Unit 7 Organic Chemistry Nomenclature of Hydrocarbons and

Unit #7 Organic Chemistry Nomenclature of Hydrocarbons and of the Derivatives of Hydrocarbons

Organic Chemistry The study of compounds containing carbon. The majority of known compounds are organic (several million such compounds have been described). There are thousands of new organic compounds discovered each year.

You encounter many organic compounds every day. Alcohols (all of them) Sugars (glucose, sucrose, fructose…) Medications Fuels Pesticides Tires & Plastics Ethylene glycol (alcohol + sugar) (Antifreeze)

Three reasons why Carbon is integral to so many different compounds: 1. It can bond with other carbons to form “chains” and/or “rings”. Other atoms can do this, but only with up to a few atoms.

2. Carbon is “tetravalent”. This allows the “chains” to branch into other “chains” and/or “rings”.

3. The “SPONCH” elements also readily bond with carbon via single, double, and triple bonds.

Hydrocarbons Compounds containing only Hydrogen and Carbon (and sometimes oxygen). They are the simplest of all organic molecules. All hydrocarbons can be put into one of two classifications. 1. Aliphatic Hydrocarbons 2. Aromatic Hydrocarbons

Aliphatic Hydrocarbons All hydrocarbons that do not contain Benzene rings. They may be either chains or simple rings. Benzene A ring of six carbons with alternating single and double carbon-carbon bonds

Aromatic Hydrocarbons All hydrocarbons that contain Benzene rings or similar structural features. We will start with the Aliphatic Hydrocarbons first.

All Aliphatic Hydrocarbons can be placed into one of two groups: 1. Saturated Hydrocarbons 2. Unsaturated Hydrocarbons

Saturated Hydrocarbons in which all carbon atoms are bonded to the maximum number of Hydrogen atoms. (There are no carbon to carbon double or triple bonds).

Unsaturated Hydrocarbons in which all carbon atoms ARE NOT bonded to the maximum number of Hydrogen atoms. (There are carbon to carbon double or triple bonds). We will focus on the “Saturated Hydrocarbons”.

Alkanes Straight or branched chain hydrocarbons with Carbon-carbon single bonds. A. K. A. - Paraffins. Do not react with many reagents. General formula: Cn. H(2 n +2)

Alkanes constitute a “Homologous Series”. Homologous Series A series of compounds in which one compound differs from a preceding one by a “-CH 2 -” group.

Members of Homologous Series have similar chemical properties and their physical properties change throughout the series in a regular way. Examples: Melting and boiling points increase with molecular weight. This results from increasing intermolecular forces.

Structural Formula A formula showing the location and type of bond in a chemical compound. The dashes represent the bonds. Examples of the structural formulas of Methane, Ethane, Propane, and Butane follow.
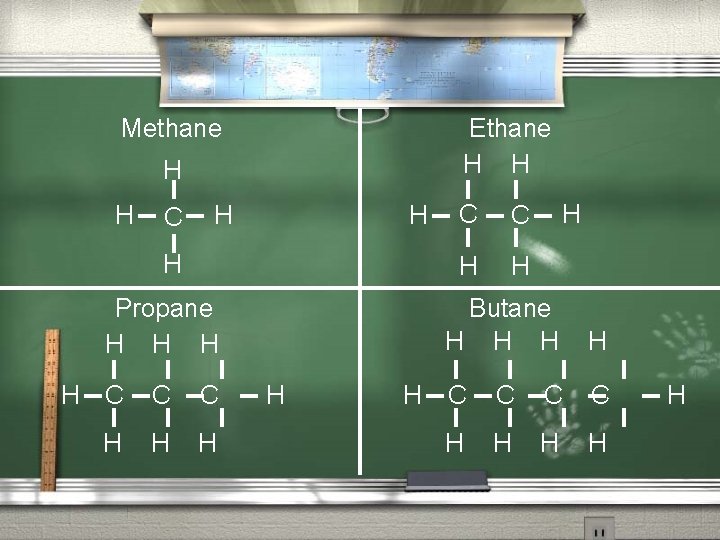
Methane Ethane H H C H H H Propane H H H H C C C H H H C C H H H Butane H H H H C C H H H

