Unit 6 Changing the DNA Blueprint Chapter 28

Unit 6: Changing the DNA Blueprint Chapter 28 - Bacterial Genetics Molecular Biology, 3 rd edition by Clark, Pazdernik and Mc. Gehee Copyright © 2019 by Academic Cell. All rights reserved.
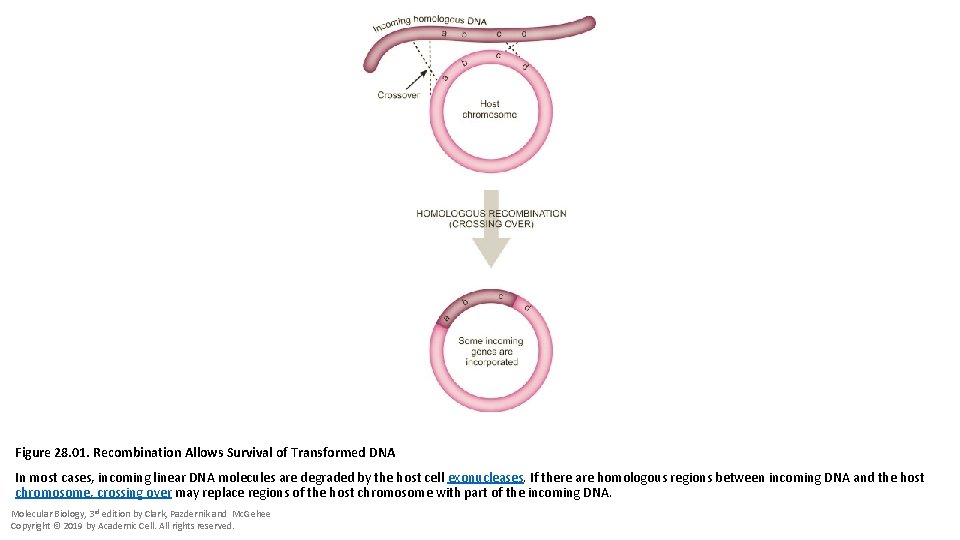
Figure 28. 01. Recombination Allows Survival of Transformed DNA In most cases, incoming linear DNA molecules are degraded by the host cell exonucleases. If there are homologous regions between incoming DNA and the host chromosome, crossing over may replace regions of the host chromosome with part of the incoming DNA. Molecular Biology, 3 rd edition by Clark, Pazdernik and Mc. Gehee Copyright © 2019 by Academic Cell. All rights reserved.
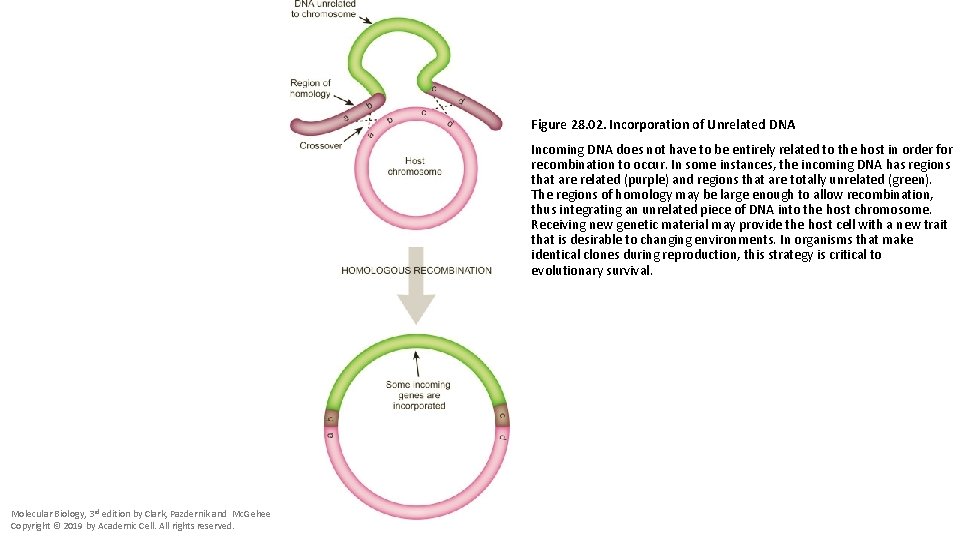
Figure 28. 02. Incorporation of Unrelated DNA Incoming DNA does not have to be entirely related to the host in order for recombination to occur. In some instances, the incoming DNA has regions that are related (purple) and regions that are totally unrelated (green). The regions of homology may be large enough to allow recombination, thus integrating an unrelated piece of DNA into the host chromosome. Receiving new genetic material may provide the host cell with a new trait that is desirable to changing environments. In organisms that make identical clones during reproduction, this strategy is critical to evolutionary survival. Molecular Biology, 3 rd edition by Clark, Pazdernik and Mc. Gehee Copyright © 2019 by Academic Cell. All rights reserved.
