UNIT 6 PHYSICAL BEHAVIOR OF MATTER I Classification

UNIT 6: PHYSICAL BEHAVIOR OF MATTER
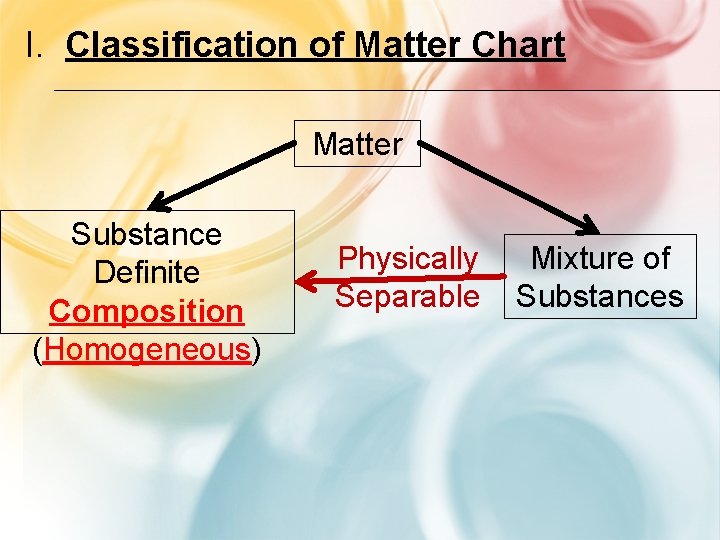
I. Classification of Matter Chart Matter Substance Definite Composition (Homogeneous) Physically Separable Mixture of Substances
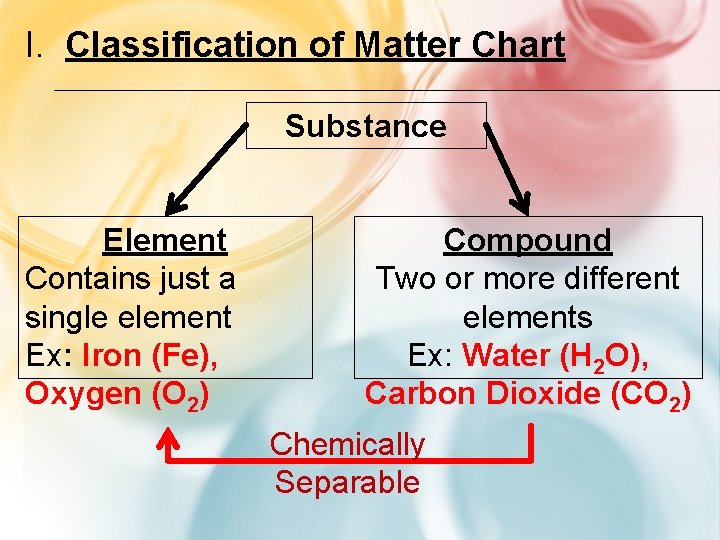
I. Classification of Matter Chart Substance Element Contains just a single element Ex: Iron (Fe), Oxygen (O 2) Compound Two or more different elements Ex: Water (H 2 O), Carbon Dioxide (CO 2) Chemically Separable

CHECKS FOR UNDERSTANDING 1. A) B) C) D) A compound differs from an element in that a compound Is homogeneous Has a definite composition Has a definite melting point Can be decomposed by a chemical reaction 2. Which of the following substances cannot be separated by chemical change? A) Nitrogen (g) B) Sodium chloride (s) C) Carbon dioxide (g) D) Magnesium Sulfate (aq)
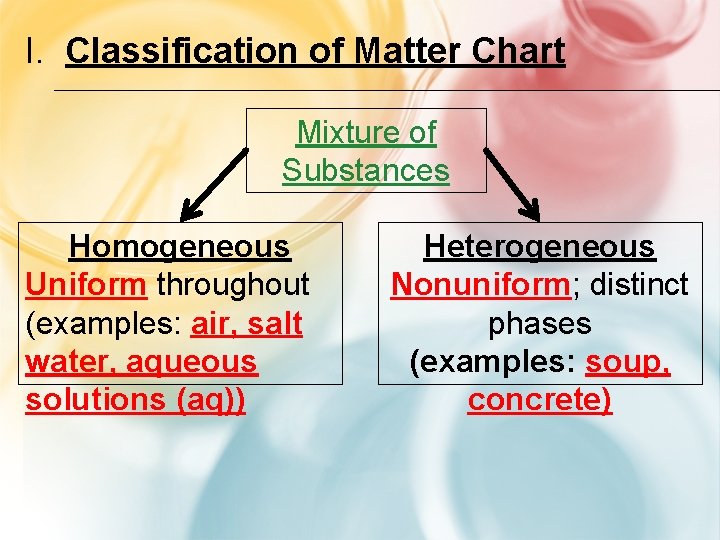
I. Classification of Matter Chart Mixture of Substances Homogeneous Uniform throughout (examples: air, salt water, aqueous solutions (aq)) Heterogeneous Nonuniform; distinct phases (examples: soup, concrete)