Condensed Structural Formula A formula that does not show the individual chemical bonds. However, all of the Hydrogen atoms attached to a given Carbon are written immediately after that Carbon.
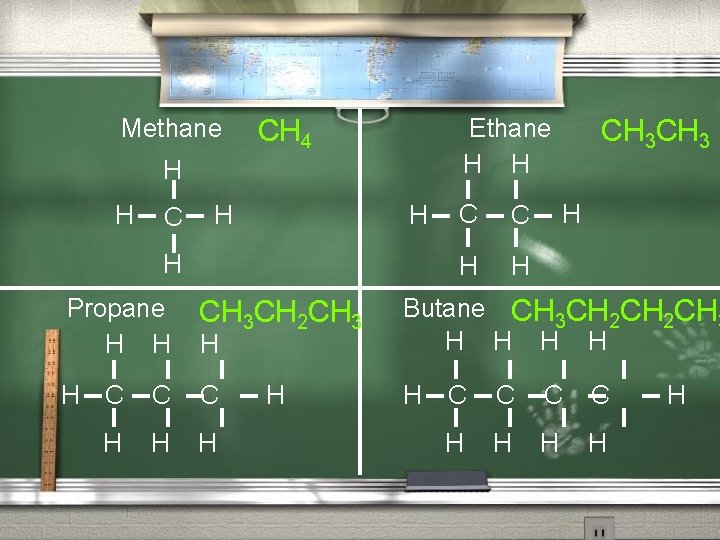
Methane Ethane H H CH 4 H H C H H H C C H H CH 3 H Propane CH 3 CH 2 CH 3 H H H Butane CH 3 CH 2 CH 3 H H H C C C H H H H H C C H H H

Skeletal Formula Also called line-angle or shorthand formula, A skeletal formula shows the skeletal structure or skeleton of a molecule, which is composed primarily of the SPONC (no H) elements that make up the molecule. Examples on next slide
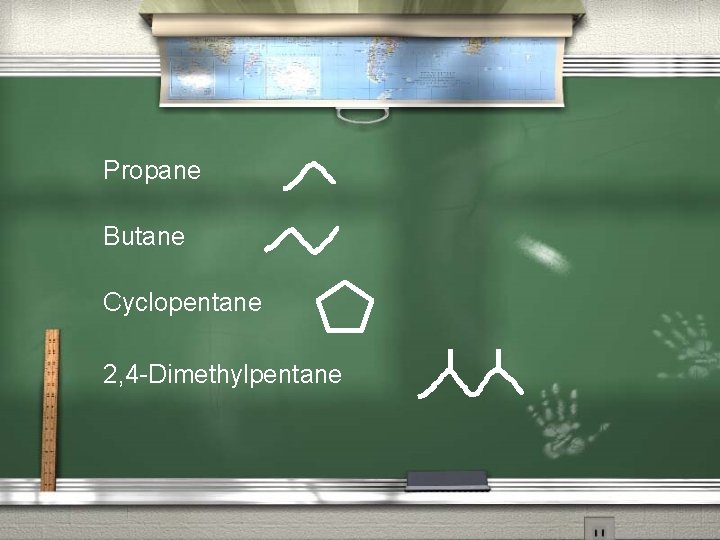
Propane Butane Cyclopentane 2, 4 -Dimethylpentane

Straight Chain Alkanes (you must memorize the first ten) - # of Carbons Structure Name 1 CH 4 Methane 2 CH 3 3 CH 3 CH 2 CH 3 4 CH 3 CH 2 CH 3 Butane 5 CH 3 CH 2 CH 2 CH 3 Pentane Ethane Propane

Straight Chain Alkanes (continued) # of Carbons Structure Name 6 CH 3 CH 2 CH 2 CH 3 Hexane 7 CH 3 CH 2 CH 2 CH 2 CH 3 Heptane 8 CH 3 CH 2 CH 2 CH 2 CH 3 Octane 9 CH 3 CH 2 CH 2 CH 3 Nonane 10 CH 3 CH 2 CH 2 CH Decane 3

“Alkyl” Groups are also often needed to name Organic compounds. While they are directly related to their straight chain namesake. They are named by dropping the “-ane” suffix and adding “-yl” to what remained of the name. Examples on the next slide

