Unit 8 Acids and Bases Arrhenius Acids Svante

Unit 8: Acids and Bases

Arrhenius Acids • Svante Arrhenius: Scientist who proposed a theory to explain the properties of acids and bases • Arrhenius Acid: Any substance that gives up a hydrogen ion H+(H+ or proton) in solution Cl. HCl + HCl H+ Cl-

Characteristic of Acids • To be an acid, it must be dissolved in water (aq) • When acids dissolve in water, they break apart to form FREELY MOVING IONS. • Ionization: when a covalent compound breaks into ions in water due to the high polarity (ex) HCl • Dissociation: when an ionic compound breaks into ions in water (ex) HNO 3 • Any compound that dissolves in water to form ions will conduct electricity or is called an electrolyte. • Electrolytes are all: ACIDS, BASES, AND SALTS (IONIC SUBSTANCES)

Characteristic of Acids • Acids react with metals (those above H 2 on Table J) to produce H 2 gas (single replacement reaction) • (ex) Mg(s) + 2 HCl Mg. Cl 2 + H 2(g) • (ex) 2 Al(s) + 6 HBr • Acids taste sour like citric acid and vinegar
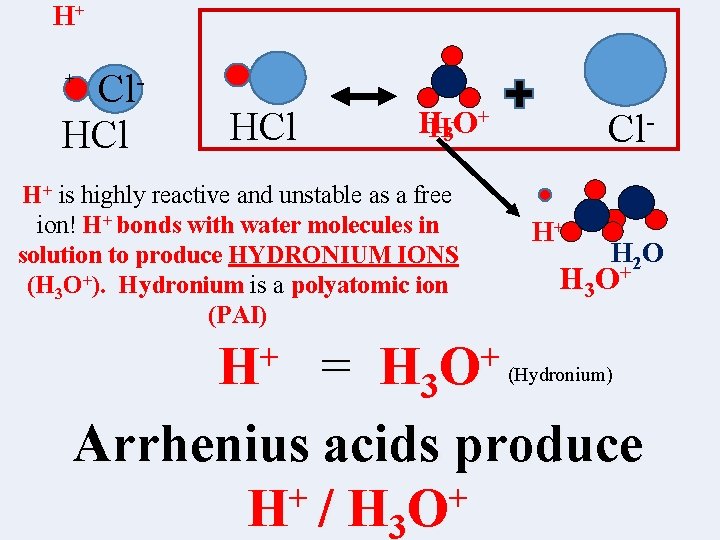
H+ Cl. HCl + HCl HH 3+O+ H+ is highly reactive and unstable as a free ion! H+ bonds with water molecules in solution to produce HYDRONIUM IONS (H 3 O+). Hydronium is a polyatomic ion (PAI) + H Cl. H+ H 2 O H 3 O + + H 3 O (Hydronium) = Arrhenius acids produce + + H / H 3 O
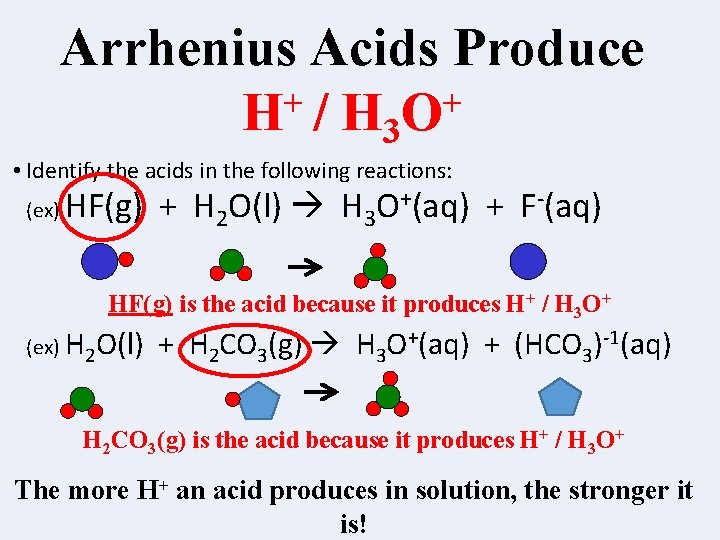
Arrhenius Acids Produce + + H / H 3 O • Identify the acids in the following reactions: (ex) HF(g) + H 2 O(l) H 3 O+(aq) + F-(aq) HF(g) is the acid because it produces H+ / H 3 O+ (ex) H 2 O(l) + H 2 CO 3(g) H 3 O+(aq) + (HCO 3)-1(aq) H 2 CO 3(g) is the acid because it produces H+ / H 3 O+ The more H+ an acid produces in solution, the stronger it is!
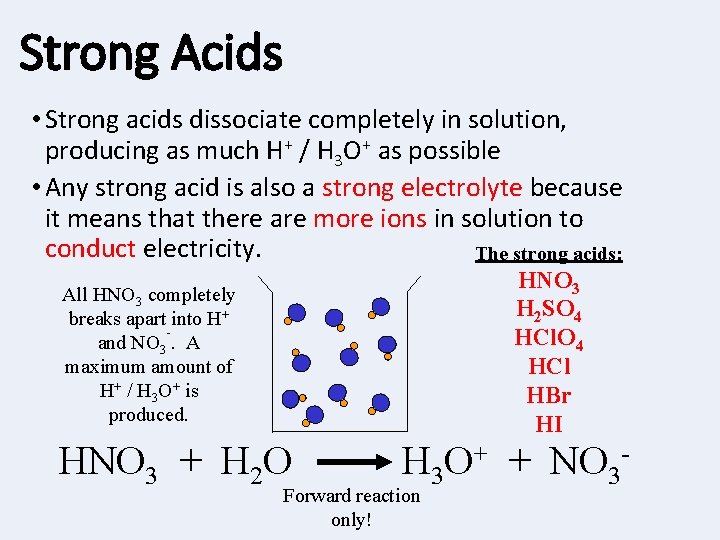
Strong Acids • Strong acids dissociate completely in solution, producing as much H+ / H 3 O+ as possible • Any strong acid is also a strong electrolyte because it means that there are more ions in solution to conduct electricity. The strong acids: HNO 3 H 2 SO 4 HCl HBr HI All HNO 3 completely breaks apart into H+ and NO 3. A maximum amount of H+ / H 3 O+ is produced. HNO 3 + H 2 O H 3 O+ + NO 3 - Forward reaction only!

