Unit 7 The Periodic Table Card Activity 10

Unit 7: The Periodic Table

Card Activity 10 pts 1. How did your group decide to arrange the cards? (what patterns are you noticing) 2. Have you changed the arrangement with new discovered cards? 3. Where did you place 2 diamonds? Why? 4. What is the trend going down the columns (family/group)? 5. What is the trend going across the rows (period)? 6. Can you predict what cards may exist, but not yet discovered? 7. How do you believe this activity is related to the development of the periodic table?

How a card game inspired Professor Mendeleev • Solitarie (German- 1783) • Cards are arranged by suit horizontally (periods on the table) • By number vertically (groups on the table)

The Mystery of Matter: Search for the Elements Episode 2: Unruly Elements (Part 1)

Pre- Mendeleev • Key Concept 1: In the 1700 s, the first list compiled all the known elements of the time; they were grouped into Gases, Metals, Nonmetals, and Earths.

Dmitri Mendeleev 1834 -1906

Dmitri Mendeleev 1834 -1906 • Key Concept 2: Mendeleev was a Russian chemist who created a farsighted version of the periodic table of elements. • He used it to correct the properties of some already discovered elements and also to predict the properties of eight elements yet to be discovered.

Dmitri Mendeleev 1834 -1906 • One of 14 kids or one of 17 kids there are two different stories, but he was the youngest. • Known for his crazy hair and beard that he only trimmed once a year. • Taught an intro level Chemistry course at St. Petersburg University.

Dmitri Mendeleev 1834 -1906 • Unable to find a proper text for his students, he decided to write his own. • Principles of Chemistry- 1868. • Volume 1→ had only 9 of the 63 elements • Volume 2→ needed to have the other 54

Dmitri Mendeleev 1834 -1906 • Mendeleev began to toy with the elements to try to place them in a table. • The elements were currently arranged based on atomic weight Or by its common properties. • Key Concept 3: Mendeleev was determined to combine the two ways of classifying elements (atomic weight & properties).

“An Attempt at a System of Elements, Based on Their Atomic Weight and Chemical Affinity” • “Because the properties repeated themselves periodically, the invention became known as the periodic table”.
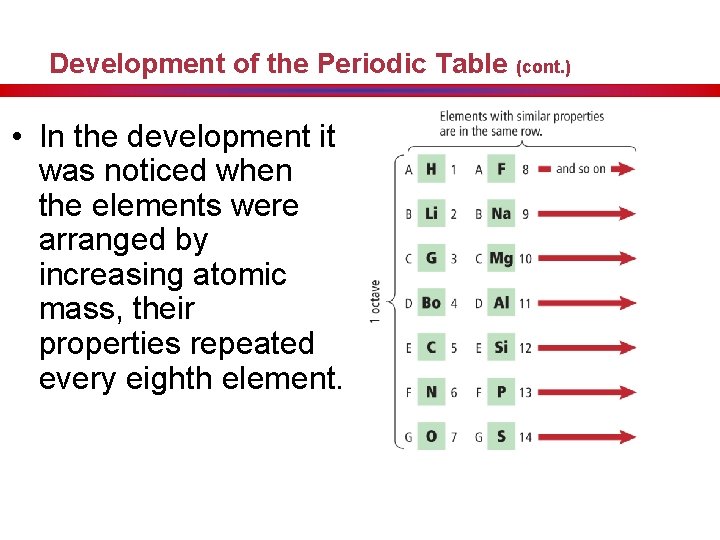
Development of the Periodic Table (cont. ) • In the development it was noticed when the elements were arranged by increasing atomic mass, their properties repeated every eighth element.

Development of the Periodic Table (cont. ) • He left gaps in the table predicting that new elements would be found.


Major Changes Post- Mendeleev to P-Table • Ramsay's 1894 discovery of Argon and Helium. • Mendeleev did not predict nor leave room for these elements on the periodic table and therefore disagreed with their discovery. • It wasn’t unit 1903 after years of research that Mendeleev agreed with Ramsay that there are inert gases. • He then created and added a new group called the Noble Gases.

Major Changes Post- Mendeleev to P-Table • Re-organize according to atomic number (protons) not weight (Thanks Moseley). • Based on IUPAC standards, there are currently 118 elements on the periodic table. This is after four new elements were added in July of 2016. • Discovery of the internal structure of the atom helped explain why properties repeated themselves periodically.

Key Concept 4: Learning Check How has the periodic table changed overtime? Why have these changes occurred?

End of Lesson 1
Importance of Tables
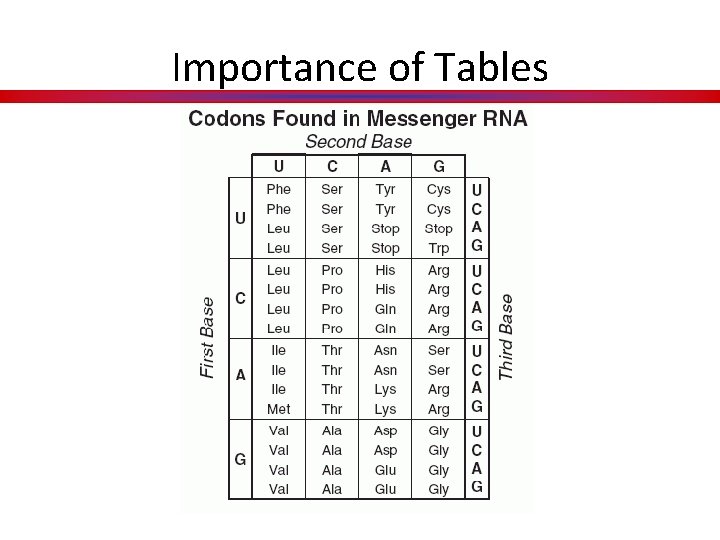
Importance of Tables
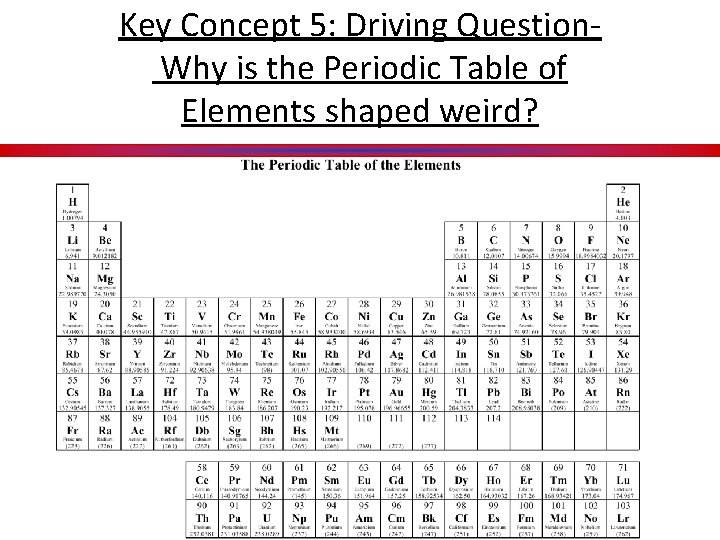
Key Concept 5: Driving Question Why is the Periodic Table of Elements shaped weird?
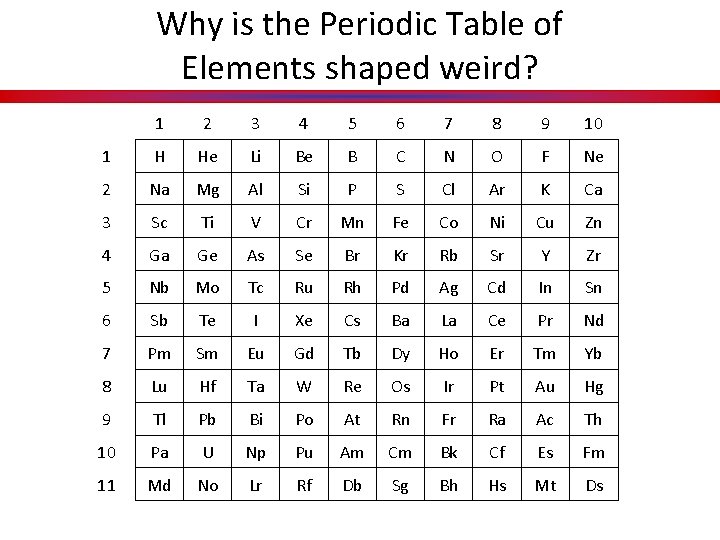
Why is the Periodic Table of Elements shaped weird? 1 2 3 4 5 6 7 8 9 10 1 H He Li Be B C N O F Ne 2 Na Mg Al Si P S Cl Ar K Ca 3 Sc Ti V Cr Mn Fe Co Ni Cu Zn 4 Ga Ge As Se Br Kr Rb Sr Y Zr 5 Nb Mo Tc Ru Rh Pd Ag Cd In Sn 6 Sb Te I Xe Cs Ba La Ce Pr Nd 7 Pm Sm Eu Gd Tb Dy Ho Er Tm Yb 8 Lu Hf Ta W Re Os Ir Pt Au Hg 9 Tl Pb Bi Po At Rn Fr Ra Ac Th 10 Pa U Np Pu Am Cm Bk Cf Es Fm 11 Md No Lr Rf Db Sg Bh Hs Mt Ds
Why is the Periodic Table of Elements shaped weird?