Straight Chain Alkanes # of Carbons Structure Name 1 CH 3 - Methyl 2 CH 3 CH 2 - 3 CH 3 CH 2 - 4 CH 3 CH 2 CH 2 - Butyl 5 CH 3 CH 2 CH 2 - Pentyl Ethyl Propyl

Straight Chain Alkanes # of Carbons Structure Name 6 CH 3 CH 2 CH 2 CH 2 - Hexyl 7 CH 3 CH 2 CH 2 CH 2 - Heptyl 8 CH 3 CH 2 CH 2 CH 2 CH 2 - Octyl 9 CH 3 CH 2 CH 2 CH 2 CH 2 -Nonyl 10 CH 3 CH 2 CH 2 CHDecyl 2 -

Branched Chain Alkane Hydrocarbon with the same molecular formula as a straight chain alkane, but a different structural formula. They have chemical and physical properties that differ from their straight chain counterparts.

Alkanes (or Alkyl groups) with three or more carbons have more than one possible way to be attached. Because of this, we will need a few additional “names” for these groups. Example: CH 3 CH 2 - vs. CH 3 CHCH 3 Propyl Isopropyl

The “Iso-” prefix is used whenever a group of three carbons (up to a maximum of 6 -carbons, “Isohexyl”) are arranged in such a way that the second carbon in the chain has two carbons attached to it. Example: CH 3 CHIsopropyl CH 3 CHCH 2 Isobutyl CH 3 CHCH 2 Isopentyl

Again, notice that the base name corresponds with the total number of carbons in each group. CH 3 CH- CH 3 CHCH 2 - Isopropyl Isobutyl 3 carbons 4 carbons CH 3 CHCH 2 Isopentyl 5 carbons

There are two additional prefixes we will need to know for Alkanes (or Alkyl groups) with four carbons (that have more than one possible way to be attached together). These prefixes are the “sec-” and “tert-” (lower case, and including the hyphen that must be used).

The “sec-” prefix refers to a secondary carbon that is the bonding site of the group. Secondary carbon: A carbon with two carbons directly bonded to it. CH 3 CH 2 CH- sec-Butyl

The “tert-” prefix refers to a tertiary carbon that is the bonding site of the group. Tertiary carbon: A carbon with three carbons directly bonded to it. CH 3 CCH 3 tert-Butyl

Structural Isomers Compounds with the same molecular formula, but different structural formulas. Examples: C 4 H 10 Butane vs. 2 -Methylpropane

Nomenclature of Alkanes Rules set by IUPAC (International Union of Pure and Applied Chemists. In this section we will learn the nomenclature of both “Straight Chain” and “Branched Chain” Hydrocarbons (up to 10 Carbons long).

Nomenclature of “Branched-chain” alkanes 1. Locate the longest continuous chain (this will be the base name for the alkane). Examples:

Nomenclature of “Branched-chain” alkanes 2. Number the longest chain beginning with the end nearest the attached group. Examples:

Nomenclature of “Branched-chain” alkanes 3. Use the numbers assigned in rule #2 to designate the location of the attached groups. (“Their address”) Examples:

Nomenclature of “Branched-chain” alkanes 4. When two or more substituents are present, give each a number corresponding to its location on the longest chain. The groups should be listed alphabetically, ignoring: di, tri, tetra…, as well as sec-, and tert- (note: di, tri, tetra… are only capitalized if they are the first letters in a name. The sec- and tert- are never capitalized). Examples:

Nomenclature of “Branched-chain” alkanes 5. When two or more substituents are present on the same carbon, use that number twice. Examples:

Nomenclature of “Branched-chain” alkanes 6. When two or more substituents are identical, indicate this by the use of the prefixes di-, tri, or tetra-, and so on. Examples:

Nomenclature of “Branched-chain” alkanes 7. When two chains of equal length compete for selection as the base chain, choose the chain with the most substituents. Examples:

Nomenclature of “Branched-chain” alkanes 8. When branching first occurs at an equal distance from either end of the longest chain, choose the name that gives the lower number at the first point of difference Examples:

Alkyl Halide An Alkane with a halogen(s) as a substituent.