Weak Acids • Weak acids do not completely break into ions in water. Some of it stays together in compound form. • Weak acids are still good electrolytes because there are still ions in solution to conduct electricity. Weak Acids: • All other acids not including the strong ones Only some CH 3 COOH breaks apart into H+. The rest stays together in compound form. CH 3 COOH + H 2 O (HC 2 H 3 O 2) • Organic acids -COOH H 3 O+ + CH 3 COO- Forward and Reverse Reactions Occur!

Acids and the Reference Table Strong Acids!!

Arrhenius Bases • Arrhenius Base: Any substance that gives up a hydroxide ion (OH)- in solution Characteristics of Bases: • To be a base, it must be dissolved in water (aq) • All bases are ionic and therefore dissociate in water to form FREELY MOVING IONS. Na. OH Na+ OH-
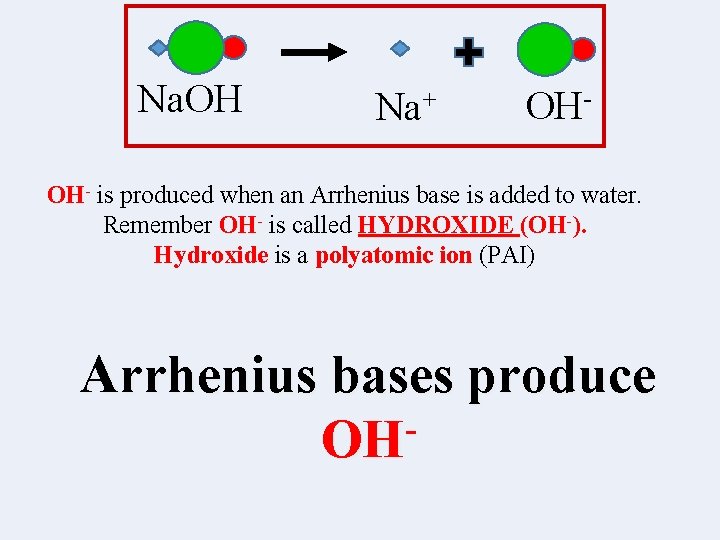
Na. OH Na+ OH- is produced when an Arrhenius base is added to water. Remember OH- is called HYDROXIDE (OH-). Hydroxide is a polyatomic ion (PAI) Arrhenius bases produce OH