Why is the Periodic Table of Elements shaped weird? How is the periodic table arranged? • Atomic Number (Proton #) • NOT BY MASS

Why is the Periodic Table of Elements shaped weird? How is the periodic table arranged? • Key Concept 6: Columns of elements are called groups (families). • Key Concept 7: Rows of elements are called periods.
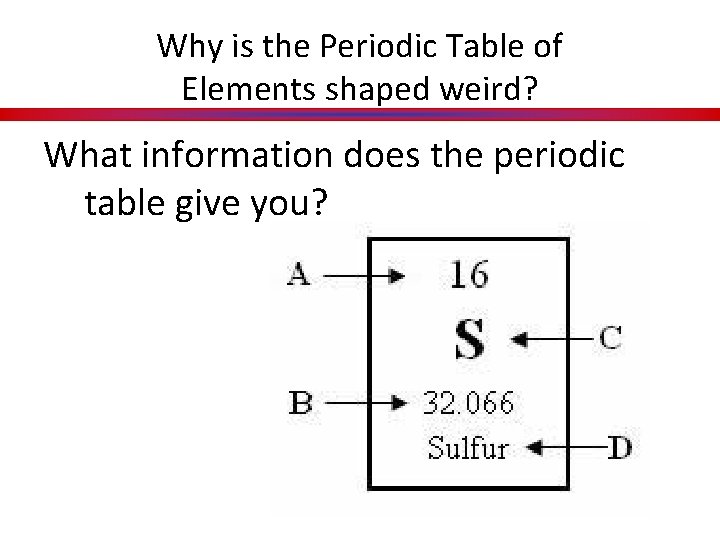
Why is the Periodic Table of Elements shaped weird? What information does the periodic table give you?


What’s in a name? Periodic • appearing or occurring at intervals. Table • a set of facts or figures systematically displayed of • expressing the relationship between a part and a whole Elements • substance that cannot be broken down by chemical means.

What’s in a name? Periodic Table of Elements The Periodic Table is a chart which arranges the chemical elements in a useful, logical manner. Key Concept 8: Elements are listed in order of increasing atomic number, lined up so that elements which exhibit similar properties are arranged in the same row or column as each other.
Would you want to shop here?

