Unit 7 Organic Chemistry Organic Chemistry Chemists referred















- Slides: 25


Unit 7 Organic Chemistry

Organic Chemistry Chemists referred to these compounds as "organic" compounds because they were produced by living organisms.

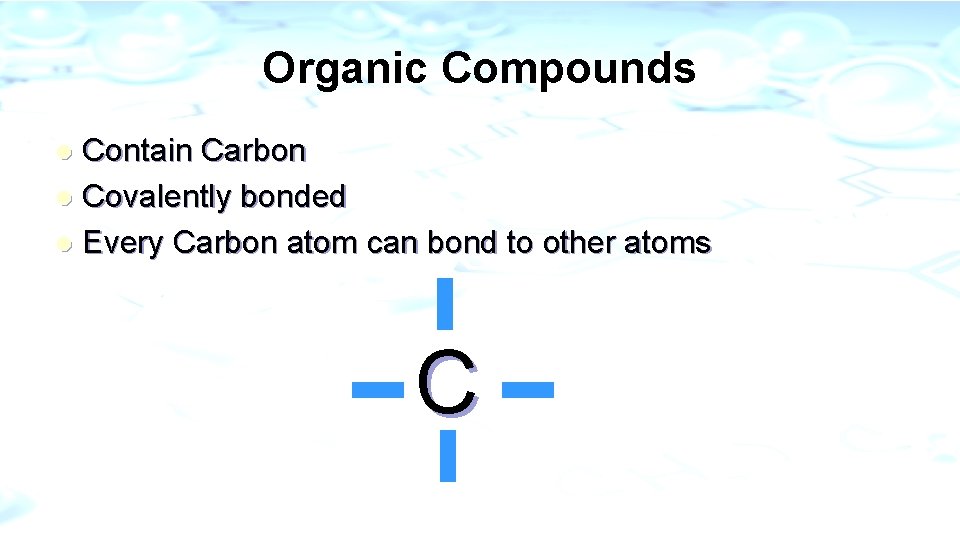
Organic Compounds Contain Carbon l Covalently bonded l Every Carbon atom can bond to other atoms l C