Characteristics of Bases • Bases are electrolytes. • Solutions of bases feel slippery like soap and cleaners. • Bases taste bitter like soap and cleaners. Identify the bases in the following reactions: (ex) Na. OH(s) + H 2 O(l) Na+(aq) + OH-(aq) Reason: Na. OH because it forms OH- when with water (ex) Ca(OH)2(s) + H 2 O(l) Ca+2(aq) + 2 OH-(aq) Reason: Ca(OH)2 because it forms OH- when with water (ex) NH 3(g) + H 2 O(l) NH 4+(aq) + OH-(aq) Reason: NH 3 because it forms OH- when with water

Strong Bases • Strong Bases: those bases that completely dissociate in water producing a maximum amount of OH- in solution. • Bases formed from group 1 and 2 metals combining with hydroxide. • Li. OH, Na. OH, Mg(OH)2, KOH, Ca(OH)2 , etc.

Weak Bases • Weak Bases: those bases that do not completely dissociate in water. • All metals not in group 1 or group 2 bonded with OH • Al(OH)3, Fe(OH)3 , Cr(OH)4 , Ti. OH, etc, • Ammonium PAI with hydroxide, ammonia • NH 4 OH, NH 3
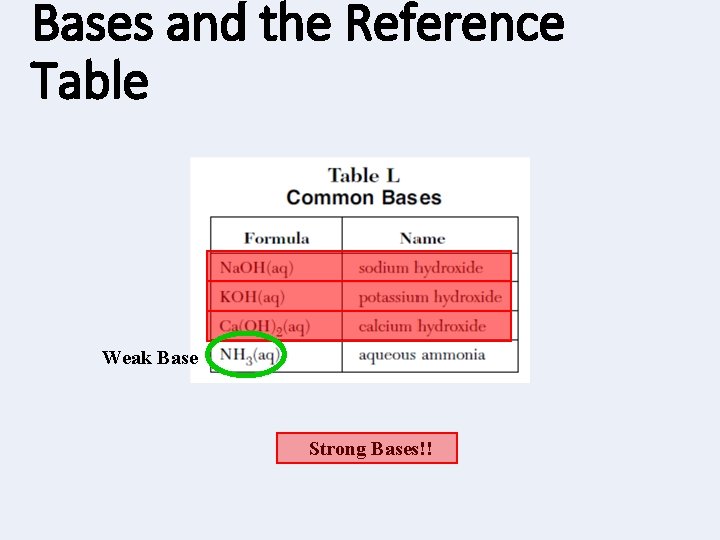
Bases and the Reference Table Weak Base Strong Bases!!

Naming Acids – Binary Acids • Binary Acids: H and a non-metal atom • Name the hydrogen in the acid hydro • Name the anion and change the ending of the name to –ic acid (ex) HCl(aq) (ex) HI (aq) (ex) HBr (aq) (ex) H 2 S (aq) Hydrochloric acid Hydroiodic acid Hydrobromic acid Hydrosulfuric acid

Naming Acids – Ternary Acids -ATE -IC, -ITE -OUS • Ternary Acids have PAI’s!!: • Go to Table E and determine the PAI • If the PAI ends in -ate , replace the -ate with -ic acid” • If the PAI ends in -ite , replace the -ite with -ous acid” (ex) H 2 CO 3(aq) (ex) HNO 2(aq) (ex) H 3 PO 4(aq) (ex) H 2 SO 3(aq) Carbonic acid Nitrous acid Phosphoric acid Sulfurous acid “ “

Naming Bases • Bases: Most bases are metal–hydroxides and so are ionic compounds therefore they are named just like ionic compounds. • Name 1 st element • Use roman numeral if the metal has more than one charge listed • 2 nd half of the name will always be hydroxide. Sodium hydroxide (ex) Na. OH(aq) (ex) Ca(OH)2(aq) Calcium hydroxide Iron (III) hydroxide (ex) Fe(OH)3(aq)

