Unit 7 Chemical Equilibrium The Equilibrium Constant Chemical

Unit 7: Chemical Equilibrium The Equilibrium Constant
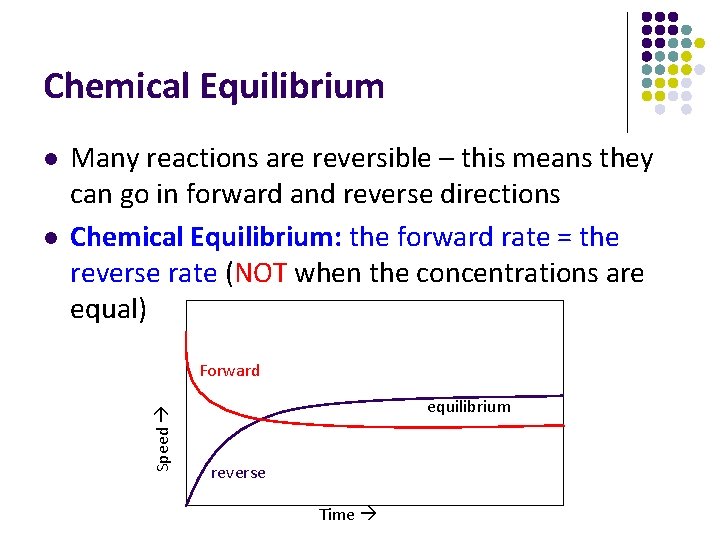
Chemical Equilibrium l Many reactions are reversible – this means they can go in forward and reverse directions Chemical Equilibrium: the forward rate = the reverse rate (NOT when the concentrations are equal) Forward Speed l equilibrium reverse Time

Equilibrium Constant Keq l For a reaction at equilibrium: a. A+b. B ↔ c. C + d. D coefficients from balanced equation molarity l Note: experiments show that equilibrium position does not depend on the amounts of pure solids and liquids present because these concentrations cannot change l Write them as “ 1” in the Keq expression or leave them out!

Equilibrium Constant Keq Example Write the equilibrium constant expression for the following reversible reaction: H 2 (g) + I 2 (g) ↔ 2 HI (g)

Equilibrium Constant Keq Practice #1 Write the equilibrium constant expression for the following reversible reaction: Ca. CO 3 (s) ↔ Ca. O (s) + CO 2 (g)

Equilibrium Constant Keq Practice #2 Write the equilibrium constant expression for the following reversible reaction: hydrogen gas + carbon dioxide gas ↔ carbon monoxide gas + water vapor

Equilibrium Constant Keq Practice #3 Write the equilibrium constant expression for the following reversible reaction: ammonia gas (NH 3) + oxygen gas ↔ water vapor + nitrogen monoxide gas

Equilibrium Constant Keq Practice #4 Write the equilibrium constant expression for the following reversible reaction: ammonia gas (NH 3) + oxygen gas ↔ water vapor + nitrogen monoxide gas

Equilibrium Constant Keq What does the equilibrium constant tell us? l Remember that the equilibrium constant is roughly: l l Bigger value of Keq = product formation favored more Smaller value of Keq = reactant formation favored more

Equilibrium Constant Keq Worksheet – Additonal Problems 1. Which reaction favors product formation the most? 2. Which reaction favors reactant formation the most? 3. For problem #3: Determine [H+] if [OH-] = 0. 037 M. 4. For problem #4: Determine [CO] if [O 2] = 1. 50 M and [CO 2] = 0. 250 M.


Equilibrium Visualizations l http: //www. chem. arizona. edu/~jpollard/fido/fid o. html

Equilibrium Book Problems l l Read Ch. 15 (some parts we won’t cover) Assigned: 15. 2, 15. 4, 15. 6, 15. 9, 15. 16, 15. 52
- Slides: 13