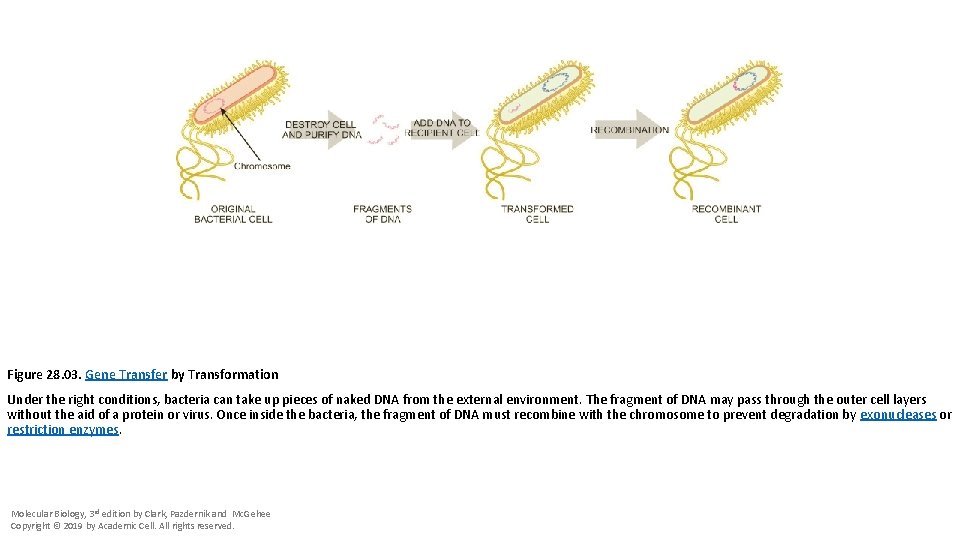
Figure 28. 03. Gene Transfer by Transformation Under the right conditions, bacteria can take up pieces of naked DNA from the external environment. The fragment of DNA may pass through the outer cell layers without the aid of a protein or virus. Once inside the bacteria, the fragment of DNA must recombine with the chromosome to prevent degradation by exonucleases or restriction enzymes. Molecular Biology, 3 rd edition by Clark, Pazdernik and Mc. Gehee Copyright © 2019 by Academic Cell. All rights reserved.
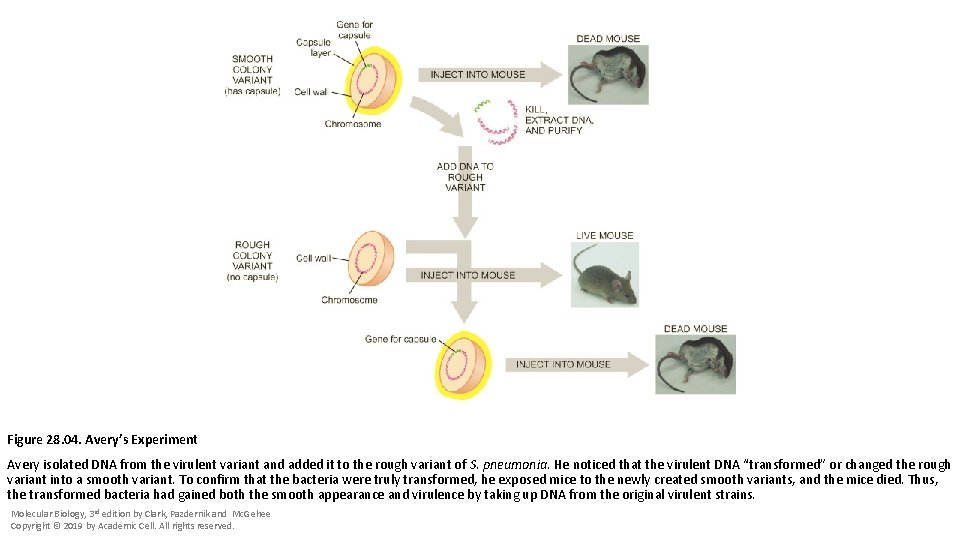
Figure 28. 04. Avery’s Experiment Avery isolated DNA from the virulent variant and added it to the rough variant of S. pneumonia. He noticed that the virulent DNA “transformed” or changed the rough variant into a smooth variant. To confirm that the bacteria were truly transformed, he exposed mice to the newly created smooth variants, and the mice died. Thus, the transformed bacteria had gained both the smooth appearance and virulence by taking up DNA from the original virulent strains. Molecular Biology, 3 rd edition by Clark, Pazdernik and Mc. Gehee Copyright © 2019 by Academic Cell. All rights reserved.