Indicators • Indicator: a substance that changes color if its in an acid or a base ** Most commonly used indicators: Acid Base Red • Litmus • Phenolphthalein (phth) Clear • Universal indicator ROY • Table M Blue Pink G BIV
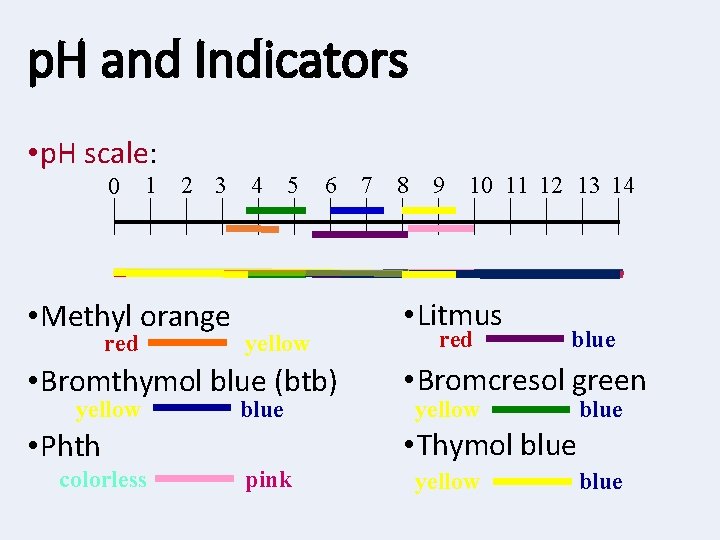
p. H and Indicators • p. H scale: 0 1 2 3 • Methyl orange red 4 5 6 yellow 7 8 9 10 11 12 13 14 • Litmus red blue • Bromthymol blue (btb) • Bromcresol green • Phth • Thymol blue yellow colorless blue pink yellow blue
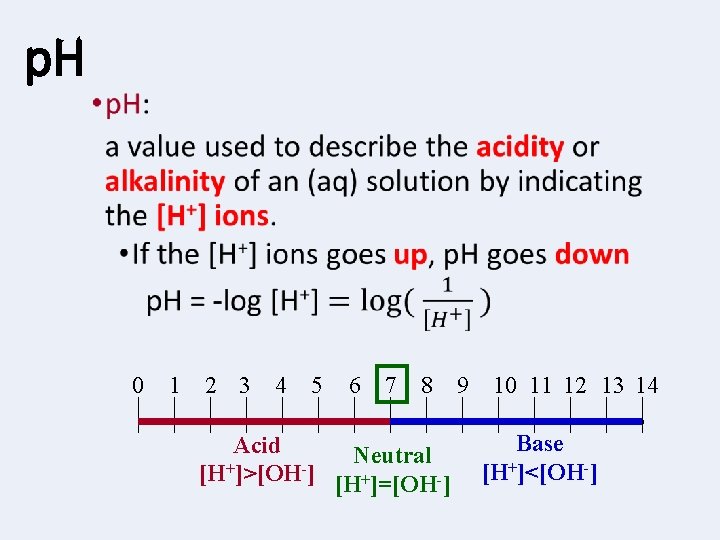
p. H • 0 1 2 3 4 5 6 7 8 Acid Neutral + [H ]>[OH ] [H+]=[OH-] 9 10 11 12 13 14 Base [H+]<[OH-]
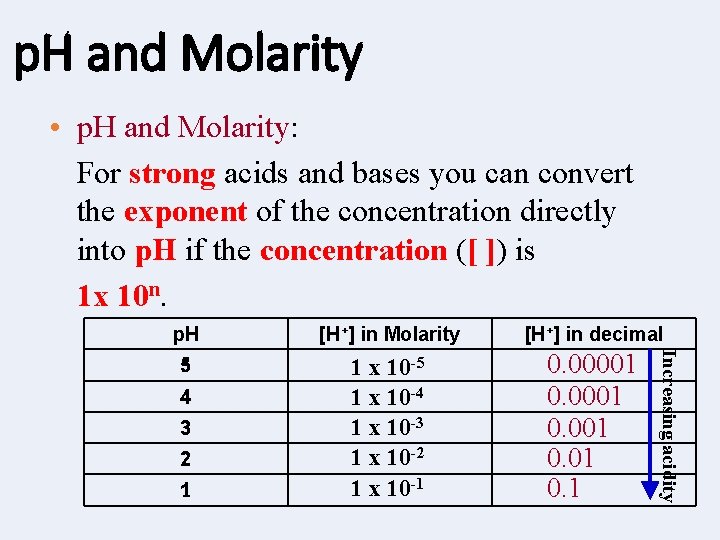
p. H and Molarity • p. H and Molarity: For strong acids and bases you can convert the exponent of the concentration directly into p. H if the concentration ([ ]) is 1 x 10 n. [H+] in Molarity [H+] in decimal 5 1 x 10 -4 1 x 10 -3 1 x 10 -2 1 x 10 -1 0. 00001 0. 1 4 3 2 1 Increasing acidity p. H

