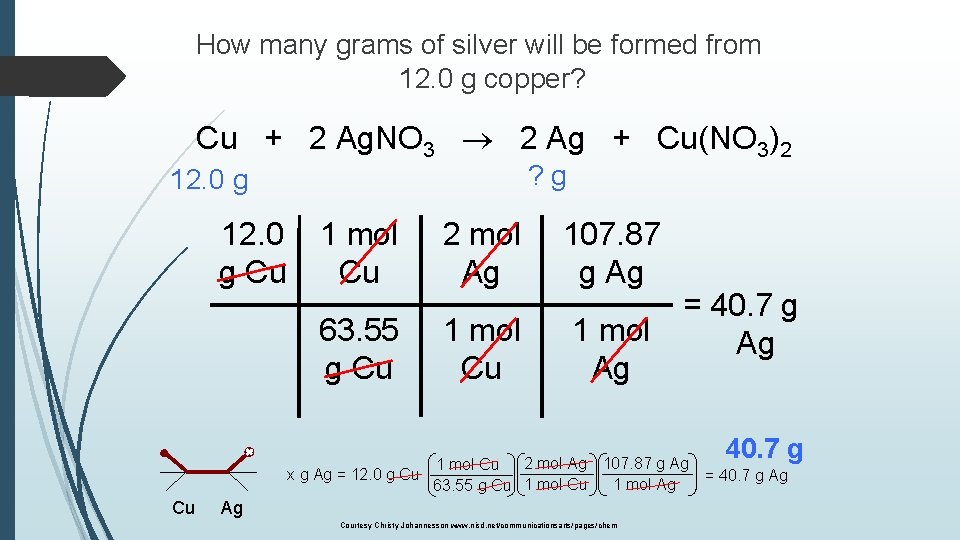
How many grams of silver will be formed from 12. 0 g copper? Cu + 2 Ag. NO 3 2 Ag + Cu(NO 3)2 ? g 12. 0 g Cu 1 mol Cu 2 mol Ag 107. 87 g Ag 63. 55 g Cu 1 mol Ag = 40. 7 g Ag 2 mol Ag 107. 87 g Ag 1 mol Cu x g Ag = 12. 0 g Cu 1 mol Ag 63. 55 g Cu 1 mol Cu Cu Ag Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem 40. 7 g = 40. 7 g Ag

Calculating Limiting Reactants STEPS: 1. Balance chemical reaction 2. Choose a reactant and calculate the amount of product. 3. Choose the other reactant and calculate the amount of product. 4. Whichever reactant makes LESS product, that is your limiting reactant. 5. The LEAST amount of product produced from Steps 3 and 4, is the amount theoretically produced by the reaction.
- Slides: 19