CHECK FOR UNDERSTANDING 1. A pure substance that is composed only of identical atoms is classified as… A) A compound B) An element C) A heterogeneous mixture D) A homogeneous mixture 2. A) B) C) D) A heterogeneous material may be… An element A compound A mixture Pure substance Once done work on page 2 of Unit 6 Work
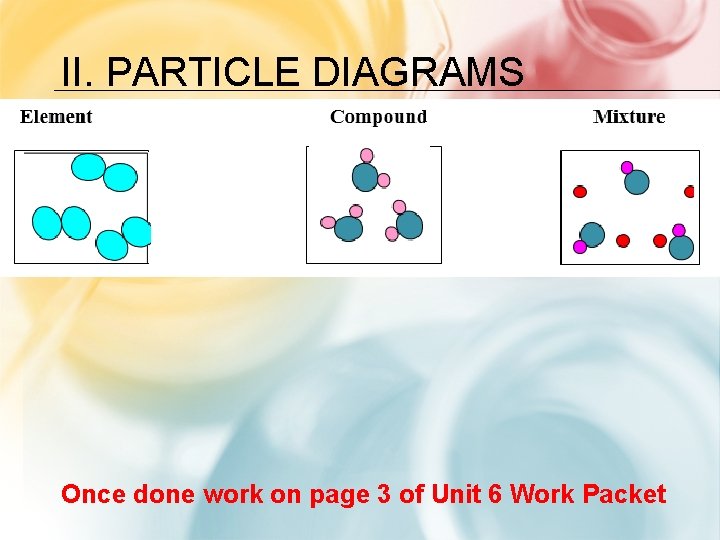
II. PARTICLE DIAGRAMS Once done work on page 3 of Unit 6 Work Packet

III. SEPARATING MATTER • Certain types of matter can be separated using various methods. • Monatomic Elements - _______ be CANNOT decomposed (broken apart) using CHEMICAL PHYSICAL _______ or _______ means. • Diatomic Elements and Compounds (ie – O 2 CHEMICAL MEANS and H 2 O) – can be decomposed using _________ only

III. SEPARATING MATTER Mixtures – can be separated using MEANS PHYSICAL __________ PARTICLE Separation by Filtration – SIZE _______ Separation BOILING by Evaporation – POINT ______ POLARITY Separation by Chromatography – ______ Separation by _______ BOILING Distillation – POINT

CHECK FOR UNDERSTANDING 1. Which of the substances could be decomposed by a chemical change? A) sodium B) aluminum C) magnesium D) ammonia 2. A sample of a material is passed through a filter paper. A white deposit remains on the paper, and is a heterogeneous a clear liquid passes. It through. The clear liquid is mixture then evaporated, leaving a white residue. What can you determine about the nature of the sample? In a mixture the elements are not bonded with each are other and of can bedifferences physically between separated. 3. What some the a In a compound elements and can mixture of ironthe and oxygen are andbonded compound Once done work on page 4 of Unit 6 Work Packet

THINK ABOUT THIS 1. What would a particle diagram look like for each of these phases? 2. What happens to the spacing and speed of particles at each of the phases? SOLID LIQUID GAS Once done work on page 4 of Unit 6 Work Packet