p. H and Molarity cont’d (ex) Calculate the p. H of a. 0001 M HCl (aq) solution. Is this an acid or base? (ex) Calculate the p. H of a solution whose concentration of H+ is 1 x 10 -10. (ex) Calculate the p. H of a solution whose concentration of H+ is 1 x 10 -1.
![p. OH • 0 1 2 3 4 Base [H+]<[OH-] 5 6 7 8 p. OH • 0 1 2 3 4 Base [H+]<[OH-] 5 6 7 8](http://slidetodoc.com/presentation_image_h2/4a2dfc2f961e1fe3319206a1946a6b9d/image-24.jpg)
p. OH • 0 1 2 3 4 Base [H+]<[OH-] 5 6 7 8 Neutral [H+]=[OH-] 9 10 11 12 13 14 Acid [H+]>[OH-]
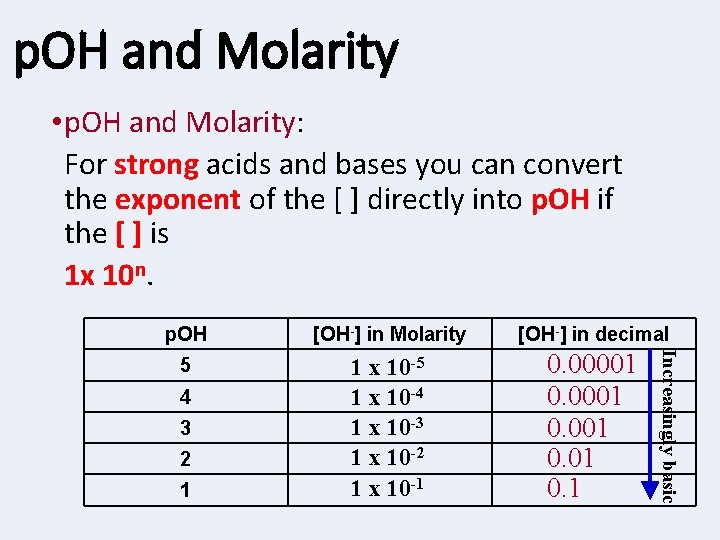
p. OH and Molarity • p. OH and Molarity: For strong acids and bases you can convert the exponent of the [ ] directly into p. OH if the [ ] is 1 x 10 n. [OH-] in Molarity [OH-] in decimal 5 1 x 10 -4 1 x 10 -3 1 x 10 -2 1 x 10 -1 0. 00001 0. 1 4 3 2 1 Increasingly basic p. OH

p. OH and Molarity cont’d (ex) Calculate the p. OH of a. 0001 M Na. OH (aq) solution. Is this an acid or a base? (ex) Calculate the p. OH of a solution whose concentration of OH- is 1 x 10 -10. (ex) Calculate the p. OH of a solution whose concentration of OH- is 1 x 10 -1.