The Nomenclature of Alkyl halides

Rules for naming the alkyl halides 1. Use the same rules as discussed for branched alkanes, But…

Rules for naming the alkyl halidess 2. Name the halogen substituent as Fluoro-, Chloro-, Bromo-, or Iodo-; followed by the base chain’s name (note: if the halogen is on carbon #1, then no “address” is required).

Rules for naming the alkyl halidess 3. The base chain’s name is based on the longest chain containing the halogen, even if it is not the longest chain.

Rules for naming the alkyl halidess 4. Number the chain so that the halogen has the lowest possible number.

Rules for naming the alkyl halidess 5. Halogen have preference in the numbering system over other alkyl groups (excluding alcohols).
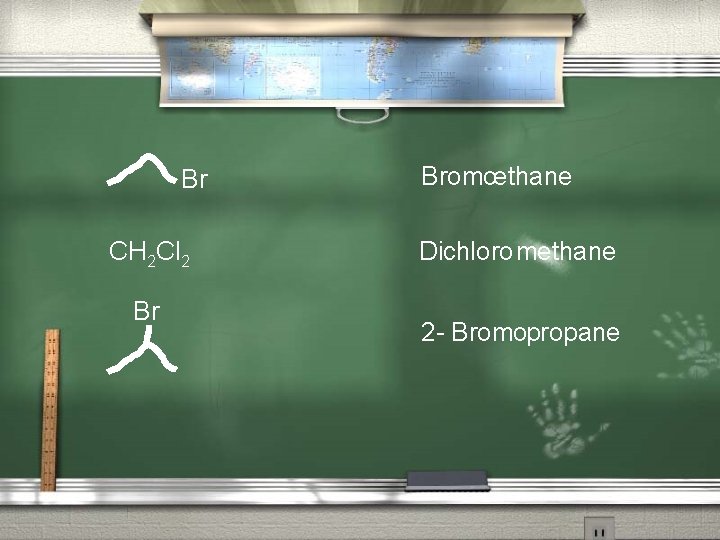
Br CH 2 Cl 2 Br Bromoethane Dichloro methane 2 - Bromopropane

Additional Practice Examples:

The Nomenclature of Alcohols

Name Molecular Formula Methanol (methyl alcohol) CH 3 OH Ethanol (ethyl alcohol) C 2 H 5 OH Propanol C 3 H 7 OH Butanol C 4 H 9 OH
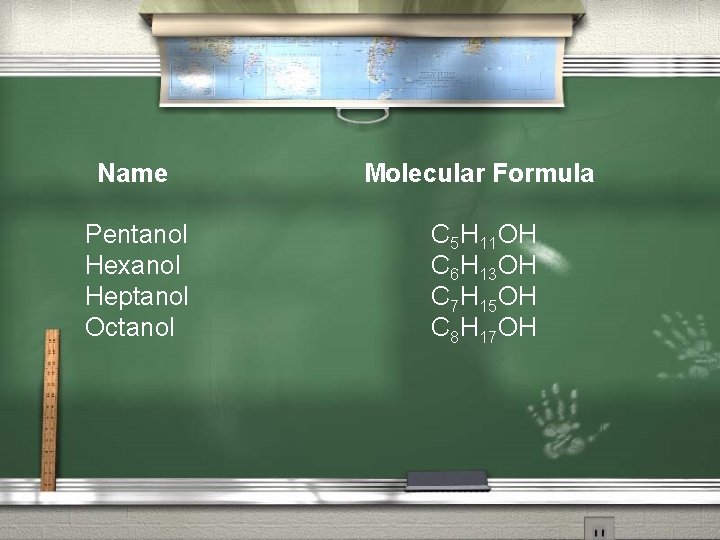
Name Pentanol Hexanol Heptanol Octanol Molecular Formula C 5 H 11 OH C 6 H 13 OH C 7 H 15 OH C 8 H 17 OH

Rules for naming the alcohols 1. Find the longest chain containing the hydroxyl group (OH). Branches on the base chain will be named as substituents. Change the base name to indicate an alcohol by dropping the final “e” and adding an “ol”.

