UNIT 7 SOLUTIONS Molarity Molality Colligative Properties Solubility

UNIT 7: SOLUTIONS Molarity, Molality, Colligative Properties, Solubility

Solutions • Solution – a homogeneous mixture which means that all of the parts are evenly distributed and not easily distinguished • Solutions are made of 2 parts: the solute and the solvent • Solute – dissolved particles in a solution • What is being dissolved • Solvent – the dissolving medium in a solution • what the solute is being dissolved in
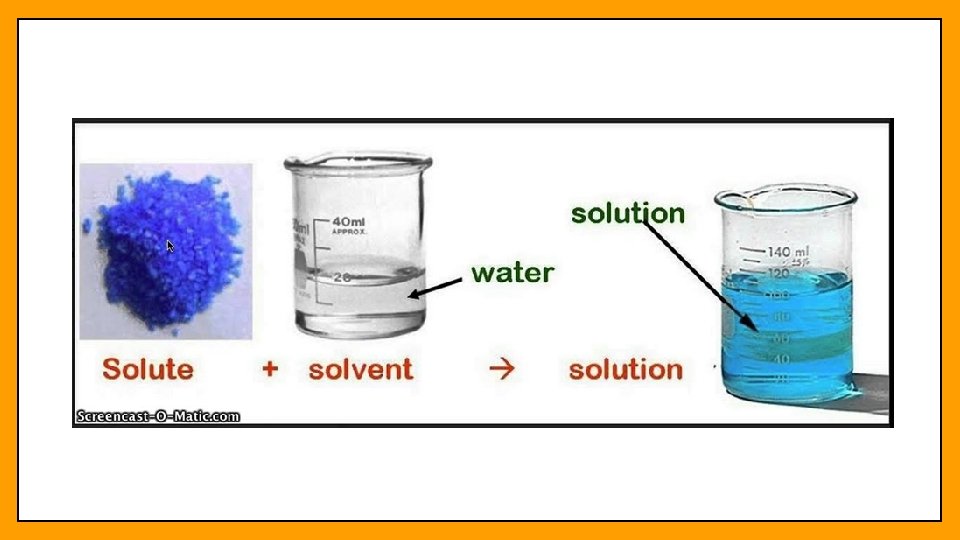

Concentration • Concentration – a measurement of the amount of solute that is dissolved in a given quantity of solvent • Concentration can be measured in two major ways • Molarity • Molality • Molarity is the more popular way to express concentration

MOLARITY
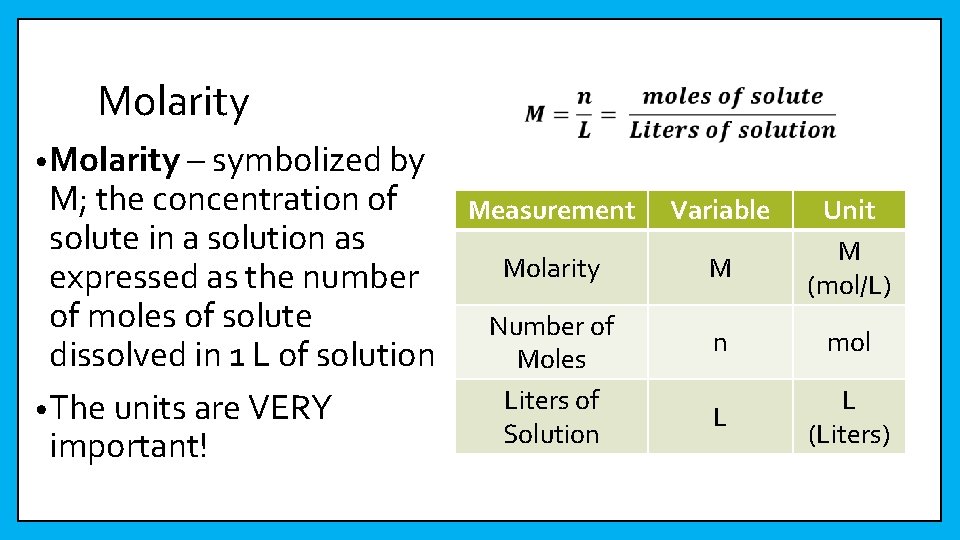
Molarity • Molarity – symbolized by M; the concentration of solute in a solution as expressed as the number of moles of solute dissolved in 1 L of solution • The units are VERY important! Measurement Variable Molarity M Number of Moles Liters of Solution Unit M (mol/L) n mol L L (Liters)

