Unit 8 Solution Chemistry and Acids and Bases

Unit 8: Solution Chemistry and Acids and Bases

Lesson 85: Pass the Proton Acid-Base Theories

Chem. Catalyst Which of these four solutions conduct electricity: 0. 10 M HCl (hydrochloric acid), 0. 10 M CH 3 COOH (acetic acid), 0. 10 M Na. Cl (sodium chloride), 0. 10 M C 12 H 22 O 11 (sugar)? Explain. The first three solutions conduct, the fourth does not. The first three will dissociate into ions, which have charges. When these are flowing, the solution is able to conduct electricity. Sugar does not dissociate into ions.

Key Question How are acids and bases defined?

You will be able to: • define Arrhenius and Brønsted-Lowry acids and bases • explain the behavior of acids and bases on a particulate level • explain the difference between strong and weak acids and bases
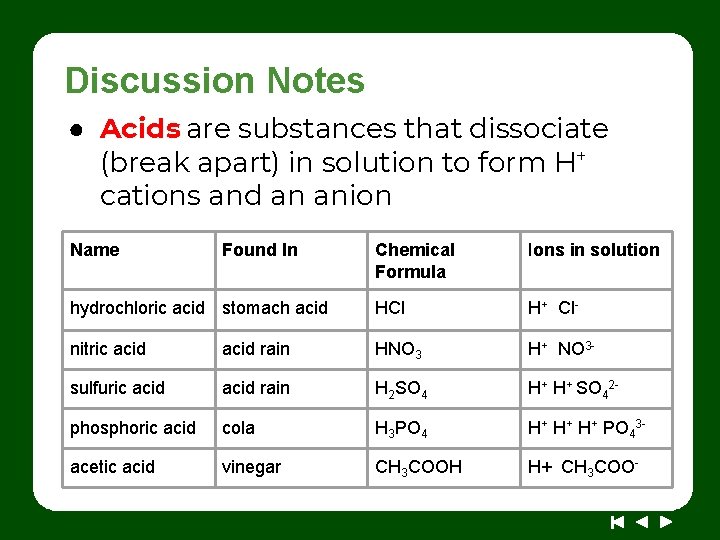
Discussion Notes ● Acids are substances that dissociate (break apart) in solution to form H+ cations and an anion Name Found In Chemical Formula Ions in solution hydrochloric acid stomach acid HCl H+ Cl- nitric acid rain HNO 3 H+ NO 3 - sulfuric acid rain H 2 SO 4 H+ H+ SO 42 - phosphoric acid cola H 3 PO 4 H+ H+ H+ PO 43 - acetic acid vinegar CH 3 COOH H+ CH 3 COO-

● Many bases contain a metal atom and a hydroxide ion, OH● Some compounds, such as ammonia and sodium carbonate, produce OH- ions when dissolved in water even though there is no OH- in the chemical formula Name Found In Chemical formula Ions in solution sodium hydroxide drain cleaner Na. OH Na+ (OH)- magnesium nydroxide antacid tablets Mg(OH)2 Mg 2+ (OH)- ammonia window cleaner NH 3 NH+4 (OH)-

Discussion Notes (cont. ) Arrhenius Definition of Acids and Bases: An acid is any substance that adds hydrogen ion (H+) to solution. A base is any substance that adds hydroxide ion (OH-) to solution. This definition does not explain how substances without hydroxide (OH-) in their formula, like ammonia (NH 3) can be bases. Therefore, a broader definition was developed

Discussion Notes (cont. ) Brønsted-Lowry Definition of Acids and Bases: An acid is a proton donor. A base is a proton acceptor. Acids and bases that break apart (dissociate) completely in solution are called strong acids and strong bases. Acids and bases that do not dissociate completely in solution are called weak acids and weak bases.
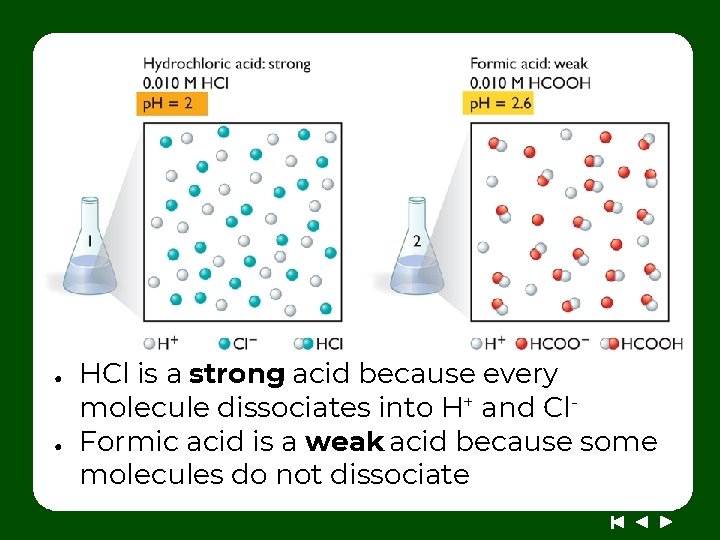
● ● HCl is a strong acid because every molecule dissociates into H+ and Cl. Formic acid is a weak acid because some molecules do not dissociate

Wrap Up How are acids and bases defined? • According to the Arrhenius definition, an acid is a substance that adds hydrogen ions, H+, to an aqueous solution. • According to the Arrhenius definition, a base is a substance that adds hydroxide ions, OH–, to an aqueous solution.

Wrap Up (cont. ) • Some substances add OH– by removing H+ from water. Brønsted and Lowry define an acid as a proton (H+) donor and a base as a proton (H+) acceptor. • Strong acids and strong bases dissociate completely into ions, while weak acids and weak bases do not.

Check-In Which substances do you expect will conduct electricity: hydrocyanic acid, HCN; magnesium hydroxide, Mg(OH)2; methanol, CH 3 OH? Explain your thinking.

Homework Questions 1) 2) 3) a. b. c. d. e. According to the Arrhenius theory, what is an acid and what is a base? What is the difference between a strong acid and a weak acid? Label each substance as either an acid or base. List the ions you would find in solution hydroiodic acid, HI formic acid, HCOOH rubidium hydroxide, Rb. OH hypochlorous acid, HOCl calcium hydroxide, Ca(OH)2

Homework Questions 4) Consider a solution of magnesium hydroxide, Mg(OH)2 a. If you draw a particle view of this base with 10 Mg 2+ ions, how many OH- ions would you need? 2+ ions b. Sketch a particle view with 10 Mg
- Slides: 15