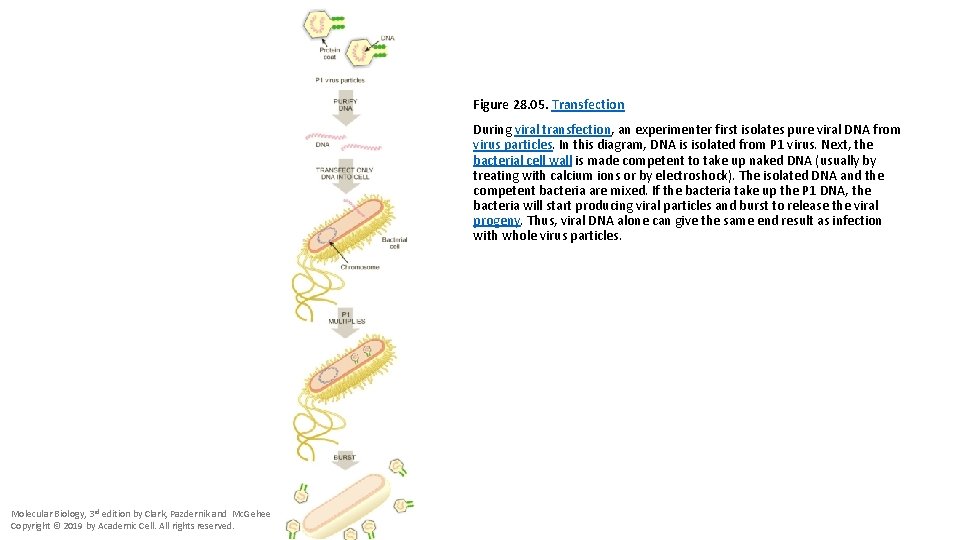
Figure 28. 05. Transfection During viral transfection, an experimenter first isolates pure viral DNA from virus particles. In this diagram, DNA is isolated from P 1 virus. Next, the bacterial cell wall is made competent to take up naked DNA (usually by treating with calcium ions or by electroshock). The isolated DNA and the competent bacteria are mixed. If the bacteria take up the P 1 DNA, the bacteria will start producing viral particles and burst to release the viral progeny. Thus, viral DNA alone can give the same end result as infection with whole virus particles. Molecular Biology, 3 rd edition by Clark, Pazdernik and Mc. Gehee Copyright © 2019 by Academic Cell. All rights reserved.
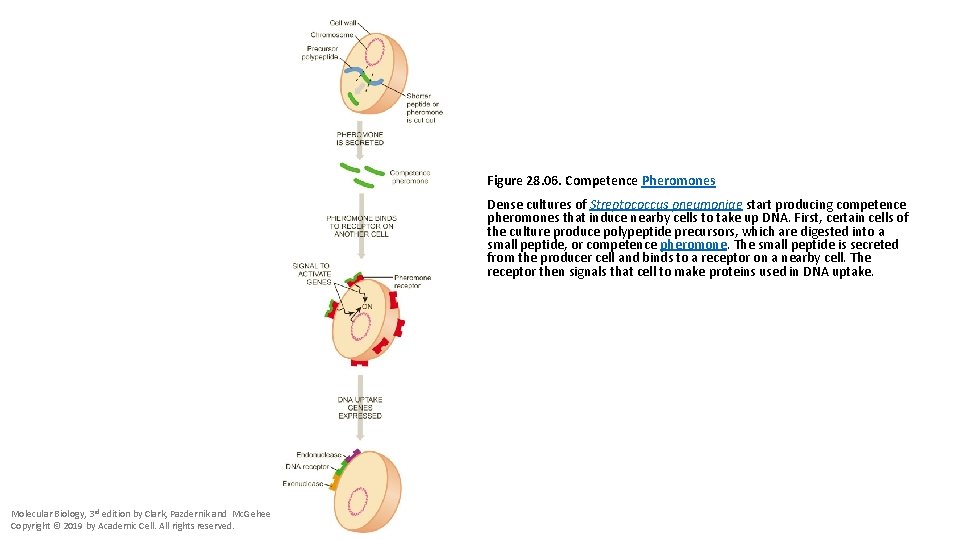
Figure 28. 06. Competence Pheromones Dense cultures of Streptococcus pneumoniae start producing competence pheromones that induce nearby cells to take up DNA. First, certain cells of the culture produce polypeptide precursors, which are digested into a small peptide, or competence pheromone. The small peptide is secreted from the producer cell and binds to a receptor on a nearby cell. The receptor then signals that cell to make proteins used in DNA uptake. Molecular Biology, 3 rd edition by Clark, Pazdernik and Mc. Gehee Copyright © 2019 by Academic Cell. All rights reserved.
