Unit 7 Metallic Bonding Bonding in Metals l

Unit 7 Metallic Bonding

Bonding in Metals l OBJECTIVES: l. Model the valence electrons of metal atoms. l. Describe the arrangement of atoms in a metal. l. Explain the importance of alloys.

What about metals? l The atoms of metals are electron donors, so they cannot form ionic bonds with each other. Metals want to give away their valence electrons!

Metallic Bonds are… l How metal atoms are held together in the solid. l Metals hold on to their valence electrons very weakly. l Think of them as positive ions (cations) floating in a sea of electrons.

METALLIC BOND - bond found in metals - holds metal atoms together very strongly

Sea of Electrons l Electrons are free to move through the solid. l Metals conduct electricity. + + +

Metallic Bonding l Metallic bonding refers to the attraction between the delocalized electrons and the metal nuclei.

Metals Metallic bonds explain many physical properties of metals, such as l Electrical conductivity l High melting points l Malleability and Ductility l Luster

Metals are good conductors of electricity: The loose electrons are free to move throughout the metal. These mobile electrons can act as charge carriers in the conduction of electricity.

Metals have high melting points l In order to melt any substance, you have to pull apart the particles: Metals tend to have high melting and boiling points because of the strength of the metallic bond. l The strong bond means that it takes a lot of energy (heat) to pull apart the metal atoms. l

Malleability and Ductility l Metals are described as malleable (can be beaten into sheets) and ductile (can be pulled out into wires). These properties are caused by the ability of the atoms to roll over each other into new positions without breaking the metallic bond.