You wouldn’t want to shop here either? 1 2 3 4 5 6 7 8 9 10 1 H He Li Be B C N O F Ne 2 Na Mg Al Si P S Cl Ar K Ca 3 Sc Ti V Cr Mn Fe Co Ni Cu Zn 4 Ga Ge As Se Br Kr Rb Sr Y Zr 5 Nb Mo Tc Ru Rh Pd Ag Cd In Sn 6 Sb Te I Xe Cs Ba La Ce Pr Nd 7 Pm Sm Eu Gd Tb Dy Ho Er Tm Yb 8 Lu Hf Ta W Re Os Ir Pt Au Hg 9 Tl Pb Bi Po At Rn Fr Ra Ac Th 10 Pa U Np Pu Am Cm Bk Cf Es Fm 11 Md No Lr Rf Db Sg Bh Hs Mt Ds
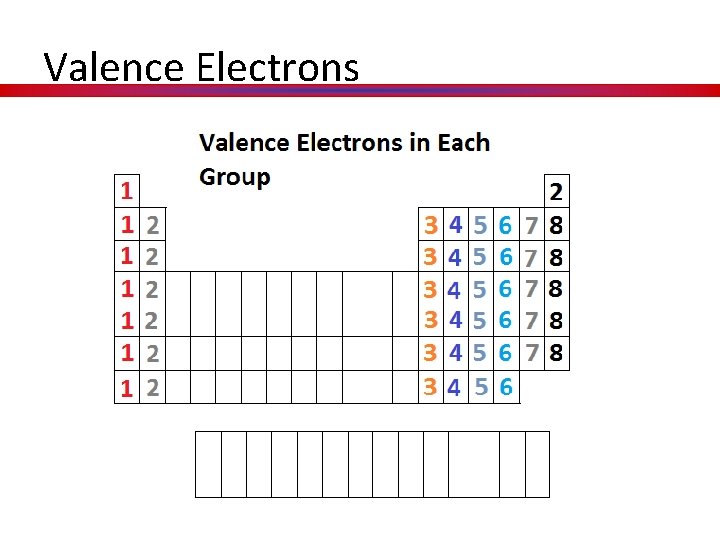
Valence Electrons
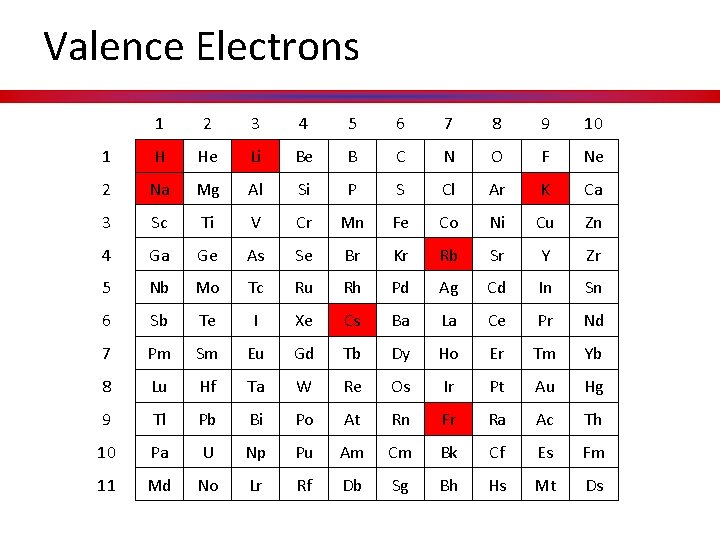
Valence Electrons 1 2 3 4 5 6 7 8 9 10 1 H He Li Be B C N O F Ne 2 Na Mg Al Si P S Cl Ar K Ca 3 Sc Ti V Cr Mn Fe Co Ni Cu Zn 4 Ga Ge As Se Br Kr Rb Sr Y Zr 5 Nb Mo Tc Ru Rh Pd Ag Cd In Sn 6 Sb Te I Xe Cs Ba La Ce Pr Nd 7 Pm Sm Eu Gd Tb Dy Ho Er Tm Yb 8 Lu Hf Ta W Re Os Ir Pt Au Hg 9 Tl Pb Bi Po At Rn Fr Ra Ac Th 10 Pa U Np Pu Am Cm Bk Cf Es Fm 11 Md No Lr Rf Db Sg Bh Hs Mt Ds
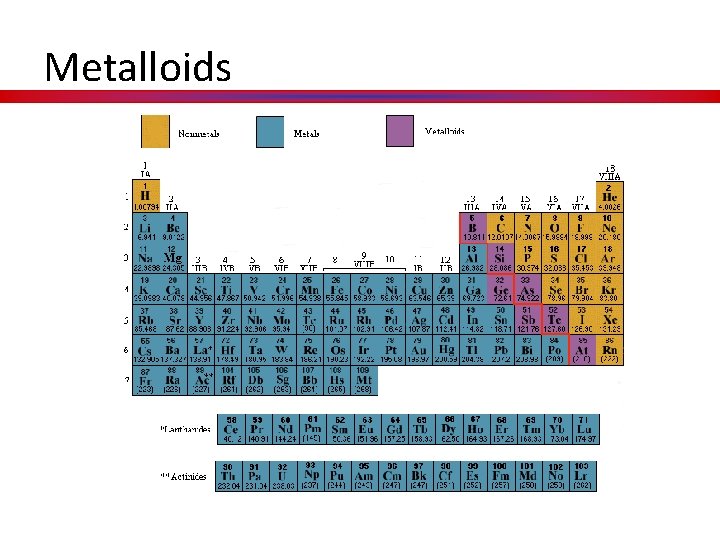
Metalloids
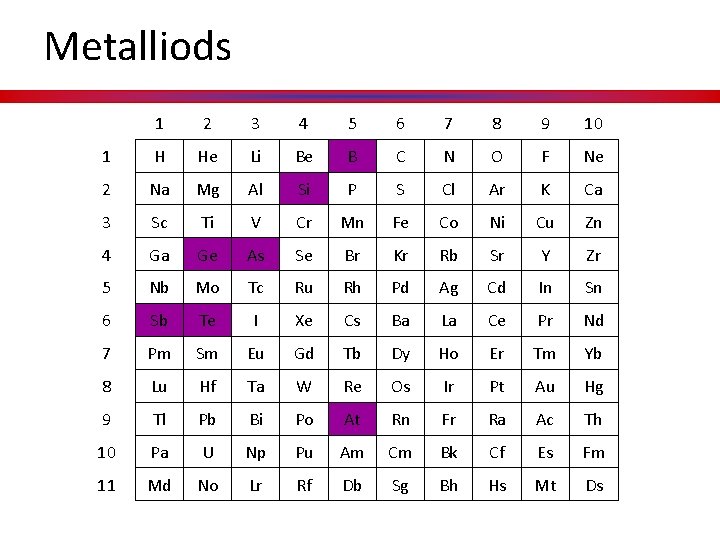
Metalliods 1 2 3 4 5 6 7 8 9 10 1 H He Li Be B C N O F Ne 2 Na Mg Al Si P S Cl Ar K Ca 3 Sc Ti V Cr Mn Fe Co Ni Cu Zn 4 Ga Ge As Se Br Kr Rb Sr Y Zr 5 Nb Mo Tc Ru Rh Pd Ag Cd In Sn 6 Sb Te I Xe Cs Ba La Ce Pr Nd 7 Pm Sm Eu Gd Tb Dy Ho Er Tm Yb 8 Lu Hf Ta W Re Os Ir Pt Au Hg 9 Tl Pb Bi Po At Rn Fr Ra Ac Th 10 Pa U Np Pu Am Cm Bk Cf Es Fm 11 Md No Lr Rf Db Sg Bh Hs Mt Ds
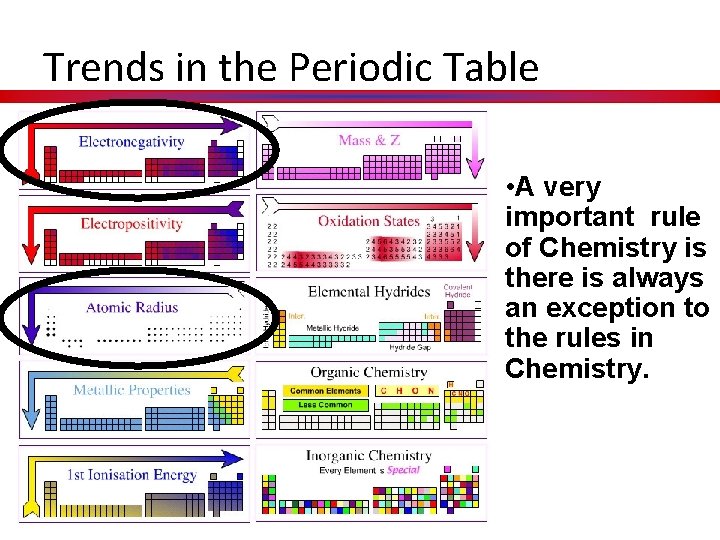
Trends in the Periodic Table • A very important rule of Chemistry is there is always an exception to the rules in Chemistry.
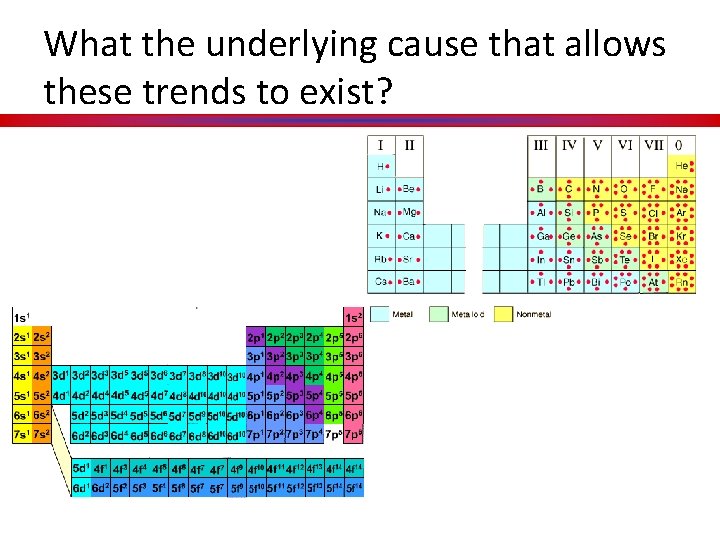
What the underlying cause that allows these trends to exist?

Development of the Periodic Table (cont. ) (KC-9) Answer to Key Concept 1: As the periodic table developed over time it took on it’s weird shape due to the electron configuration of each element discovered.