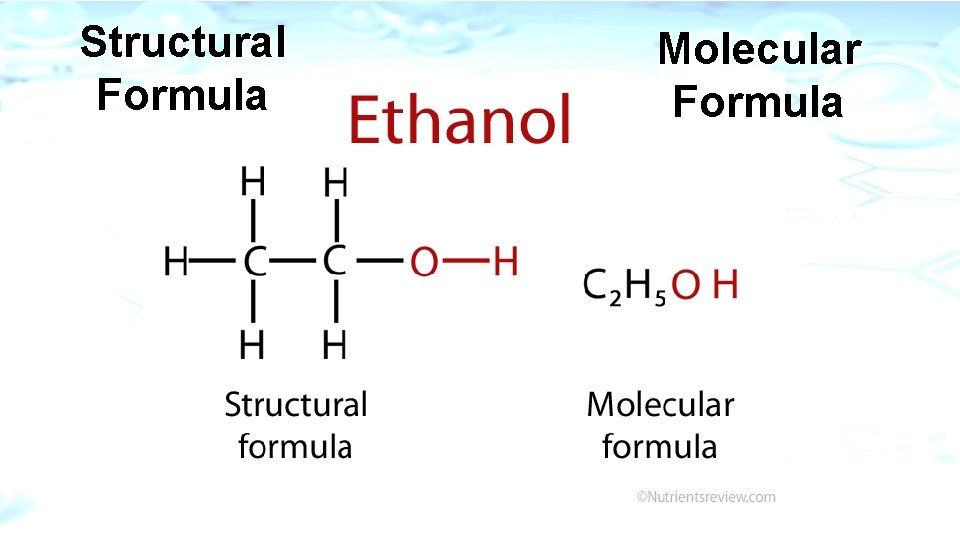
Structural Formula Molecular Formula

Organic Compounds l Hydrocarbons Functional groups Polymers

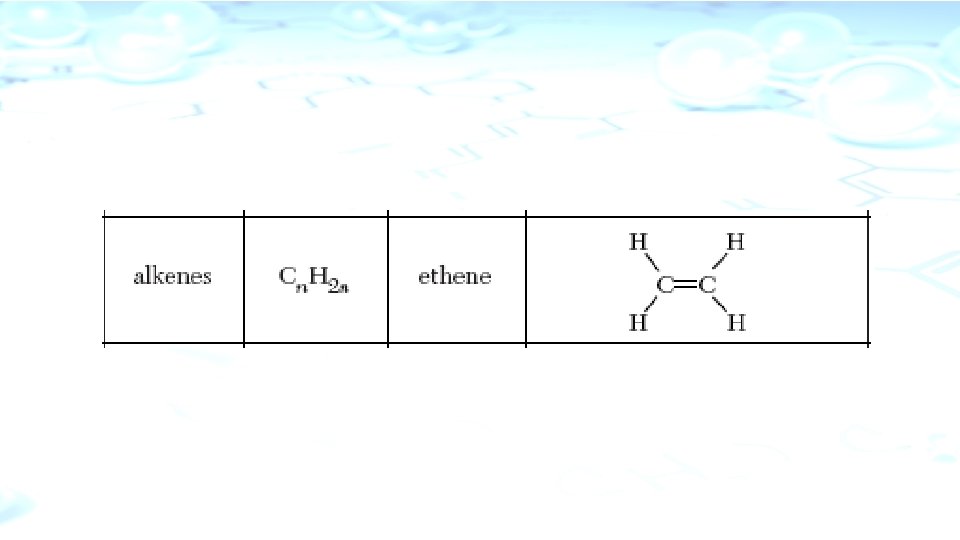
Hydrocarbons Simplest organic compound composed only of the elements carbon AND hydrogen. 4 Types 1. 1. Alkane 2. 2. Alkenes 3. 3. Alynes 4. 4. Aromatic Compounds

Saturated vs Unsaturated l Saturated - A hydrocarbon that contains only single bonds. l Unsaturated - A hydrocarbon that contains at least one double or triple bond between carbon atoms.

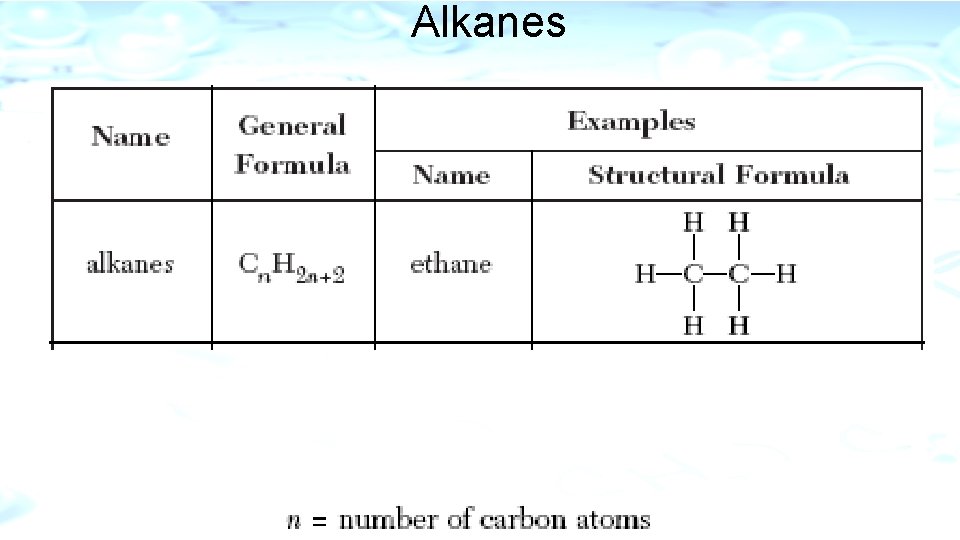
l. Alkanes l. Only Saturated Hydrocarbon l. Only single bonds between carbons l. Name ends in –ane l. Stable l. Insoluble in water l. Examples: Methane CH 4 and Ethane C 2 H 6

Alkanes

l. Alkenes l. Contain one double bond l. Name ends in –ene l. Unsaturated Hydrocarbon l. Insoluble in water but soluble in non-polar solvents l. Reactive l. Examples: Ethene or Ehylene


l Alkynes l. Contain one triple bond l. Name ends in –yne l. Unsaturated Hydrocarbon l. Non-polar, insoluble in water but soluble in non -polar solvents l. Triple bonds are unstable

Alkynes

Aromatic Compounds All contain at least one benzene l 6 electrons form 3 double bonds and are shared equally l All the carbon atoms are bonded to each with single bond l Non-polar l