IV. FORMS OF MECHANICAL ENERGY Kinetic Energy of movement (similar to temperature) (how fast atoms are moving) Potential Energy Stored energy (energy of position) More spread out (gas) = High PE Closer together (solid) = Low PE
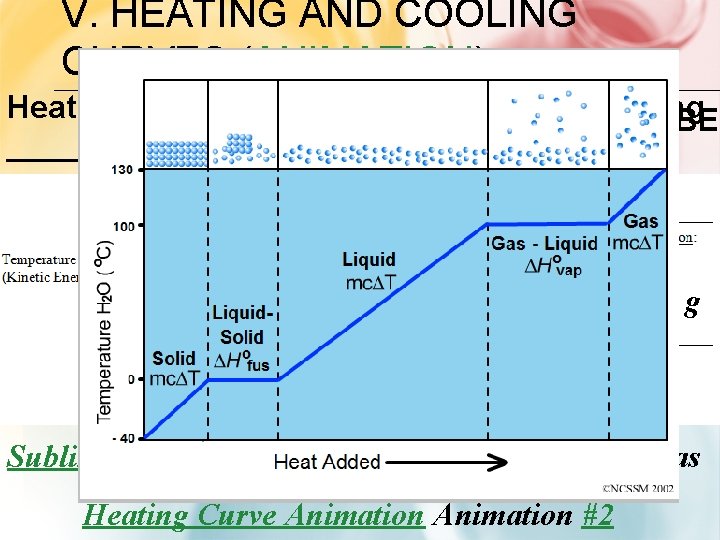
V. HEATING AND COOLING CURVES (ANIMATION) Heating Curve: ______ - Energy is being ENDOTHERMIC ABSORBED ____ l g s l gas liquid s g solid Sublimation (video)- Solid changes directly to a gas Heating Curve Animation #2
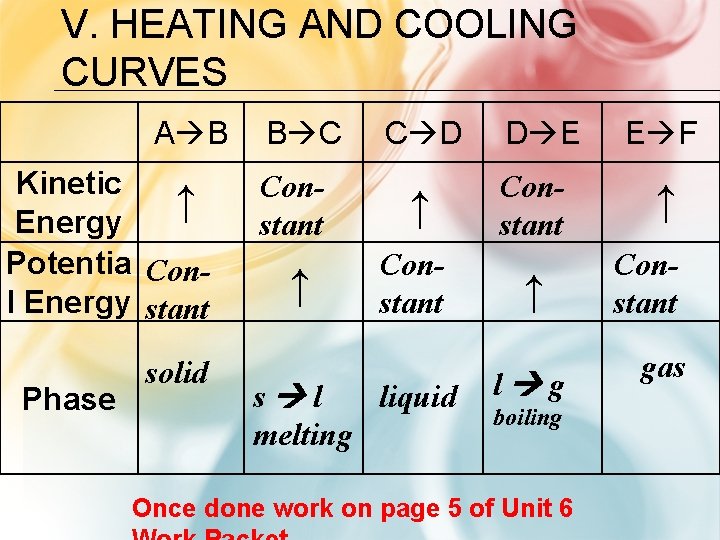
V. HEATING AND COOLING CURVES A B Kinetic ↑ Energy Potentia Conl Energy stant Phase solid B C C D Constant ↑ ↑ Constant s l liquid melting D E Constant ↑ l g boiling Once done work on page 5 of Unit 6 E F ↑ Constant gas