Key Concept 10: Learning Check 1. Breakdown the title and explain the name “Periodic Table of Elements”. 2. Why is the Periodic Table shaped weird?

The Element Song

Periodic Table is just a Treasure Map in Disguise

Let’s go on a Treasure Hunt

What information is on the p-table? • Columns of elements are called groups (families). • Rows of elements are called periods. • Elements in groups 1 A, 2 A, and 3 A-8 A possess a wide variety of chemical and physical properties and are called the representative elements. • Elements in groups “B” are known as the transition metals and inner transition metals.
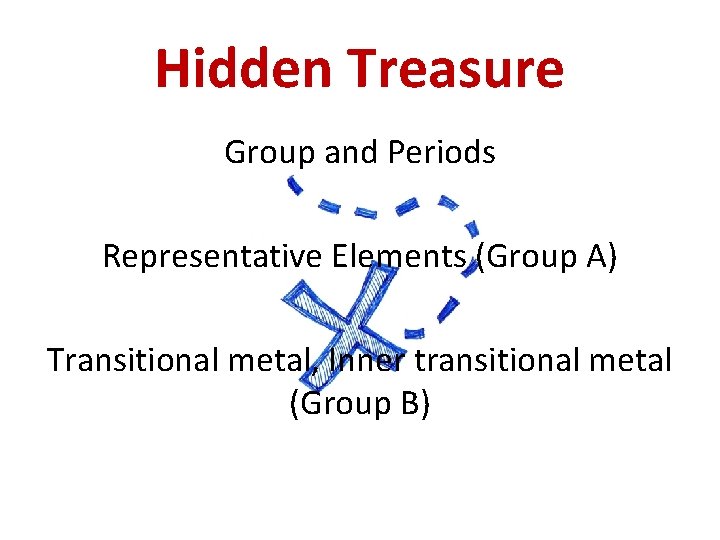
Hidden Treasure Group and Periods Representative Elements (Group A) Transitional metal, Inner transitional metal (Group B)

What information is on the p-table? • The two sets of inner transition metals are called the lanthanide series and actinide series and are located at the bottom of the periodic table. • How do you think they got their name?

Hidden Treasure Lanthanide series and actinide series

What information is on the p-table? • Atoms groups/ families have similar properties. WHY? • They are similar because they all have the same number of valence (outer shell) electrons, which governs their chemical behavior.

Hidden Treasure Valence Electrons
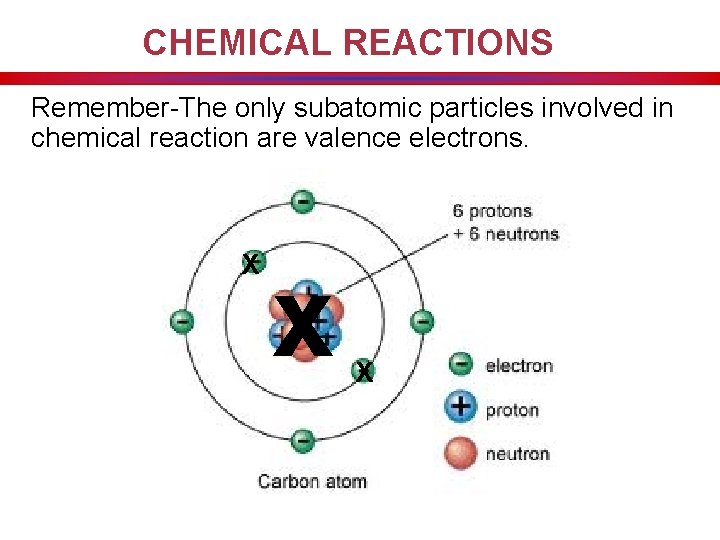
CHEMICAL REACTIONS Remember-The only subatomic particles involved in chemical reaction are valence electrons. X x X
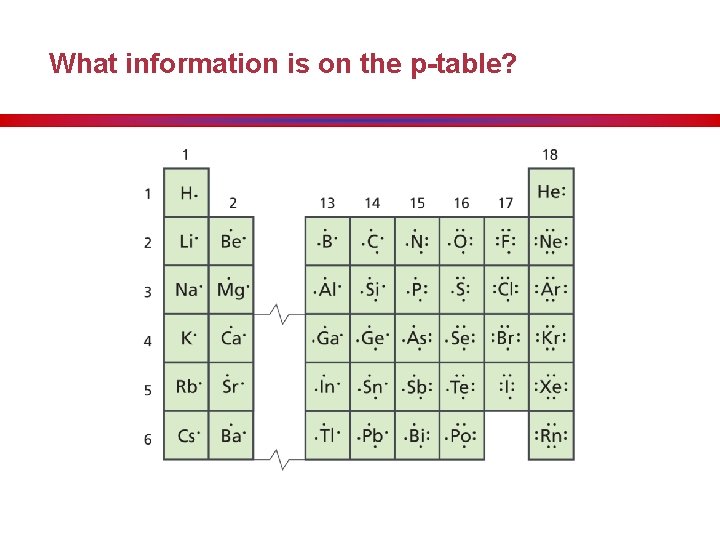
What information is on the p-table?
Hidden Treasure Lewis Dot Structures for Representative Elements WHY- Oxidation number exist
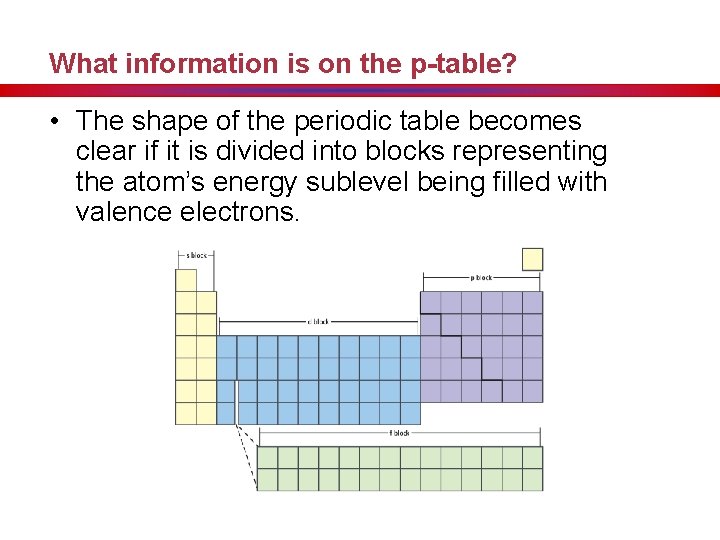
What information is on the p-table? • The shape of the periodic table becomes clear if it is divided into blocks representing the atom’s energy sublevel being filled with valence electrons.
Hidden Treasure S- Block P-Block D-Block F-Block Energy Levels of Electrons
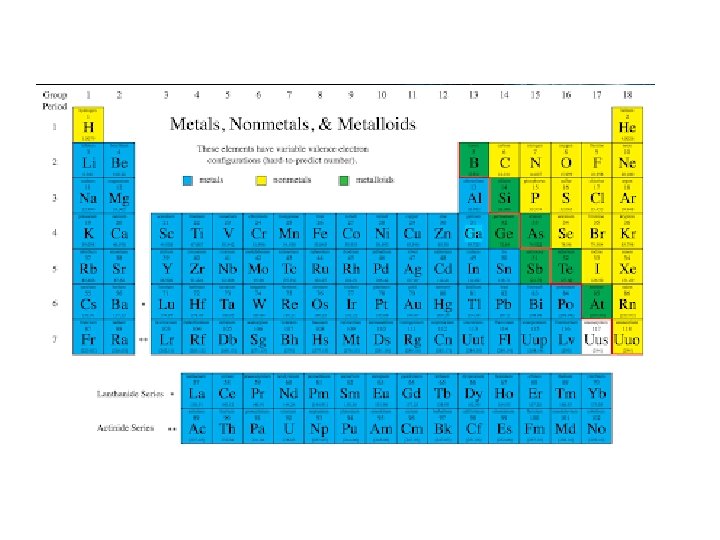

Metals • Metals are lustrous (shiny), malleable, ductile, and are good conductors of heat and electricity. • They are mostly solids at room temp. • What is one exception?