p. H and p. OH p. H + p. OH always equals______ 14 (ex) If the p. H of a solution is 3, the p. OH is 11 ________. (ex) If the p. OH of a solution is 7, the p. H is ________. 7 [H+] p. H [OH-] p. OH Acid, base or neutral? 0. 0001 M 4 1. 0 x 10 -10 M 10 1. 0 x 10 -7 M 7 acid neutral 1. 0 x 10 -11 M 11 6 1. 0 x 10 -3 M base 1. 0 x 10 -8 M 3 8 9 1. 0 x 10 -5 M 5 base 1. 0 x 10 -6 M 1. 0 x 10 -9 M acid
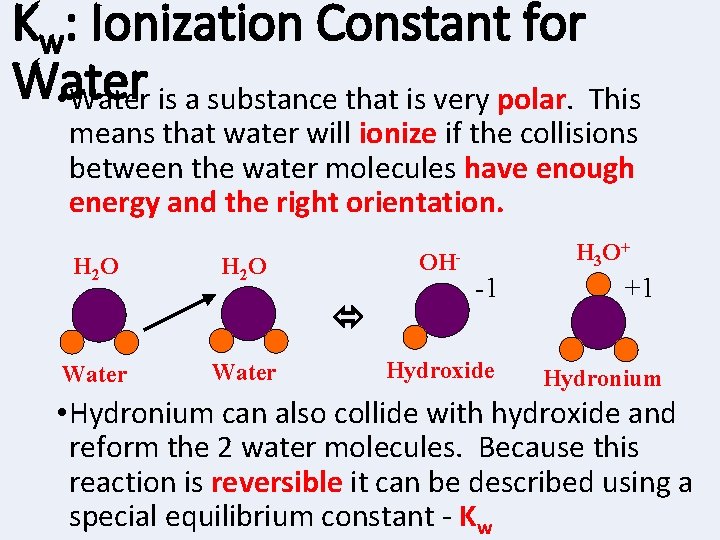
Kw: Ionization Constant for Water • Water is a substance that is very polar. This means that water will ionize if the collisions between the water molecules have enough energy and the right orientation. H 2 O OH- H 2 O Water H 3 O + -1 Hydroxide +1 Hydronium • Hydronium can also collide with hydroxide and reform the 2 water molecules. Because this reaction is reversible it can be described using a special equilibrium constant - Kw

Kw cont’d • The equation for the self-ionization of water is: H 2 O(l) + H 2 O(l) H 3 O+(aq) + OH-(aq) At STP, Kw = [H+][OH-] = 1 x 10 -14 • We can use the Kw to calculate the [H+] or [OH-].
![Kw cont’d • Therefore, in pure water at 273 K, the [H 3 O+] Kw cont’d • Therefore, in pure water at 273 K, the [H 3 O+]](http://slidetodoc.com/presentation_image_h2/4a2dfc2f961e1fe3319206a1946a6b9d/image-30.jpg)
Kw cont’d • Therefore, in pure water at 273 K, the [H 3 O+] and [OH-] are each 1 x 10 -7 M. Kw = [H+][OH-] = (1 x 10 -7) = 1 x 10 -14 **this is because pure water is neutral, therefore [H 3 O+]=[OH-]. • Remember, Kw means that there is an equilibrium. That means if the [ ] of one thing changes, everything else also changes. ** As [H 3 O+] goes up, [OH-] goes down.

Bronsted-Lowry Theory • This theory DOES NOT replace the Arrhenius theory, it only broadens it to include more substances as bases. THE DEFINITION OF AN ACID IS EXACTLY THE SAME!! • For example, NH 3 (aq) and Na 2 CO 3 (aq) are two compounds that act like bases, however they do not contain OH- ions. Instead they steal a H+ from water to FORM an OH-. NH 3(g) + H 2 O(l) NH 4+(aq) + OH-(aq)

Bronsted-Lowry Theory cont’d • With Bronsted and Lowry it’s all about the H+ (proton). And we always look at the reverse reaction as well. • B-L Acid: Any substance that donates an H+ + Any substance that accepts (steals) an H • B-L Base: (ex) HCl (aq) + H 2 O H 3 O+ (aq) + Cl- (aq) (ex) NH 3 (aq) + H 2 O NH 4+ (aq) + OH- (aq)
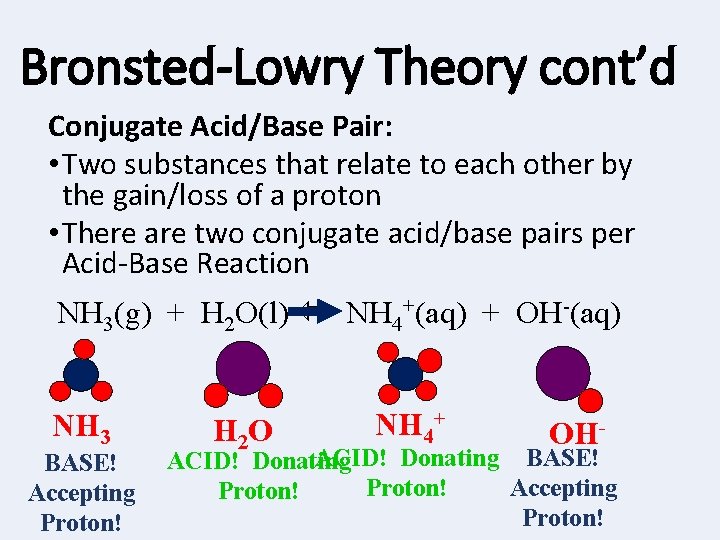
Bronsted-Lowry Theory cont’d Conjugate Acid/Base Pair: • Two substances that relate to each other by the gain/loss of a proton • There are two conjugate acid/base pairs per Acid-Base Reaction NH 3(g) + H 2 O(l) NH 4+(aq) + OH-(aq) NH 3 BASE! Accepting Proton! H 2 O NH 4+ OH- ACID! Donating BASE! ACID! Donating Accepting Proton!