OH octanol octane

Rules for naming the alcohols 2. Number the chain to give the carbon with the hydroxyl group the lower number. With the exception of carbonyl groups such as ketones and aldehydes (a carbon double bonded to an oxygen atom), the alcohol or hydroxyl groups have first priority for naming.
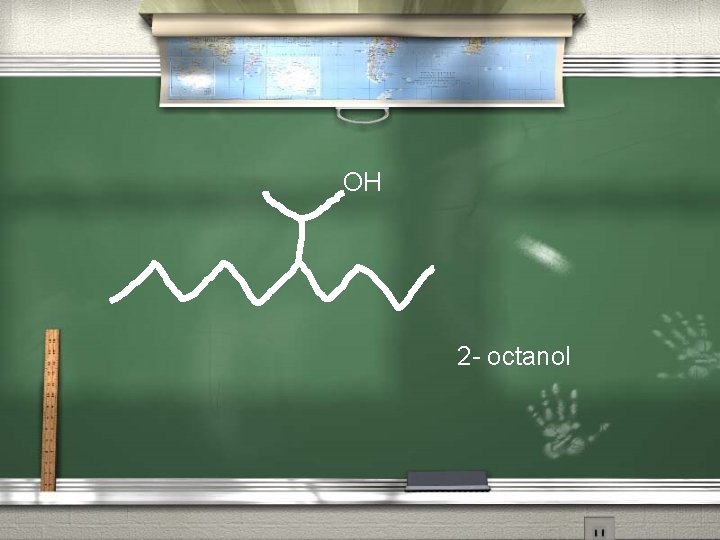
OH 2 - octanol

Rules for naming the alcohols 3. Follow the nomenclature rules for other branched chain alkanes.

OH 3 -Butyl- 2 - octanol

Rules for naming the alcohols 4. Alcohols receive preference over other alkyl groups and halogens in the numbering system. OH Cl 4 -Chloro-1 -butanol
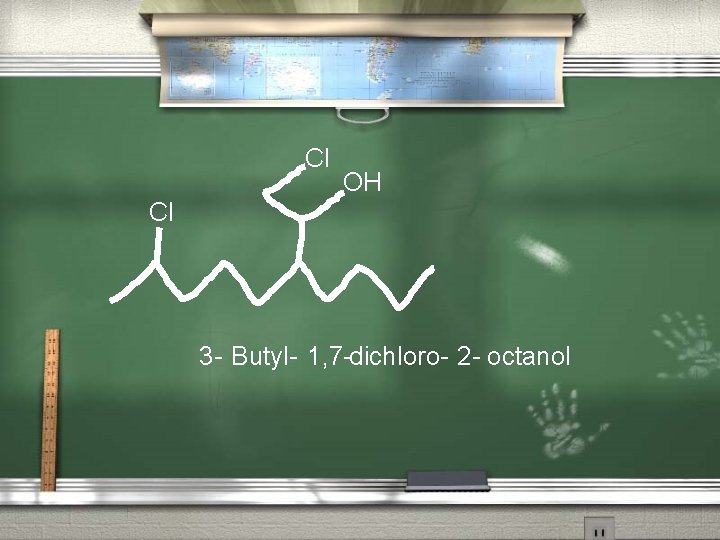
Cl OH Cl 3 - Butyl- 1, 7 -dichloro- 2 - octanol

Practice Examples:

Rules for naming the alcohols 5. Alcohols with two hydroxyl groups are commonly called “glycols”. In the IUPAC nomenclature system they are called “diols”. List the “address” of the hydroxyl groups, followed by the base chain name, followed by “diol”.

Practice Examples:

Glycols aka Diols (the IUPAC name) Alcohols with 2 hydroxyl groups. Nomenclature: list the “address” of the 2 hydroxyl groups, followed by the base chain name, followed by diol.

Practice Examples:

Cycloalkane An alkane with a ring(s) of carbon. Monocyclic alkane A cycloalkane with only one ring in it’s structure. Bicyclic alkane A cycloalkane with two fused rings in it’s structure.