Metals There are two metal groups that you have to know there name. • Alkali metals are all the elements in group 1 A except hydrogen, and are very reactive. • Alkaline earth metals are in group 2 A, and are also highly reactive.

Nonmetals • Nonmetals are the opposite. • They are dull, brittle, nonconductors (insulators). • Some are solid, but many are gases, and Bromine is a liquid.

The Modern Periodic Table (cont. ) • There are two non-metal groups that you have to know the name of. • Group 7 A is composed of highly reactive elements called halogens. • Group 8 A gases are extremely unreactive and commonly called noble gases.

Metalloids • Metalloids, aka semimetals are just that. • They have characteristics of both metals and nonmetals. • They are shiny but brittle. • And they are semiconductors.
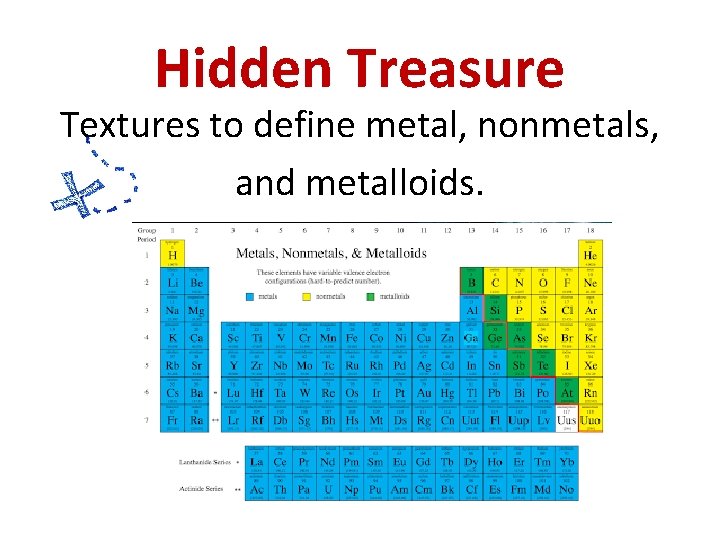
Hidden Treasure Textures to define metal, nonmetals, and metalloids.

Hidden Treasure Family Names Group 1 A, 2 A, 7 A, and 8 A
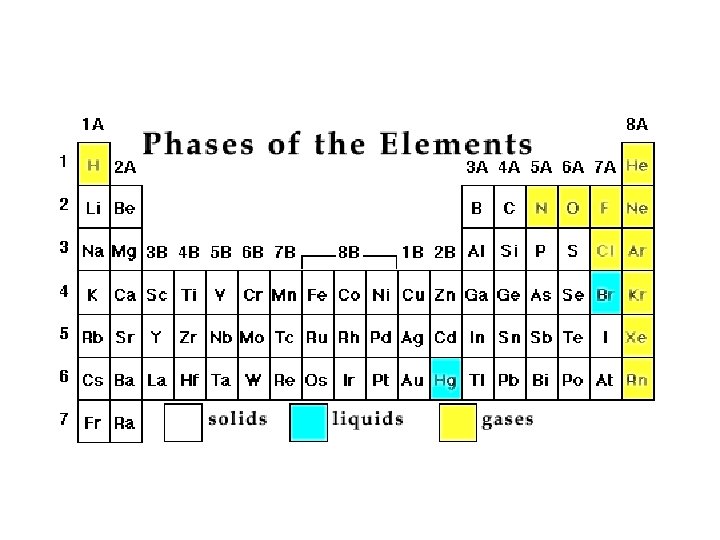

Hidden Treasure Solid, Liquids, and Gas at Room Temperature

Add Treasure/ Information • Group and Periods • Representative Elements (Group A) • Transitional metal, Inner transitional metal (Group B) – Lanthanide series and actinide series • Valence electrons, Lewis Dot Structure examples, Oxidation Numbers • s, p, d, & f blocks

Add Treasure/ Information • Metal/ Nonmetals/ Metalloids • 4 Family Names • Phases at Room Temperature

End of Lesson 2

Demo Time • Demo 1 • Demo 4
Hidden Treasure “F” Reactivity Tend

End of Lesson 3

Describe what you see in the cartoon.

Focus questions: 1. What does the word radius mean? 2. Draw a picture of a circle and indicate the radius.
Atomic Radius • Atomic size (radius) is a periodic trend influenced by electron configuration. • Key Concept 11: The outer limit of an electron cloud is defined as the spherical surface within which there is 90% probability of finding an electron. • Therefore atomic size is defined by how closely an atom lies to a neighboring atom.
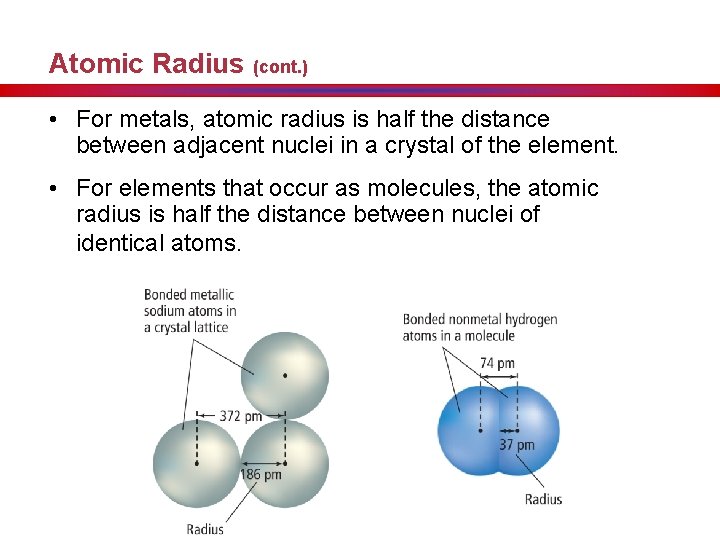
Atomic Radius (cont. ) • For metals, atomic radius is half the distance between adjacent nuclei in a crystal of the element. • For elements that occur as molecules, the atomic radius is half the distance between nuclei of identical atoms.

