Unit 7 Covalent Bonding covalent bond formed by
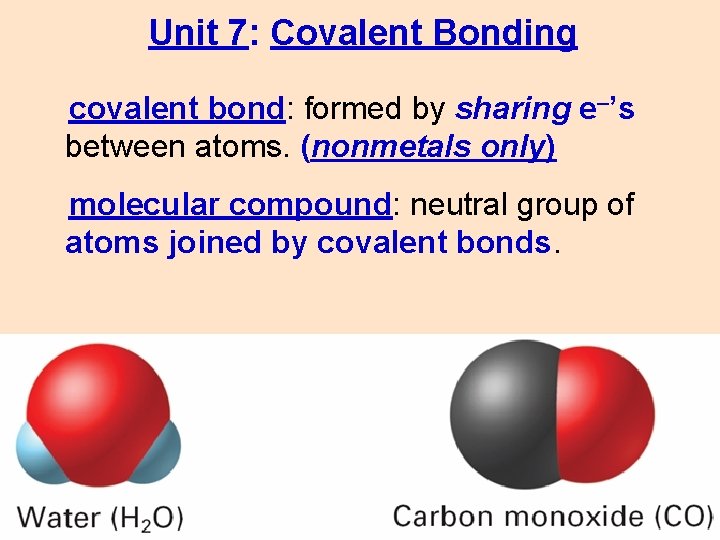
Unit 7: Covalent Bonding covalent bond: formed by sharing e–’s between atoms. (nonmetals only) molecular compound: neutral group of atoms joined by covalent bonds.

Ionic Bonds transfer electrons between a metal and non-metals (usually). Covalent Bonds share electrons between non-metals. Idea is still to get a full valence shell “ 8 electrons…” around each atom. Whether it’s covalent or ionic is dictated by the difference in the atom’s electronegativities.

diatomic molecules (all covalently bonded): two atoms of the same element. There are Seven Diatomic Molecules Hydrogen (Gas) Nitrogen (Gas) Oxygen (Gas) Fluorine (Gas) *YOU MUST KNOW THESE. FROM NOW Chlorine Bromine (Liquid)

molecular formula: shows how many atoms of each element

Properties of molecular compounds: Ø lower m. p. ’s than ionic compounds. (usually liquid or gas at room Temp. ) Ø poor conductors


Quick Quiz! 1. A molecular compound usually consists of A. two metal atoms and a nonmetal atom. B. two nonmetal atoms and a metal atom. C. two or more metal atoms. D. two or more nonmetal atoms.

Quick Quiz. 2. A molecular formula shows… A. how many atoms of each element a molecule contains. B. a molecule's structure. C. which atoms are bonded together. D. how atoms are arranged in space.

Quick Quiz. 3. Compared to ionic compounds, molecular compounds tend to have relatively A. low melting points. B. high melting points. C. high conductivity. D. low conductivity and high melting points.
- Slides: 9