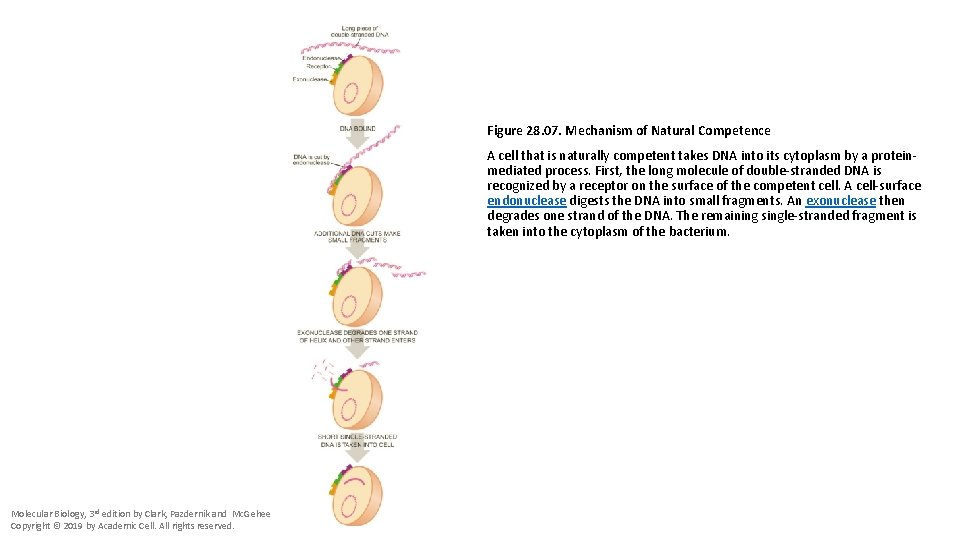
Figure 28. 07. Mechanism of Natural Competence A cell that is naturally competent takes DNA into its cytoplasm by a proteinmediated process. First, the long molecule of double-stranded DNA is recognized by a receptor on the surface of the competent cell. A cell-surface endonuclease digests the DNA into small fragments. An exonuclease then degrades one strand of the DNA. The remaining single-stranded fragment is taken into the cytoplasm of the bacterium. Molecular Biology, 3 rd edition by Clark, Pazdernik and Mc. Gehee Copyright © 2019 by Academic Cell. All rights reserved.
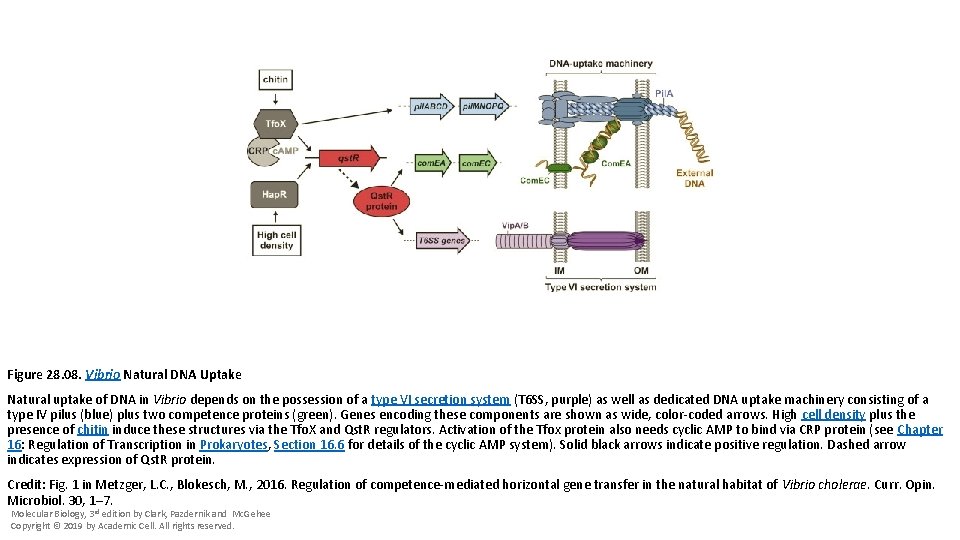
Figure 28. 08. Vibrio Natural DNA Uptake Natural uptake of DNA in Vibrio depends on the possession of a type VI secretion system (T 6 SS, purple) as well as dedicated DNA uptake machinery consisting of a type IV pilus (blue) plus two competence proteins (green). Genes encoding these components are shown as wide, color-coded arrows. High cell density plus the presence of chitin induce these structures via the Tfo. X and Qst. R regulators. Activation of the Tfox protein also needs cyclic AMP to bind via CRP protein (see Chapter 16: Regulation of Transcription in Prokaryotes, Section 16. 