Focus Question: Atomic Radius (cont. ) 3. What elements do you think has the biggest radius. Please summarize your hypothesis in writing on your board. Hydrogen or Sodium 4. What elements do you think has the biggest radius. Please summarize your hypothesis in writing on your board. Sodium or Chlorine

Focus Question: Atomic Radius (cont. ) What if I told you that chlorine atomic radius is smaller than sodium. 5. With your partner come up with a hypothesis in writing why?
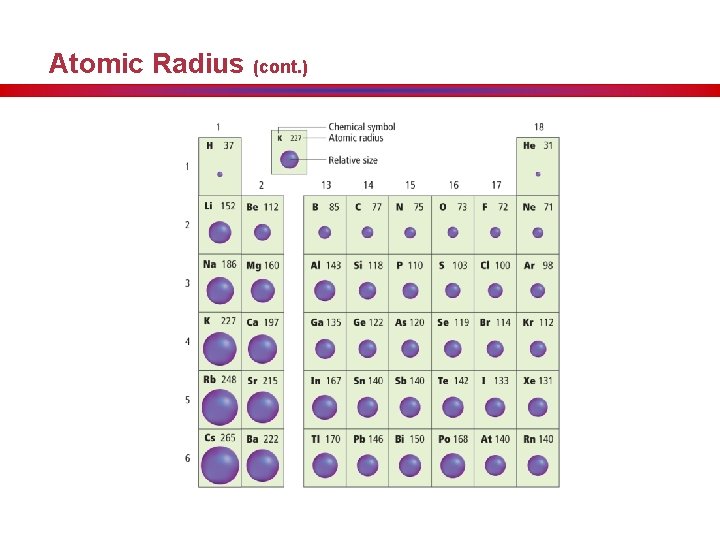
Atomic Radius (cont. )

On your Periodic Table: • Your arrows will always point in the increasing way. • You will be adding a total of 4 trends to your periodic table. Atomic Radius Ionization Energy Electronegativity M. P. and B. P.
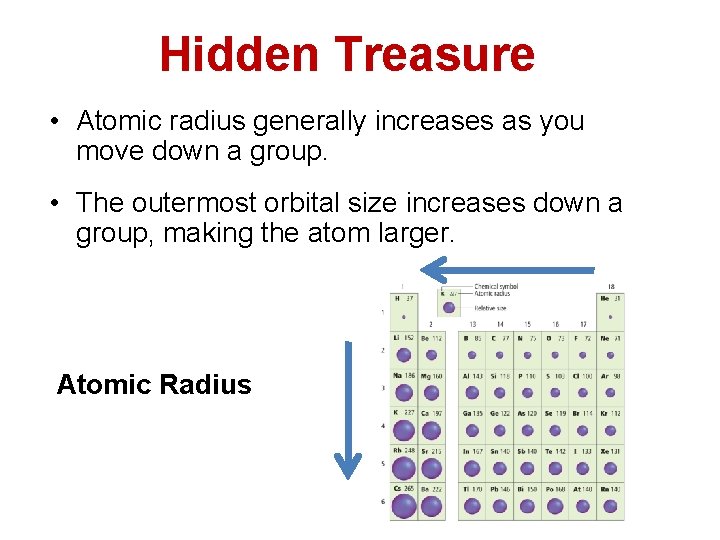
Hidden Treasure • Atomic radius generally increases as you move down a group. • The outermost orbital size increases down a group, making the atom larger. Atomic Radius

Focus Questions: Review 6. An atom that gains one or more electrons will have a __________ charge. 7. An atom that loses one or more electrons will have a __________ charge. 8. An atom that gains or loses one or more electrons is called an ______. 9. A positive ion is called a _______ and a negative ion is called an ________.

Ionic Radius • An ion is an atom or bonded group of atoms with a positive or negative charge. • Key Concept 12: When atoms lose electrons to form cation, they always become SMALLER for two reasons: 1. The loss of a valence electron can leave an empty outer orbital resulting in a small radius. 2. Electrostatic repulsion decreases (more p+ vs e-) allowing the electrons to be pulled closer to the radius.

Ionic Radius (cont. ) • When atoms gain electrons and becomes an anion, they can become LARGER, because the addition of an electron increases electrostatic repulsion.

Ionic Radius (cont. )

Ionic Radius (cont. ) Bigger or Smaller Beryllium Be 2+ Fluoride F- Oxygen O 2 - Boron B 3+ Carbon C 4 - C 4+ Carbon

Focus Questions: Ionization Energy • Key Concept 13: Ionization energy is defined as the energy required to remove an electron from a gaseous atom. • The energy required to remove the first electron is called the first ionization energy. Answer this question: 10. What group (alkali metals or noble gases) do you believe to have the highest ionization energy? WHY?

Ionization Energy (cont. ) • Removing the second electron requires more energy, and is called the second ionization energy. • Each successive ionization requires more energy, but it is not a steady increase.
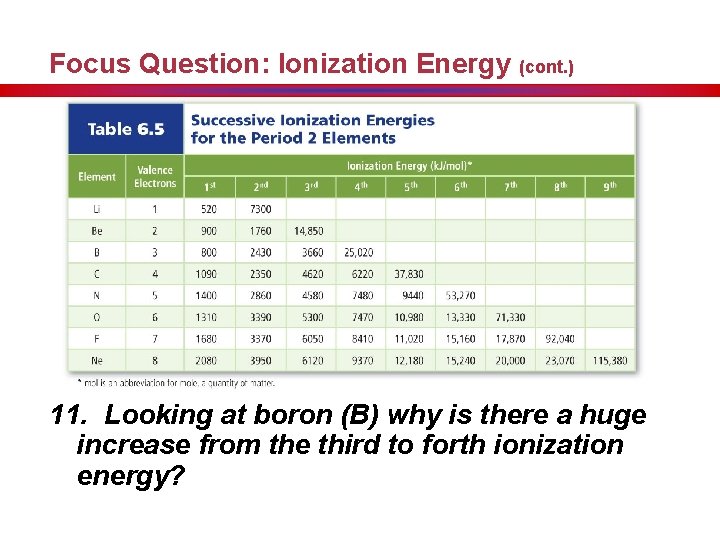
Focus Question: Ionization Energy (cont. ) 11. Looking at boron (B) why is there a huge increase from the third to forth ionization energy?

Ionization Energy (cont. ) • The ionization at which the large increase in energy occurs is related to the number of valence electrons. 12. Draw a large rectangle to represent the periodic table, then predict the trend of first ionization energy. • First ionization energy increases from left to right across a period. • First ionization energy decreases down a group because atomic size increases and less energy is required to remove an electron farther from the nucleus.
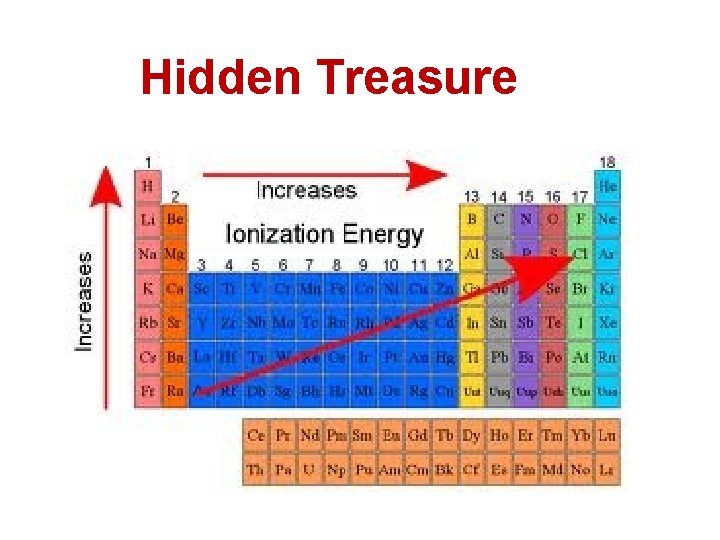
Hidden Treasure

