Unit 61 Engineering Thermodynamics Lesson 10 Entropy Objective

Unit 61: Engineering Thermodynamics Lesson 10: Entropy

Objective • The purpose of this lesson is to derive a a thermodynamic property called entropy.

Definition • From previous, reversible incremental work done was given by… d. Wrev = pd. V And the total work done during the process between two states was given by… 2 Wrev = pd. V 1

Definition • Suppose now that a system undergoes the same reversible process between two states 1 and 2 and that during the infinitesimal (incremental) part of the process an amount of heat d. Qrev is transferred to or from the system at temperature T, then a property called entropy can be defined such that the change in entropy during the infinitesimal part of the process is given by…

Definition d. Qrev = Td. S hence d. S = d. Qrev/T thus the total change in entropy for the complete process is found by integrating between the two states… 2 S 2 – S 1 = d. Qrev/T 1

The Carnot Cycle • We often sketch a temperature-entropy diagram for cycles or processes of interest. The Carnot cycle provides a simple display when plotting temperature v entropy.
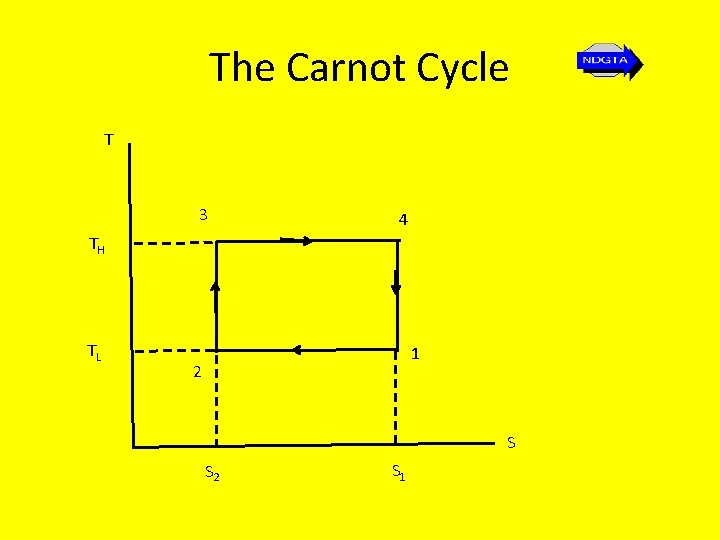
The Carnot Cycle T 3 4 TH TL 1 2 S S 2 S 1

Carnot Cycle • The change in entropy for the first process from state 1 to state 2 is given by… 2 S 2 – S 1 = d. Qrev/T = - QL/TL • The entropy 1 change for the adiabatic process from state 2 to state 3 is zero • For the process from state 3 to state 4 the entropy change is numerically equal to that of the first process • From state 4 to 1 the entropy change is zero

Heat Transfer • The heat transfer during a reversible process can be expressed as… d. Q = Td. S • Hence the area under the curve represents the heat transfer during any reversible process. • Thus the area of the rectangle represents the net heat transfer during the Carnot cycle

Heat Transfer • Since the heat transfer is equal to the work done for a cycle, the area also represents the net work accomplished by the system during the cycle i. e. Qnet = Wnet = ΔTΔS • By considering the 1 st law the equation becomes… Td. S – Pd. V = d. U

Entropy v Enthalpy • Dividing by the mass we have… Tds – Pdv = du Where specific entropy s = S/m • We can also relate entropy change to enthalpy change i. e. dh = du + pdv +vd. P Thus… Tds = dh - vd. P

Entropy for an Ideal Gas ds = dh/T – vd. P/T = Cvd. T/T + Rdv/v Where… du = Cvd. T and Pv = RT Integrating thus gives… s 2 – s 1 = Cvln(T 2/T 1) + Rln(V 2/V 1) Similarly… s 2 – s 1 = Cpln(T 2/T 1) – Rln(P 2/P 1)

Entropy Change • So what does the property of entropy or more importantly to engineers, entropy change signify? • From the discussion of reversibility that an ideal process is one in which friction losses were zero and the process was reversible. • In real thermodynamic systems such as combustion engines, turbines, heat pumps, compressors, etc we try to keep losses to a minimum and these will not only include friction losses by heat transfer losses.

Entropy Change • Thus for real systems we try to make them adiabatic as well as reversible. • Thus for reversible adiabatic (isentropic) processes… – No heat transfer i. e. d. Qrev = 0 and Q = 0, then sthere should be no change in entropy i. e. s 2 s 1 = 0 so that cpln(T 2/T 1) – Rln(P 2/P 1) = 0

Isentropic Process • The ideal process with which comparisons may be made with real processes and cycles is the isentropic process. • Hence the need to understand the concepts of entropy, entropy change and isentropic efficiencies against which real cycles and systems can be measured. • Thus for a process to be isentropic it must be adiabatic and must have constant entropy
- Slides: 15