6 for details of the cyclic AMP system). Solid black arrows indicate positive regulation. Dashed arrow indicates expression of Qst. R protein. Credit: Fig. 1 in Metzger, L. C. , Blokesch, M. , 2016. Regulation of competence-mediated horizontal gene transfer in the natural habitat of Vibrio cholerae. Curr. Opin. Microbiol. 30, 1– 7. Molecular Biology, 3 rd edition by Clark, Pazdernik and Mc. Gehee Copyright © 2019 by Academic Cell. All rights reserved.
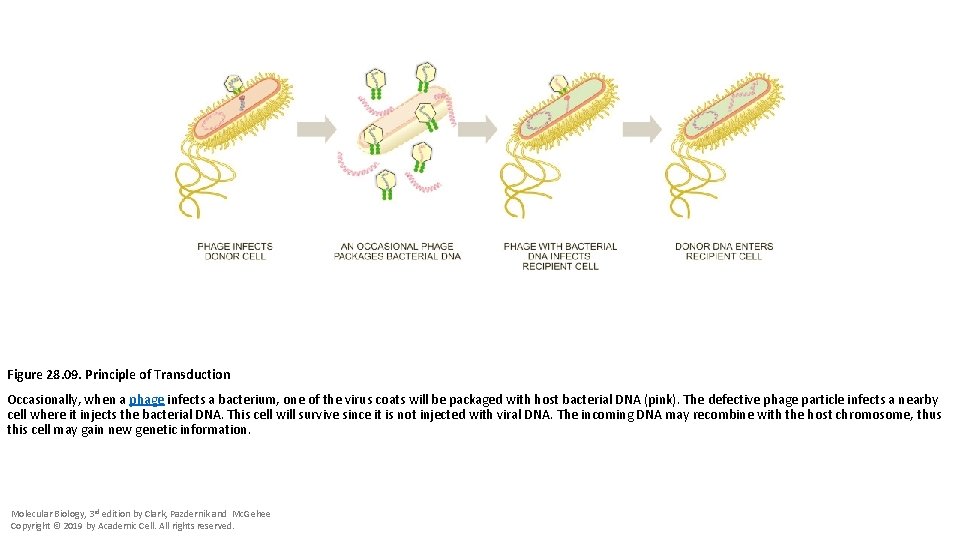
Figure 28. 09. Principle of Transduction Occasionally, when a phage infects a bacterium, one of the virus coats will be packaged with host bacterial DNA (pink). The defective phage particle infects a nearby cell where it injects the bacterial DNA. This cell will survive since it is not injected with viral DNA. The incoming DNA may recombine with the host chromosome, thus this cell may gain new genetic information. Molecular Biology, 3 rd edition by Clark, Pazdernik and Mc. Gehee Copyright © 2019 by Academic Cell. All rights reserved.