Conjugate Acids and Bases • Conjugate Acid: the particle formed when a base gains an H+ ion. • Conjugate Base: the particle formed when an acid loses an H+ ion. An acid and its conjugate base are always on opposite sides of the reaction. A base and its conjugate acid are always on opposite sides of the reaction.

B-L Practice (ex) NH 3 (aq) + H 2 O NH 4+ (aq) + OH- (aq) (ex) HNO 2 (aq) + H 2 O H 3 O+ (aq) + NO 2 - (aq) (ex) HS- (aq) + NH 3 (aq) NH 4+ (aq) + S-2 (aq)

Acid/Base Neutralization • Neutralization: When equal molar quantities of an acid and a base are mixed, a neutral solution is produced. *For every 1 mole of OH- ions there needs to be 1 mole of H+ ions. • Products from mixing an acid and a base are always water and a salt. • Salt: any ionic compound that is NOT an acid or base • The products always have a p. H of 7 and [OH-] and [H+] are equal.

Practice with Neutralization **Neutralization rxns are double replacement. • If the reactants are equal volumes of a strong acid and a strong base p. H = 7 • If the reactants are equal volumes of a strong acid and a weak base p. H < 7 • If the reactants are equal volumes of a weak acid and a strong base p. H > 7

Acid/Base Titrations • Titration: a method used in the lab to determine the concentration of an unknown acid or base. • A solution of known concentration (standard sol’n) is added to a solution of unknown concentration until it is completely neutral. • The point at which neutralization occurs is called the endpoint. • The endpoint can be found by adding an indicator to the unknown.
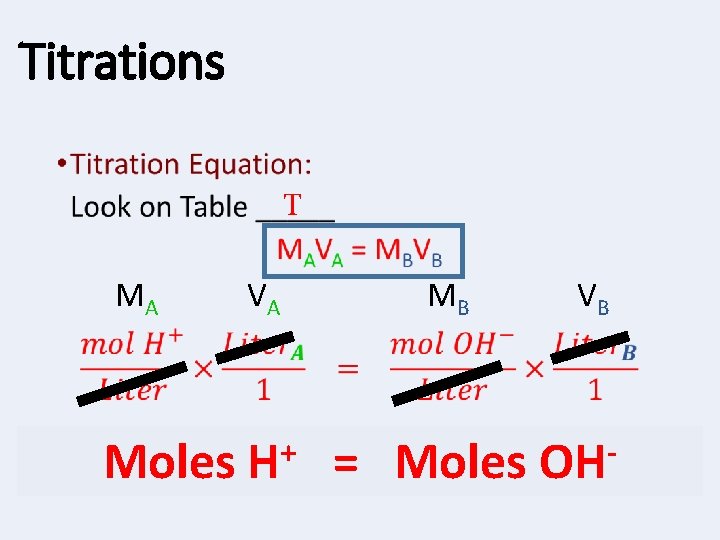
Titrations • T MA Moles VA + H MB = Moles VB OH

Titration Practice • (ex) What volume of 2. 0 M HNO 3 is needed to neutralize 40. m. L of 5. 0 M Na. OH? • (ex) You need 20 m. L of 2 M HCl, however the stock solution is 12 M. How many m. L of the stock solution do you need to make your final solution? • (ex) There is 3 moles of HBr in a solution. How many m. L of 2. 5 M Na. OH need to be added to neutralize the solution?
- Slides: 40