Practice Problems 1. What is the molarity of a solution that contains 0. 70 mol of Na. Cl dissolved in 0. 250 L of solution?

Practice Problems 2. What is the molarity of a solution that contains 0. 90 g of Na. Cl in exactly 2. 5 L of solution?

Practice Problems 3. How many moles of NH 4 NO 3 are in 335 m. L of 0. 425 M solution?

Practice Problems 4. How many liters of solution are prepared if the solution is 0. 75 M and made from 12. 3 moles of KCl?

MOLALITY
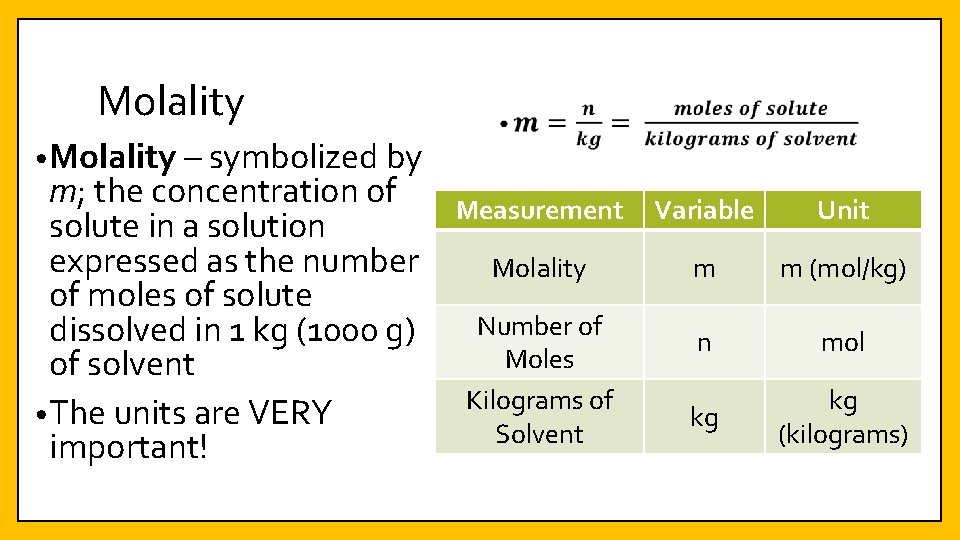
Molality • Molality – symbolized by m; the concentration of solute in a solution expressed as the number of moles of solute dissolved in 1 kg (1000 g) of solvent • The units are VERY important! Measurement Variable Unit Molality m m (mol/kg) n mol kg kg (kilograms) Number of Moles Kilograms of Solvent

Practice Problems 1. Calculate the molality of a solution prepared by dissolving 0. 575 moles of Na. Cl in 0. 675 kg of water.

Practice Problems 2. Calculate the molality of a solution prepared by dissolving 10. 0 g of Na. Cl in 600. g of water.

Practice Problems 3. How many kg of water must be used to dissolve 2. 35 moles of Mg. Cl 2 to produce a 0. 750 molal solution?

Practice Problems 4. How many grams of Na. F are needed to prepare a 0. 400 m solution that contains 750. grams of water?