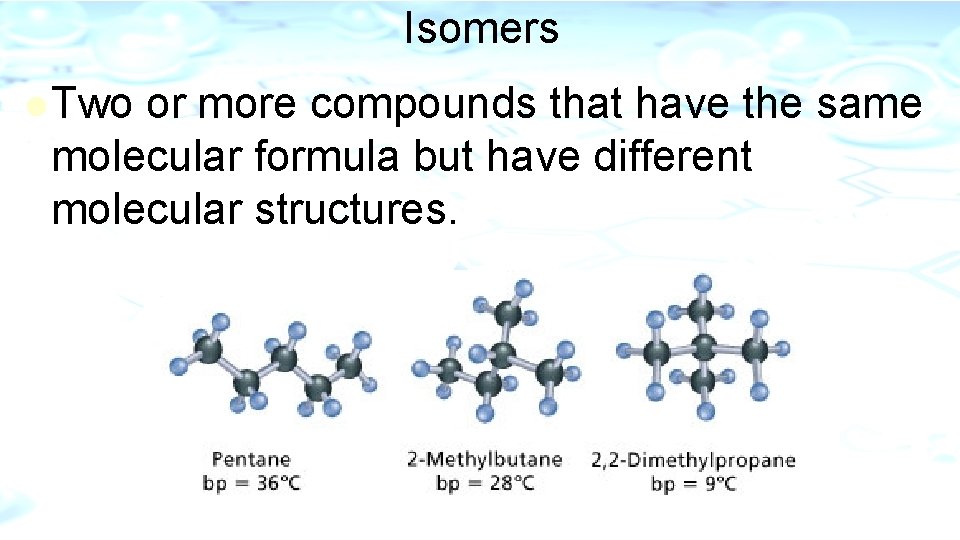
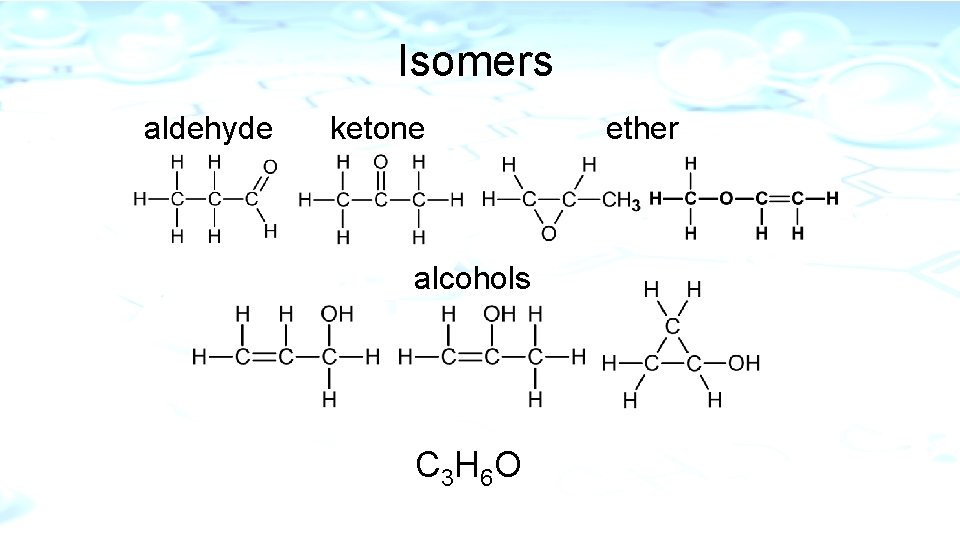
Isomers l Two or more compounds that have the same molecular formula but have different molecular structures.

Isomers aldehyde ketone alcohols C 3 H 6 O ether

Functional Groups l An atom or group of atoms that always react in a certain way in an organic molecule.