Focus Questions: Electronegativity • Key Concept 14: The electronegativity of an element indicates its relative ability to attract electrons in a chemical bond. 13. What elements do you think has the highest electronegativity. Hydrogen or Sodium 14. What elements do you think has the highest electronegativity. Sodium or Chlorine
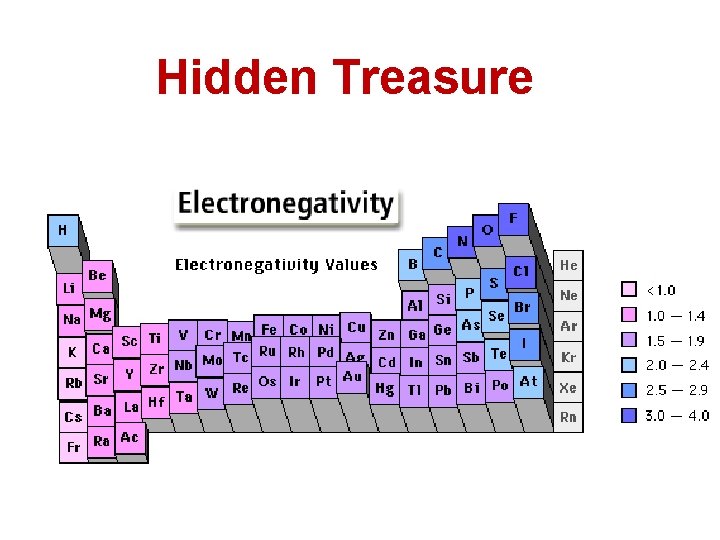
Hidden Treasure
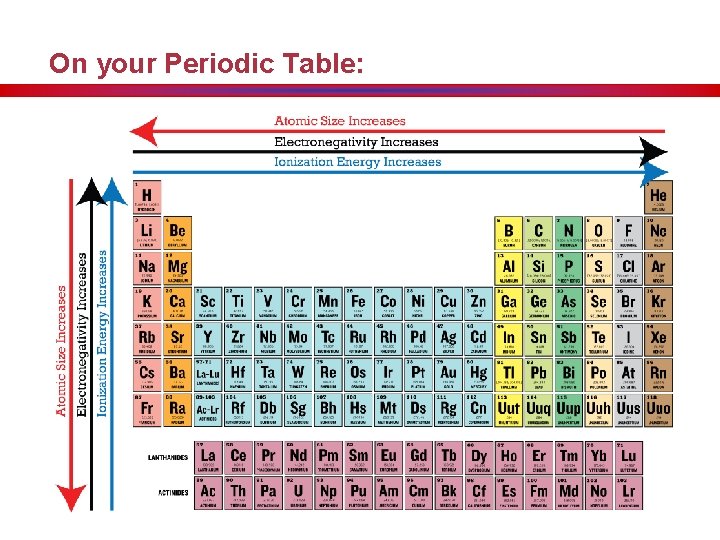
On your Periodic Table:

Hidden Treasure Melting/Boiling Pt. Trends www. ptable. com
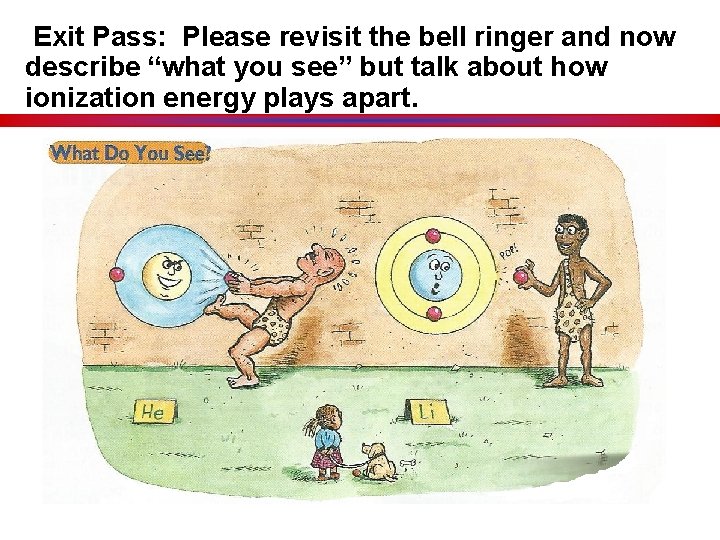
Exit Pass: Please revisit the bell ringer and now describe “what you see” but talk about how ionization energy plays apart.

This table is epic… what are the most helpful trends to do well on the Exam

EXAM…When you get a green table • • Valence Electron (Lewis Dot Structure) Common Ion Period numbers (Energy Levels) Atomic Radius/Ionization Energy Trend Electronegativity Trend Reactivity Tend Metal vs. Nonmetals

What other information do we need to know? Ionic Compounds Molecular Compounds • How to name and write the • How to name and write formulas • Use common ions to predict • Draw Lewis Structures (NOT the products of a chemical UNTIL NEXT SEMESTER) formulas 90% 10 %
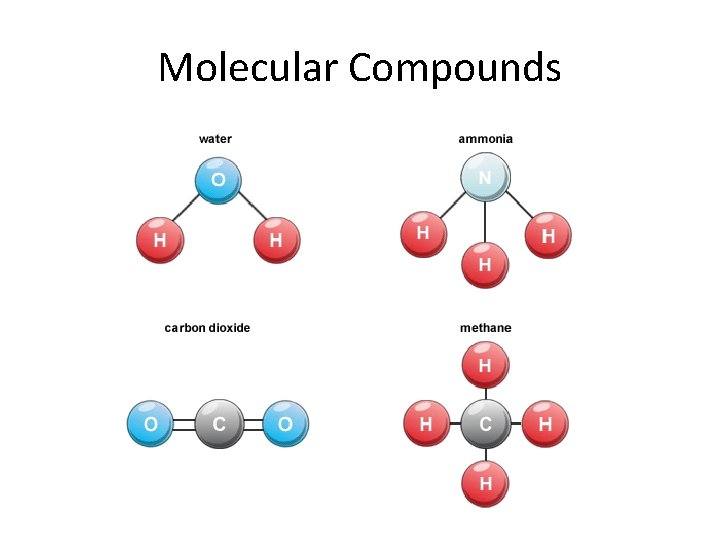
Molecular Compounds

How are covalent bonds represented? • Lewis structures are still used to represent A Lewis structure is a electrons in covalent bonds drawing of a molecule that illustrates the connectivity • Lewis structures for molecular compounds of the compound, as well as uses the same idea with valence electrons, but identifying the lone pairs of since the electrons are shared, actual bonds electrons available for need to be drawn bonding with other • Refresh your memory: draw the Lewis dot molecules. structure for Carbon, Oxygen, Sulfur, and Bromine

How are covalent bonds represented? • Key Concept 15: A Lewis structure is a drawing of a molecule that illustrates the connectivity of the compound, as well as identifying the lone pairs of electrons available for bonding with other molecules.