COLLIGATIVE PROPERTIES

Colligative Properties • When a solute is added to a solvent, the solution will have different properties • Colligative properties: a property of a solution that depends only on the NUMBER of solute particles and not upon their identities • Examples of colligative properties are: • Freezing-point depression • Boiling-point elevation

Freezing-Point Depression • Freezing-point depression: the difference in temperature between the freezing point of a solution and the freezing point of the pure solvent • When a substance freezes, the particles of the solid take on a orderly pattern • When a solute is added, the solute particles disrupt this pattern

Boiling-Point Elevation • Boiling-point elevation: the difference in temperature between the boiling point of a solution and the boiling point of the pure solvent • When a solute is added to a solvent, the solute particles become surrounded by the solvent particles, not allowing as much energy to be able to escape to the surface
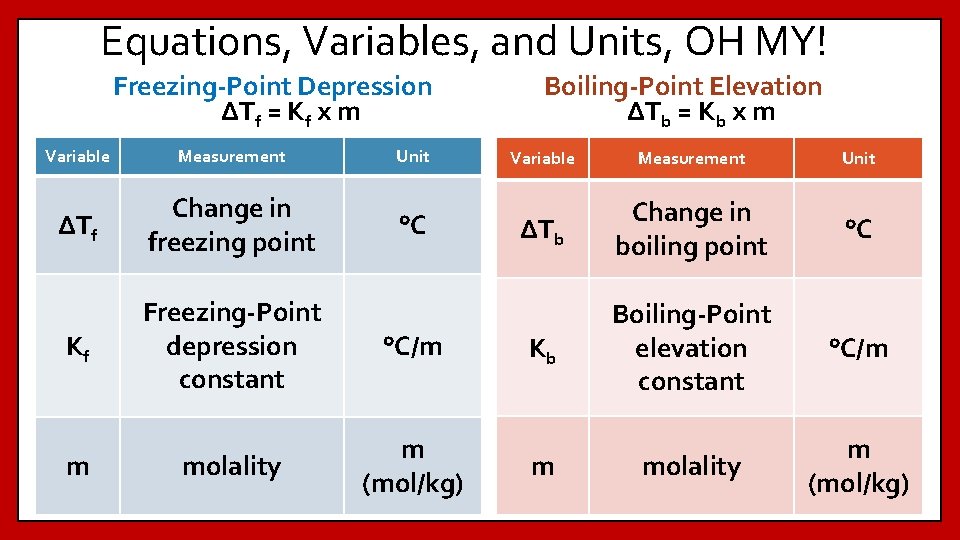
Equations, Variables, and Units, OH MY! Freezing-Point Depression ΔTf = Kf x m Variable Measurement Unit ΔTf Change in freezing point °C Kf Freezing-Point depression constant °C/m molality m (mol/kg) m Boiling-Point Elevation ΔTb = Kb x m Variable Measurement Unit ΔTb Change in boiling point °C Kb Boiling-Point elevation constant °C/m molality m (mol/kg) m

Practice • What is the new boiling point of a 1. 50 m C 6 H 12 O 6 solution? Kb = 0. 512 °C.

Practice • Antifreeze protects a car from freezing. It also protects it from over heating. Calculate the new freezing point of a solution containing 1. 61 moles of antifreeze (ethylene glycol = C 2 H 6 O 2) in 0. 500 kg of water. Kf = 1. 86 °C.

Practice • A solution is composed of sucrose and water. What is the molality of the solution of sucrose that has a freezing point of -15. 8 °C? Kf = 1. 86 °C/m

SOLUBILITY

Solution Formation • The composition (what it’s made of) of the solvent and solute determine whether the solute will dissolve in the solvent • Other factors that determine how fast the solute dissolves • Agitation (stirring) • Temperature • Surface area