The Nomenclature of Monocyclic alkanes 1. Use the previous rules, however, name the compound as either: a) An Alkylcycloalkane b) A Halocycloalkane c) An Alkylcycloalkanol

Practice Example naming as an Alkylcycloalkane: Note: if only one substituent is present, it is not necessary to give it’s “address”. CH 2 CH 3 Ethyl cyclo hexane

Practice Example naming as an Halocycloalkane: Note: if only one substituent is present, it is not necessary to give it’s “address”. Cl Chlorocyclo hexane

Practice Example naming as an Alkylcycloalkanol: Note: if only one substituent is present, it is not necessary to give it’s “address”. OH Cyclohexanol

The Nomenclature of Monocyclic alkanes (continued) 2. If more than one substituent is present, number the ring in the way that gives the next substituent the lowest number possible.
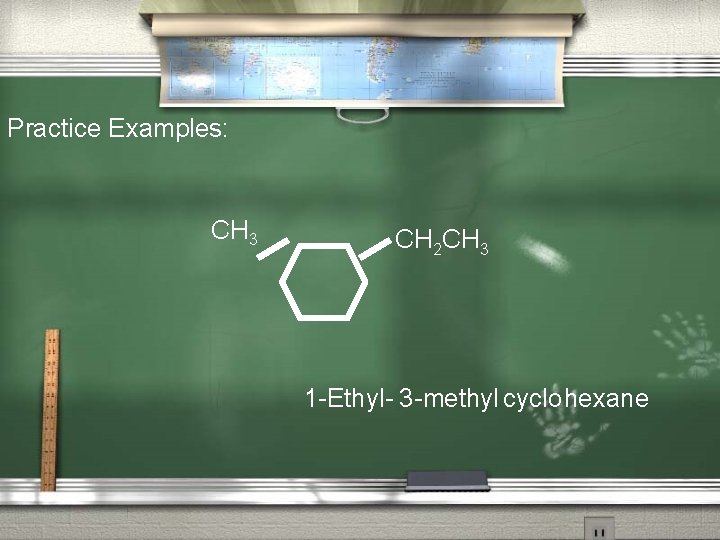
Practice Examples: CH 3 CH 2 CH 3 1 -Ethyl- 3 -methyl cyclo hexane

Practice Examples:

The Nomenclature of Monocyclic alkanes (continued) 3. cis- vs. trans- isomerism cis. The position when two adjacent substituents are on the same side of the ring.

Practice Examples with cis- isomerism:

The Nomenclature of Monocyclic alkanes (continued) 3. cis- vs. trans- isomerism trans. The position when two adjacent substituents are on the opposite sides of the ring.

Practice Examples with trans- isomerism:

The Nomenclature of Bicyclic alkanes. 1 A. Determine the total number of carbons comprising the two rings. B. Determine the base name as follows: bicyclo + the name of the alkane with “X” carbons. Example on the next slide

The number of carbons = Base name is = 7 bicycloheptane

The Nomenclature of Bicyclic alkanes (continued). 2. Insert in-between the bicyclo ____ and the alkane name, an expression in brackets that denotes the number of carbon atoms (in decending order) in each bridge. Example on the next slide
![Bicyclo heptane [2, 2, 1] Bicyclo heptane [2, 2, 1]](http://slidetodoc.com/presentation_image_h/119de8ae095645882ecaa7b214f8eec7/image-84.jpg)
Bicyclo heptane [2, 2, 1]