Lewis Structure Rules 1. Calculate the total electrons allowed in the compound (Add up valence electrons). 2. Draw skeleton of compound (Follows logic; place the element that is only contained once in the middle). 3. Add a single bond between elements and then give every element a full octet (you might break the Law of Conservation of Mass). 4. Count number of electrons in drawing from step #3 and compare to number of electrons allowed step #1. 5. If too many, draw another bond and erase 2 lone pair until you have the correct number of electrons allowed.
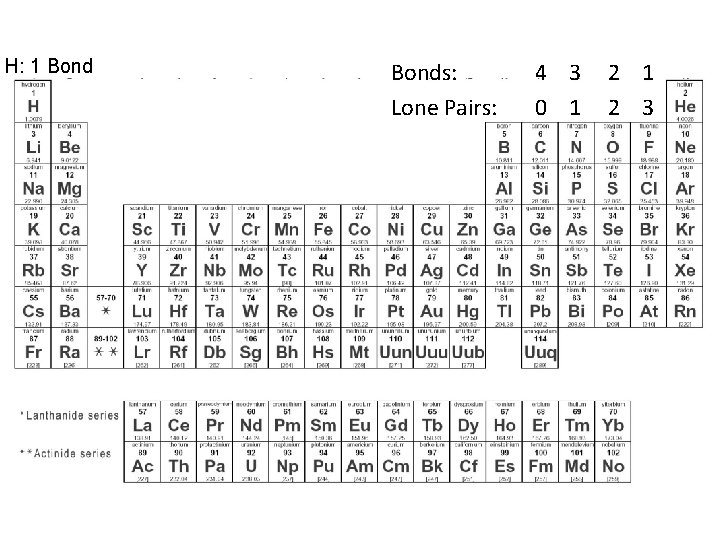
H: 1 Bonds: Lone Pairs: 4 3 2 1 0 1 2 3
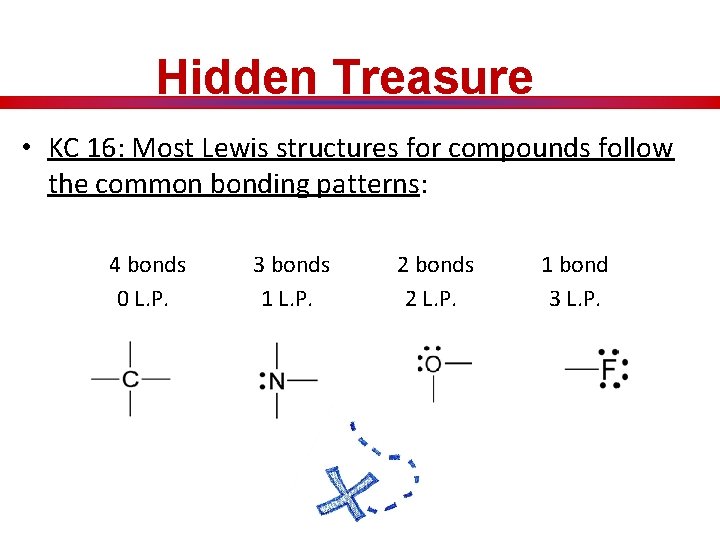
Hidden Treasure • KC 16: Most Lewis structures for compounds follow the common bonding patterns: 4 bonds 0 L. P. 3 bonds 1 L. P. 2 bonds 2 L. P. 1 bond 3 L. P.
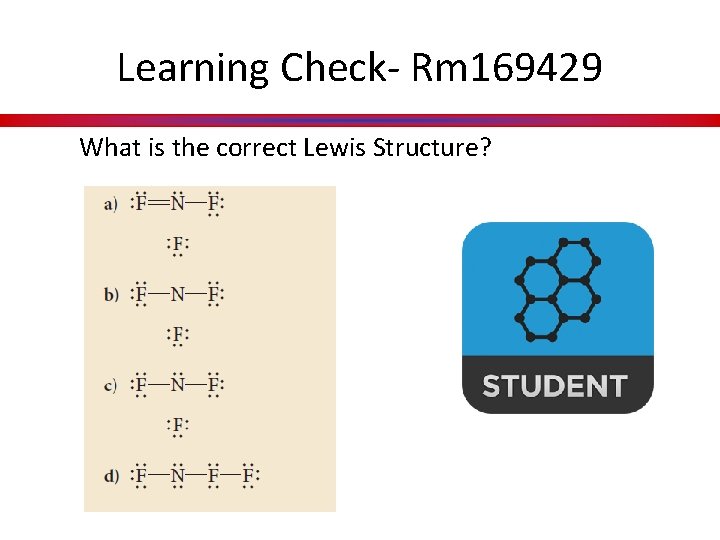
Learning Check- Rm 169429 What is the correct Lewis Structure?
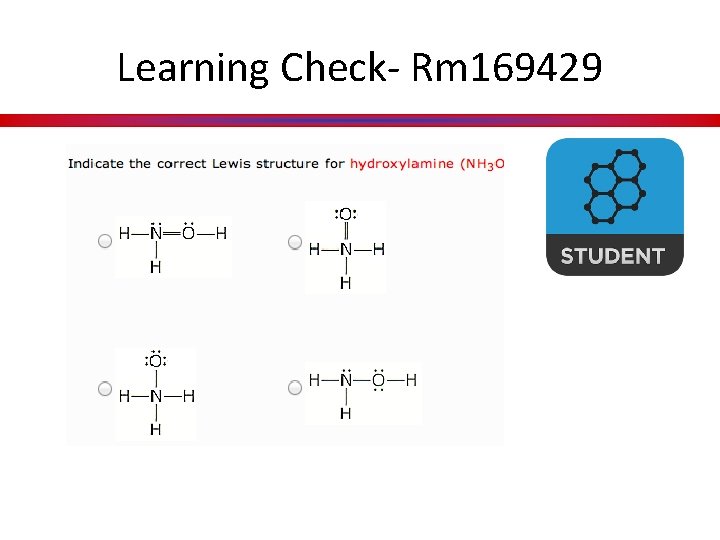
Learning Check- Rm 169429
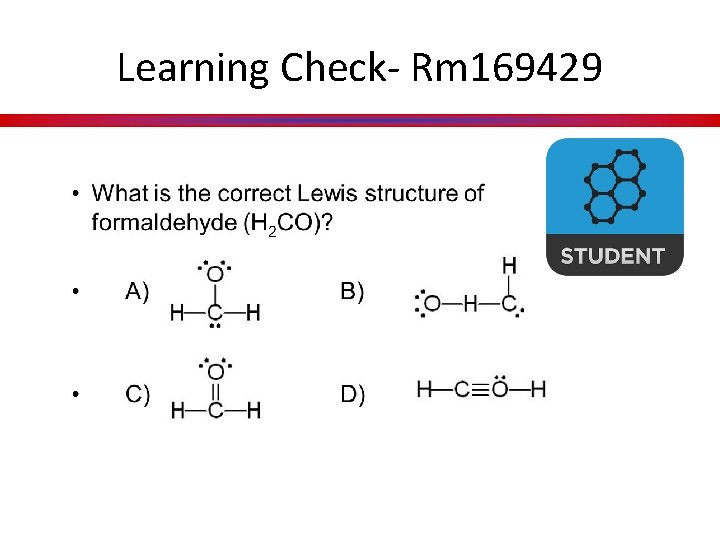
Learning Check- Rm 169429

Learning Check- Rm 169429 • For each of the following sets of atoms, rank them from highest to lowest ionization energy 1. Mg, Si, S

Learning Check- Rm 169429 • For each of the following sets of atoms, rank them from lowest to highest electronegativity 1. C, O, Ne

Learning Check- Rm 169429 Define Electronegativity

Learning Check- Rm 169429 • What common ion will the following atoms form? 1. Oxygen 2. Aluminum 3. Neon

End of Lesson 4
- Slides: 110