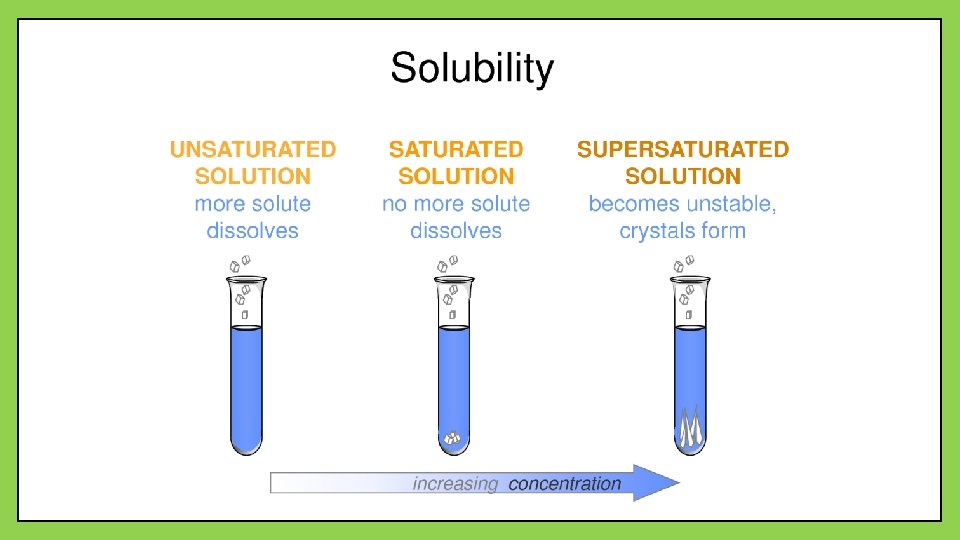
Solubility • The solubility of a substance is the amount of solute that dissolves in a given quantity of a solvent at a specific temperature and pressure to produce a saturated solution • There are three main categories of solutions • Saturated: contains the maximum amount of solute for a given quantity of solvent • Unsaturated: contains less solute than a saturated solution • Can dissolve more solute • Supersaturated: contains more solute than it can theoretically hold
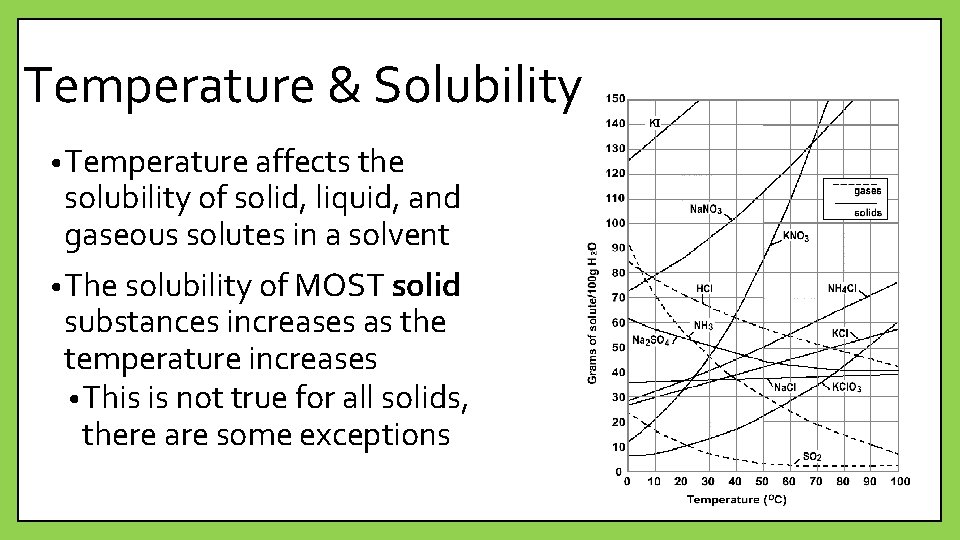
Temperature & Solubility • Temperature affects the solubility of solid, liquid, and gaseous solutes in a solvent • The solubility of MOST solid substances increases as the temperature increases • This is not true for all solids, there are some exceptions
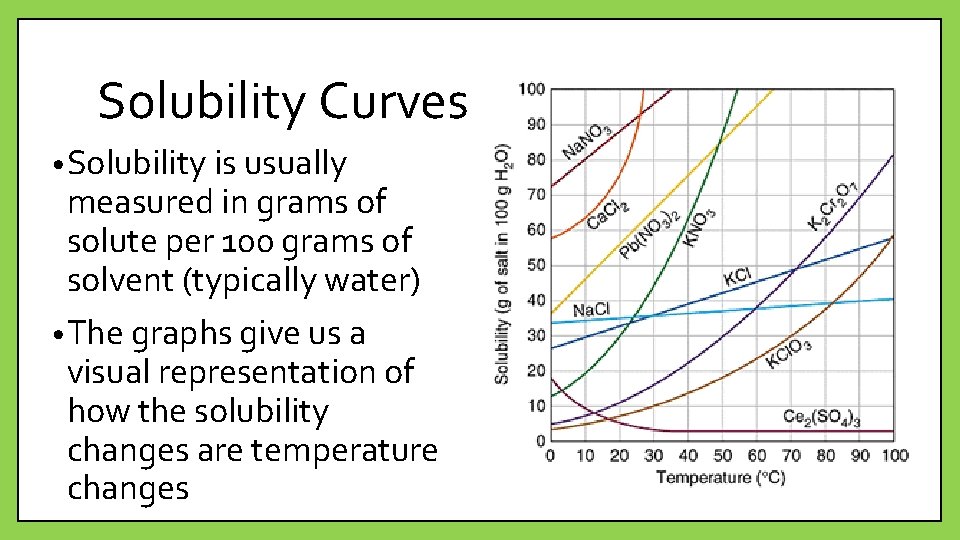