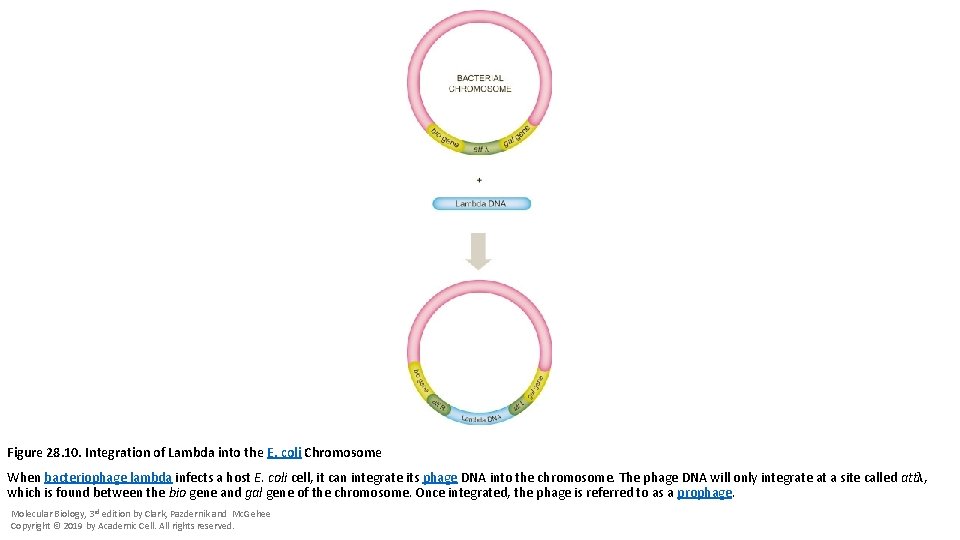
Figure 28. 10. Integration of Lambda into the E. coli Chromosome When bacteriophage lambda infects a host E. coli cell, it can integrate its phage DNA into the chromosome. The phage DNA will only integrate at a site called attλ, which is found between the bio gene and gal gene of the chromosome. Once integrated, the phage is referred to as a prophage. Molecular Biology, 3 rd edition by Clark, Pazdernik and Mc. Gehee Copyright © 2019 by Academic Cell. All rights reserved.
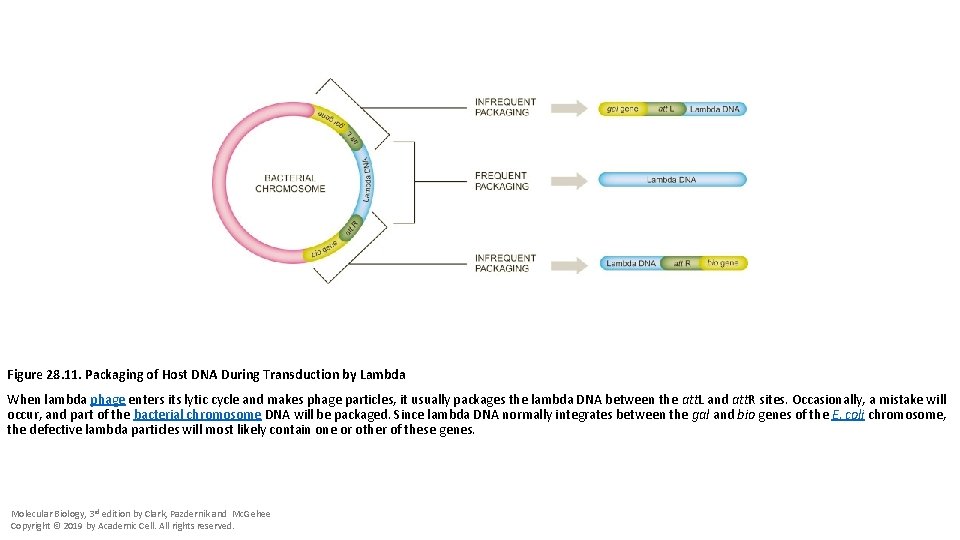
Figure 28. 11. Packaging of Host DNA During Transduction by Lambda When lambda phage enters its lytic cycle and makes phage particles, it usually packages the lambda DNA between the att. L and att. R sites. Occasionally, a mistake will occur, and part of the bacterial chromosome DNA will be packaged. Since lambda DNA normally integrates between the gal and bio genes of the E. coli chromosome, the defective lambda particles will most likely contain one or other of these genes. Molecular Biology, 3 rd edition by Clark, Pazdernik and Mc. Gehee Copyright © 2019 by Academic Cell. All rights reserved.