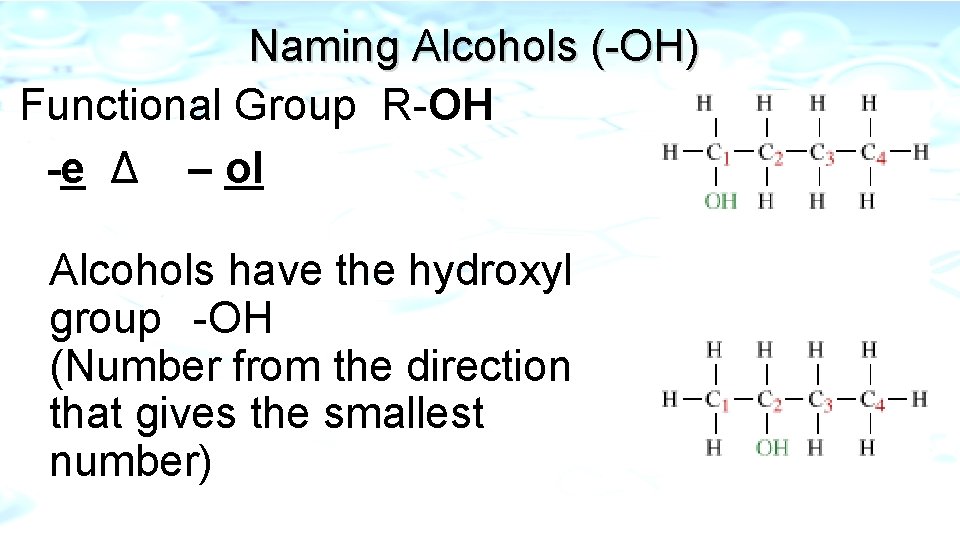
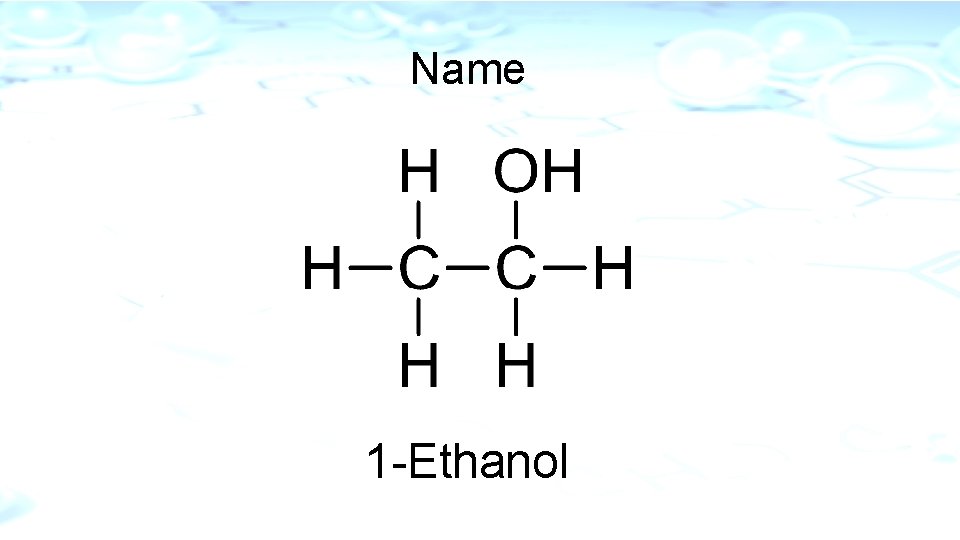
Naming Alcohols (-OH) Functional Group R-OH -e Δ – ol Alcohols have the hydroxyl group -OH (Number from the direction that gives the smallest number)

Name 1 -Ethanol

Substitution Rx - A reaction of saturated Organic Reactions organic compounds in which one atom or group of atoms in a molecule is replaced by an atom or group of atoms.

Esterification - Occurs when an acid reacts with an alcohol. l Polymers – repeating molecules

Fermentation – The process in which glucose is broken down, producing either ethanol, carbon dioxide, and energy (alcoholic fermentation) or lactic acid and energy (lactic acid fermentation).

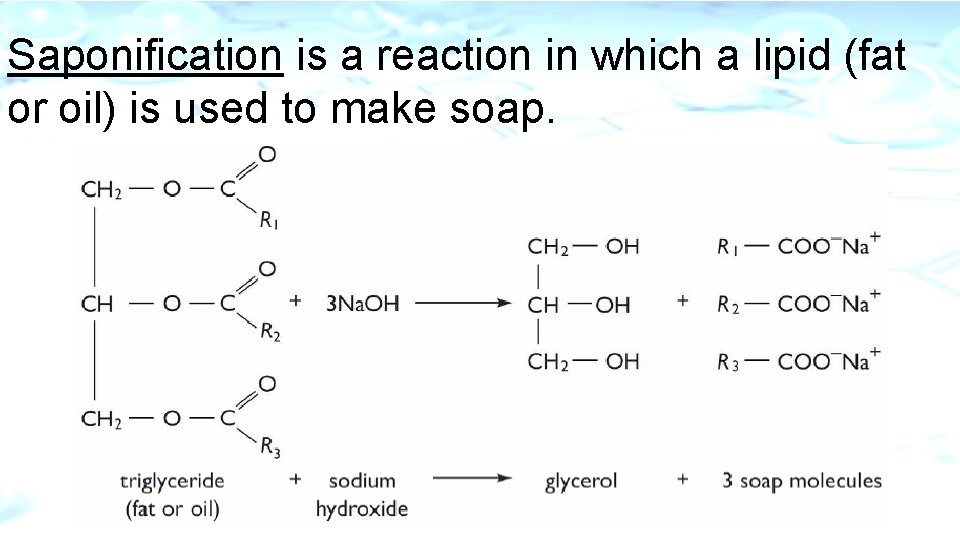
Saponification is a reaction in which a lipid (fat or oil) is used to make soap.

Fractional Distillation Separating Hydrocarbons (crude oil) using their boiling points