CHECK FOR UNDERSTANDING 1. A substance begins to a melt. What happens to the potential and kinetic energy? PE increase, KE stays the same 2. The temperature of a substance refers to what type of energy? Kinetic energy 3. How does the speed and space of water molecules compare when in a liquid phase to a gas phase Molecules move faster and more spread out in gas phase
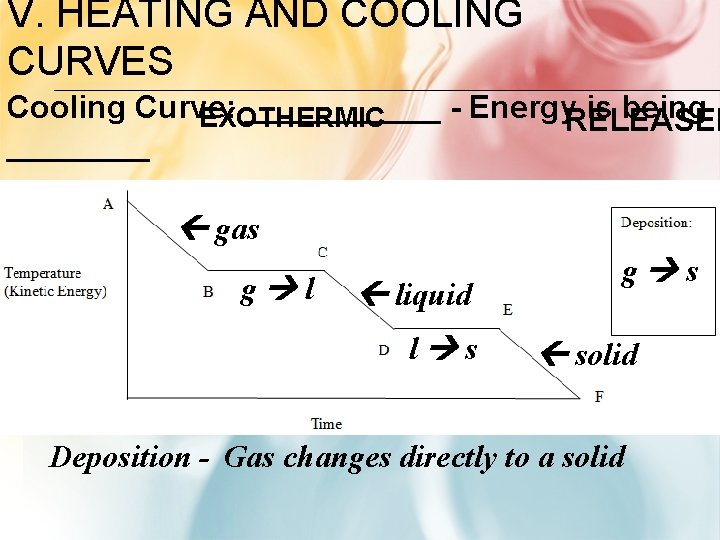
V. HEATING AND COOLING CURVES Cooling Curve: ______ - Energy. RELEASED is being EXOTHERMIC ____ gas g l liquid l s g s solid Deposition - Gas changes directly to a solid
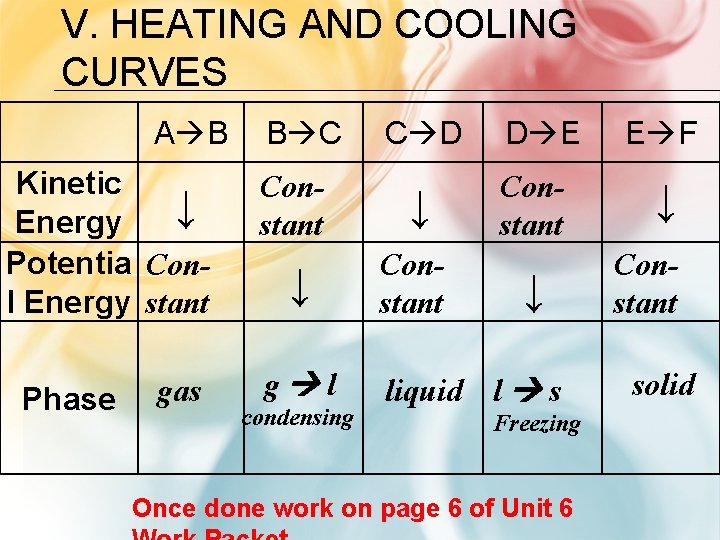
V. HEATING AND COOLING CURVES A B Kinetic ↓ Energy Potentia Conl Energy stant Phase gas B C C D Constant ↓ ↓ Constant g l condensing D E Constant ↓ liquid l s Freezing Once done work on page 6 of Unit 6 E F ↓ Constant solid

CHECK FOR UNDERSTANDING 1. As a substance condenses, what happens to its potential and kinetic energy? PE decreases, KE stays the same 2. What phase is a substance in when it has its highest kinetic energy? gas 3. How does the speed and space of water molecules compare when in a liquid phase to a solid phase Molecules move slower and are closer together in solid phase

VI. TEMPERATURE VS. HEAT Amount of energy Average kinetic energy transferred from one of its particles (how substance to fast they’re moving) another Joules (J) or Celsius (o. C) or Kelvin (K) Calories (cal) (K = o. C + 273) 1 cal = 4. 18 J T q

VI. TEMPERATURE HEAT Temperature Scales (See Ref. VS. Tabs. ): K = o. C + 273 K = Kelvin Celsius o. C = degrees Convert: 200 degrees Celsius to Kelvin o o K = C + 273 K = 200 C + 273 = 473 K Law of Conservation of Energy: Energy (heat) cannot be created or destroyed. Energy (heat) can be TRANSFERRED. Heat Transfer: HEAT ALWAYS MOVES FROM WARMER OBJECTS TO COLDER OBJECTS

CHECK FOR UNDERSTANDING 1. You wake up in the morning and your barefoot touches the ceramic floor and it feels cold. Explain which way heat is being transferred. Heat moves from your body (warm) to the floor (cold) 2. You are cooking pasta in a boiling metal pot of water. You grab the metal handles with your bare hands (ouch!). Explain which way heat is being transferred. Heat moves from metal handles (warm) to your hands (cold). 3. Why do you feel cold after you get out of a hot shower. (link)