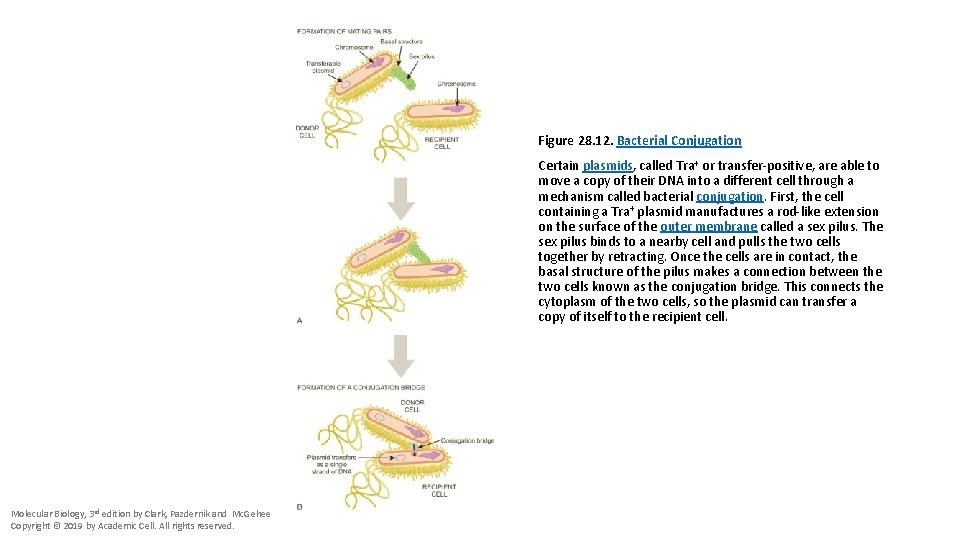
Figure 28. 12. Bacterial Conjugation Certain plasmids, called Tra+ or transfer-positive, are able to move a copy of their DNA into a different cell through a mechanism called bacterial conjugation. First, the cell containing a Tra+ plasmid manufactures a rod-like extension on the surface of the outer membrane called a sex pilus. The sex pilus binds to a nearby cell and pulls the two cells together by retracting. Once the cells are in contact, the basal structure of the pilus makes a connection between the two cells known as the conjugation bridge. This connects the cytoplasm of the two cells, so the plasmid can transfer a copy of itself to the recipient cell. Molecular Biology, 3 rd edition by Clark, Pazdernik and Mc. Gehee Copyright © 2019 by Academic Cell. All rights reserved.
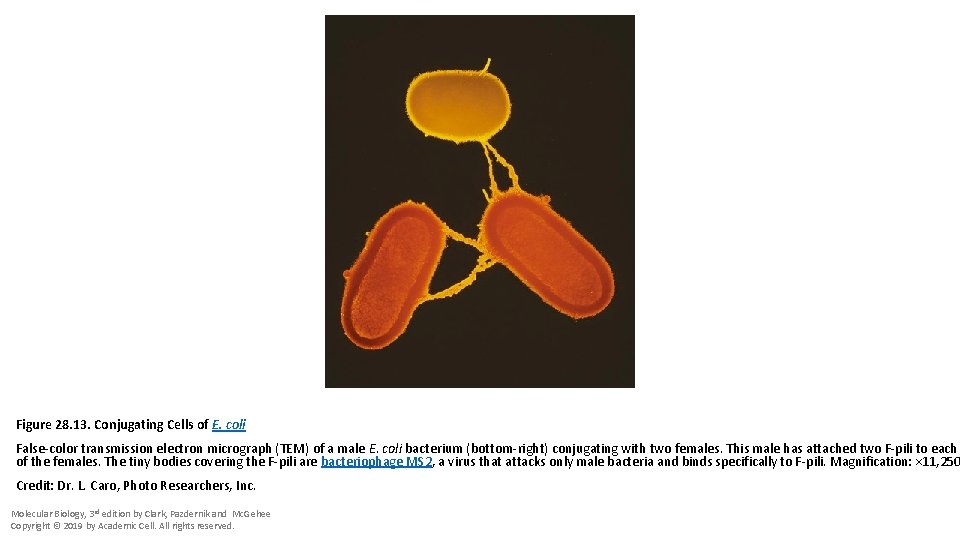
Figure 28. 13. Conjugating Cells of E. coli False-color transmission electron micrograph (TEM) of a male E. coli bacterium (bottom-right) conjugating with two females. This male has attached two F-pili to each of the females. The tiny bodies covering the F-pili are bacteriophage MS 2, a virus that attacks only male bacteria and binds specifically to F-pili. Magnification: × 11, 250 Credit: Dr. L. Caro, Photo Researchers, Inc. Molecular Biology, 3 rd edition by Clark, Pazdernik and Mc. Gehee Copyright © 2019 by Academic Cell. All rights reserved.