The number of carbons = Base name is = 4 bicyclobutane
![Bicyclo butane [1, 1, 0] Bicyclo butane [1, 1, 0]](http://slidetodoc.com/presentation_image_h/119de8ae095645882ecaa7b214f8eec7/image-86.jpg)
Bicyclo butane [1, 1, 0]
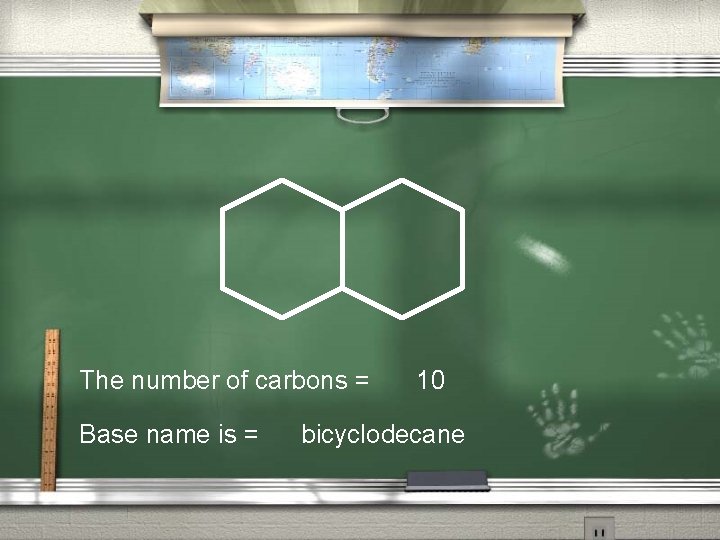
The number of carbons = Base name is = 10 bicyclodecane
![Bicyclo decane [4, 4, 0] Bicyclo decane [4, 4, 0]](http://slidetodoc.com/presentation_image_h/119de8ae095645882ecaa7b214f8eec7/image-88.jpg)
Bicyclo decane [4, 4, 0]

The Nomenclature of Bicyclic alkanes (continued). 3. If substituents are present, we must number the bridged ring system as follows: a. Begin at one of the bridge heads and proceed, first along the longest bridge to the other bridge head.

The Nomenclature of Bicyclic alkanes (continued). 3. b. Next, you will number along the next longest bridge, and back to the first bridge head. c. Finally, the shortest bridge will be numbered last. Example on the next slide
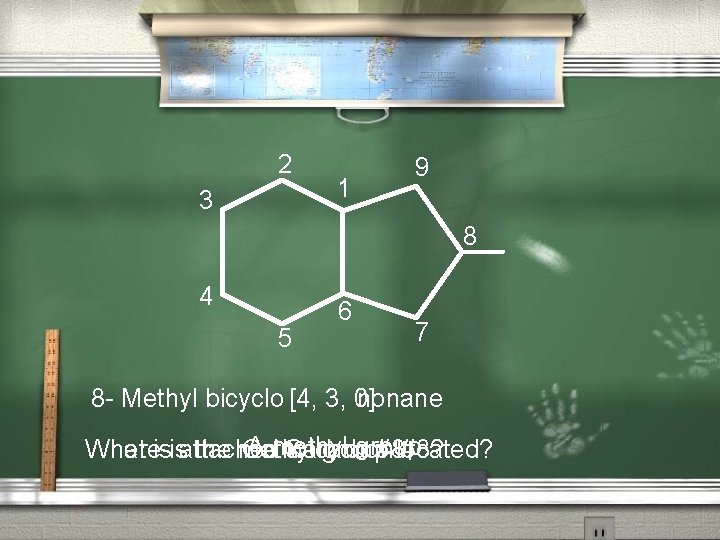
2 3 1 9 8 4 5 6 7 8 - Methyl bicyclo nonane [4, 3, 0] A methyl group Where is the methyl group located? What is attached to carbon #8? On Carbon #8
![Cl Cl 6, 7 - Dichloro bicyclo heptane [3, 2, 0] Cl Cl 6, 7 - Dichloro bicyclo heptane [3, 2, 0]](http://slidetodoc.com/presentation_image_h/119de8ae095645882ecaa7b214f8eec7/image-92.jpg)
Cl Cl 6, 7 - Dichloro bicyclo heptane [3, 2, 0]
![Draw / build the following structures: 1. Bicyclo[2, 2, 0]hexane 2. Bicyclo[4, 4, 0]decane Draw / build the following structures: 1. Bicyclo[2, 2, 0]hexane 2. Bicyclo[4, 4, 0]decane](http://slidetodoc.com/presentation_image_h/119de8ae095645882ecaa7b214f8eec7/image-93.jpg)
Draw / build the following structures: 1. Bicyclo[2, 2, 0]hexane 2. Bicyclo[4, 4, 0]decane 3. 1 -Butylcyclohexane 4. Bicyclo[2, 2, 2]octane

HOMEWORK: Draw the structures and write the names of the bicyclic compounds that are isomers of: Bicyclo[2, 2, 1]heptane.
- Slides: 94