VII. ENDOTHERMIC AND • Energy is either absorbed or released in EXOTHERMIC (REVISITED) chemical reactions • Remember: - Breaking bonds is __________ ENDOTHERMIC - Heat is ______ the reaction from the ENTERING surroundings - Ex) heat + Br 2 Br + Br EXOTHERMIC - Creating bonds is _________ EXITING - Heat is ______ the reaction into the surroundings - Ex) N+ N N 2+ energy

VII. ENDOTHERMIC AND • Where does the heat come from (or go to)? EXOTHERMIC (REVISITED) ___________ The surroundings • For exothermic reactions, heat (energy) leaves the reaction and moves into the _____________. Therefore, warmer makingsurroundings the surrounding temperature _________ • Exothermic Chemical Equation: Reactant(s) Product(s) + HEAT

VII. ENDOTHERMIC AND (REVISITED) • For. EXOTHERMIC endothermic reactions, heat (energy) leaves the surroundings and moves Into the reaction _____________. Therefore, colder making the surrounding temperature _________ • Endothermic. Reactant(s) Chemical Equation: + HEAT Product(s) ENDOTHERMIC REACTION

VII. ENDOTHERMIC AND • Enthalpy: It is a measure of the heat released or absorbed in a EXOTHERMIC (REVISITED) reaction. If heat is released then the reaction is an exothermic reaction, heat will be on the products side and the ΔH will be negative. If heat is absorbed then the reaction is an endothermic reaction, heat will appear on the reactants side and the ΔH will be positive. (see Table ____ on Reference Tables). I Examples: Tell whether each of the following reactions are endothermic or exothermic (you may have to use table I). Then tell whether the temperature of the surroundings increases or decreases as a result. Endo/Exothermic _______Surroundings Exo (-∆H) _____ warmer C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O + heat

VII. ENDOTHERMIC AND EXOTHERMIC (REVISITED) Endo/Exothermic _______Surroundings 1. ______C 6 H 12 O 6 (s) + 6 O 2(g) 6 CO 2 (g) + 6 H 2 O + heat ______ exo(-∆H) warmer 2. endo(+∆H) _____ 2 H 2 O + 484 k. J 2 H 2 + O 2 colder ________ exo(-∆H) warmer 6. _____ 2 KCl. O 3(s) 2 KCl(s) + O 2(g) + endo(+∆H) colder heat ________ 7. _____ H+(aq) + C 2 H 3 O 2(aq) + heat HC 2 H 3 O 2(l) ________

VIII. VAPOR PRESSURE (VP) • Vapor Pressure: (VP) Pressure a liquid “feels” pushing it to evaporate (turn to gas) • ______ VP = _____________ HIGH Evaporates easily Example: Ethanol has higher VP than H O, so it evaporates more easily 2 • Factors for Vapor Pressure Strength of intermolecular force: molecules held together by dipole 1. dipole (polar) have lower VP than Van der Waals (nonpolar) (H 2 O does not evaporate as fast as methane (CH 4)) 2. Temperature/Pressure: increase temp. , increase VP

• Vapor Pressure of Four Liquids (See Table _____) VIII. VAPOR PRESSURE (VP) H • Questions: k. Pa (101. 3 k. Pa = 1 atm = 760 mm. Hg = 760 torr. ) 1. Measured What isin: the vapor pressure of propanone at 35 o. C? _______ 48 k. Pa 2. What temperature does water boil at if the pressure is 70 90 degrees C k. Pa? ______ 117 degrees C 3. What is the normal boiling point of ethanoic acid? _____ Occurs because solids have very weak IMF (Intermolecular Forces). Sublimation: They go directly from s g and have HIGH VP

CHECK FOR UNDERSTANDING 1. How could you change the boiling point temperature of a substance without adding anything to the substance? Change your altitude (higher altitude (lower pressure), lower boiling point) 2. You spill a glass of water on the floor. How could you get the water to evaporate faster, without using a mop or something to soak it up? Increase the temperature of the room. Spread out the puddle (increase surface area).
- Slides: 29