Metals are Malleable l. Hammered into shape (bend). l. Also ductile - drawn into wires. l. Both malleability and ductility explained in terms of the mobility of the valence electrons
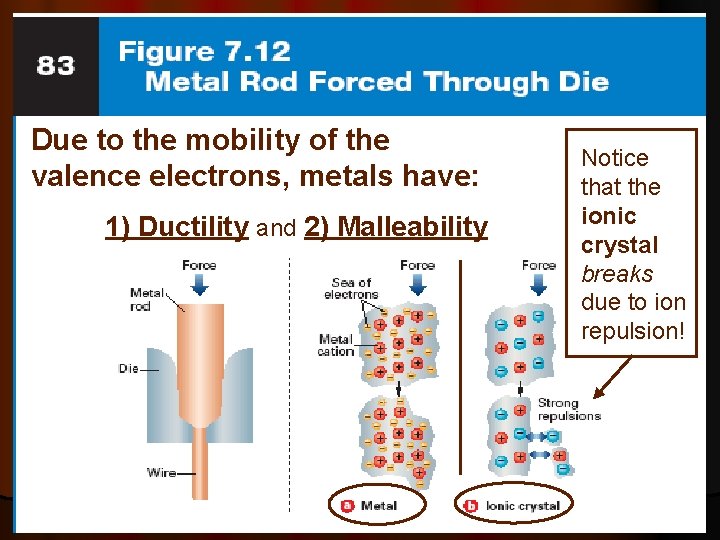
Due to the mobility of the valence electrons, metals have: 1) Ductility and 2) Malleability Notice that the ionic crystal breaks due to ion repulsion!
Malleable Force + + +
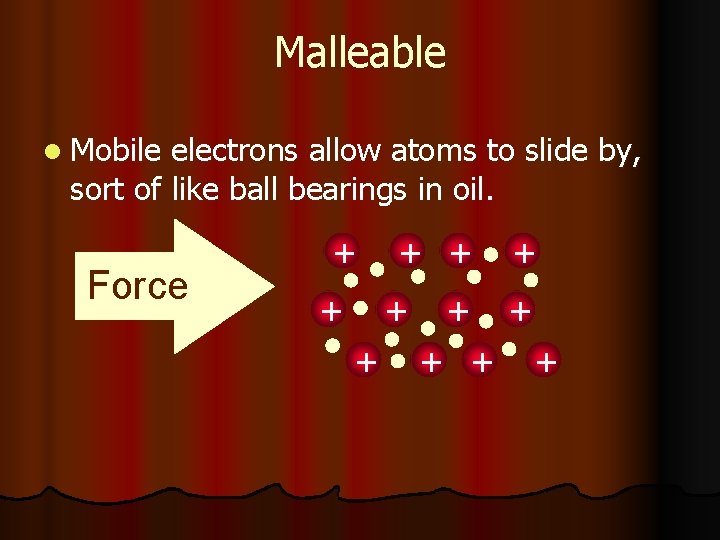
Malleable l Mobile electrons allow atoms to slide by, sort of like ball bearings in oil. Force + + +
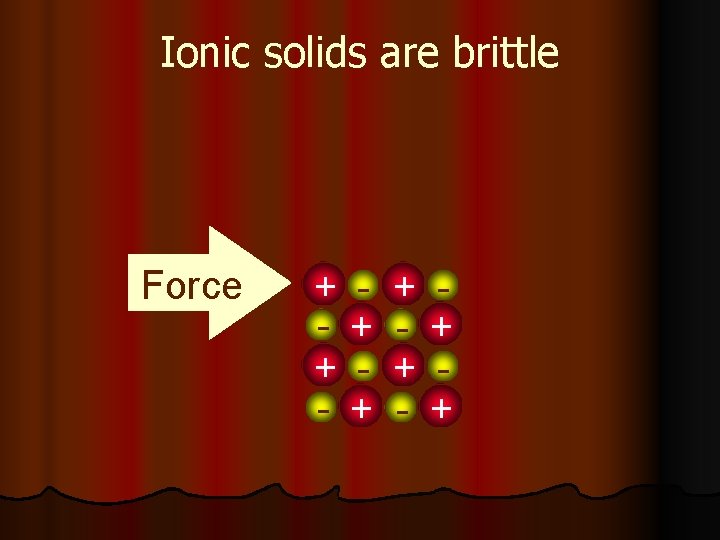
Ionic solids are brittle Force + + - + +
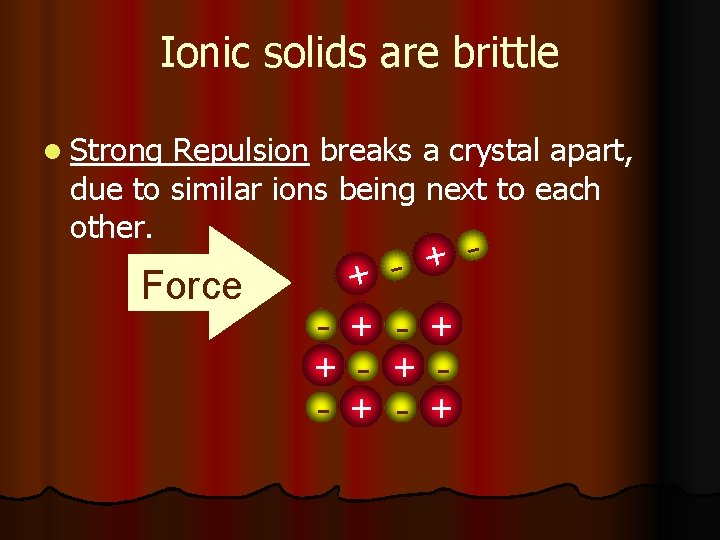
Ionic solids are brittle l Strong Repulsion breaks a crystal apart, due to similar ions being next to each other. Force + + - + - + - +

Luster l l The bright luster of metals is caused by the delocalized mobile electrons. When light falls on the surface of the metal, the loosely held electrons absorb photons of lights. The electrons get promoted to higher energy levels (excited state). These electrons readily return from the higher to the lower levels of energy by releasing energy, thus becoming a source of light radiations. Light appears to be reflected from metal surface, and the surface acquires a shining appearance, which is known as metallic luster.

Alloys l We use lots of metals every day, but few are pure metals l Alloys are mixtures of 2 or more elements, at least 1 is a metal l made by melting a mixture of the ingredients, then cooling l Brass: an alloy of Cu and Zn l Bronze: Cu and Sn

Why use alloys? l Properties are often superior to the pure element l Sterling silver (92. 5% Ag, 7. 5% Cu) is harder and more durable than pure Ag, but still soft enough to make jewelry and tableware l Steels are very important alloys l corrosion resistant, ductility, hardness, toughness, cost

More about Alloys… Types? a) substitutional alloy- the atoms in the components are about the same size l b) interstitial alloy- the atomic sizes quite different; smaller atoms fit into the spaces between larger l “Amalgam”- dental use, contains Hg
- Slides: 21