Solubility Curves • Solubility is usually measured in grams of solute per 100 grams of solvent (typically water) • The graphs give us a visual representation of how the solubility changes are temperature changes
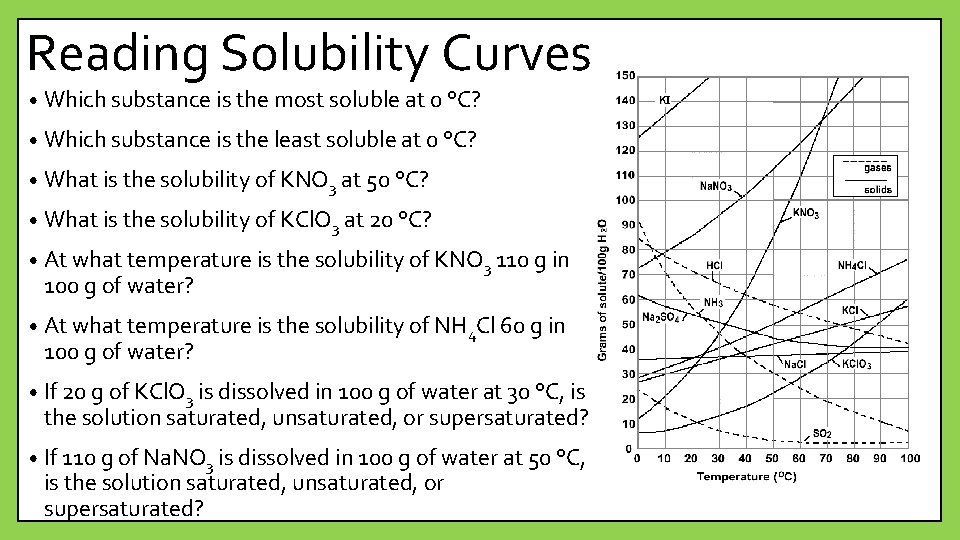
Reading Solubility Curves • Which substance is the most soluble at 0 °C? • Which substance is the least soluble at 0 °C? • What is the solubility of KNO 3 at 50 °C? • What is the solubility of KCl. O 3 at 20 °C? • At what temperature is the solubility of KNO 3 110 g in 100 g of water? • At what temperature is the solubility of NH 4 Cl 60 g in 100 g of water? • If 20 g of KCl. O 3 is dissolved in 100 g of water at 30 °C, is the solution saturated, unsaturated, or supersaturated? • If 110 g of Na. NO 3 is dissolved in 100 g of water at 50 °C, is the solution saturated, unsaturated, or supersaturated?
- Slides: 31