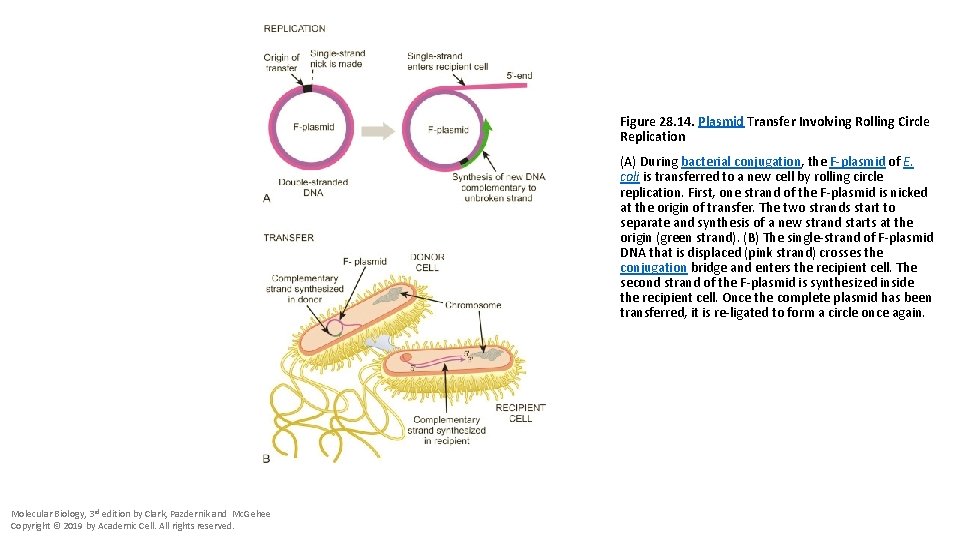
Figure 28. 14. Plasmid Transfer Involving Rolling Circle Replication (A) During bacterial conjugation, the F-plasmid of E. coli is transferred to a new cell by rolling circle replication. First, one strand of the F-plasmid is nicked at the origin of transfer. The two strands start to separate and synthesis of a new strand starts at the origin (green strand). (B) The single-strand of F-plasmid DNA that is displaced (pink strand) crosses the conjugation bridge and enters the recipient cell. The second strand of the F-plasmid is synthesized inside the recipient cell. Once the complete plasmid has been transferred, it is re-ligated to form a circle once again. Molecular Biology, 3 rd edition by Clark, Pazdernik and Mc. Gehee Copyright © 2019 by Academic Cell. All rights reserved.
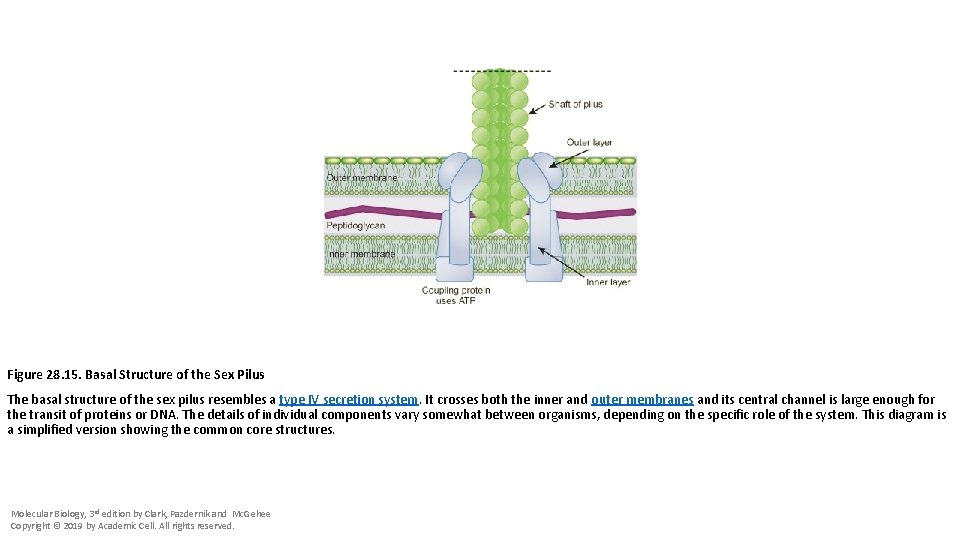
Figure 28. 15. Basal Structure of the Sex Pilus The basal structure of the sex pilus resembles a type IV secretion system. It crosses both the inner and outer membranes and its central channel is large enough for the transit of proteins or DNA. The details of individual components vary somewhat between organisms, depending on the specific role of the system. This diagram is a simplified version showing the common core structures. Molecular Biology, 3 rd edition by Clark, Pazdernik and Mc. Gehee Copyright © 2019 by Academic Cell. All rights reserved.
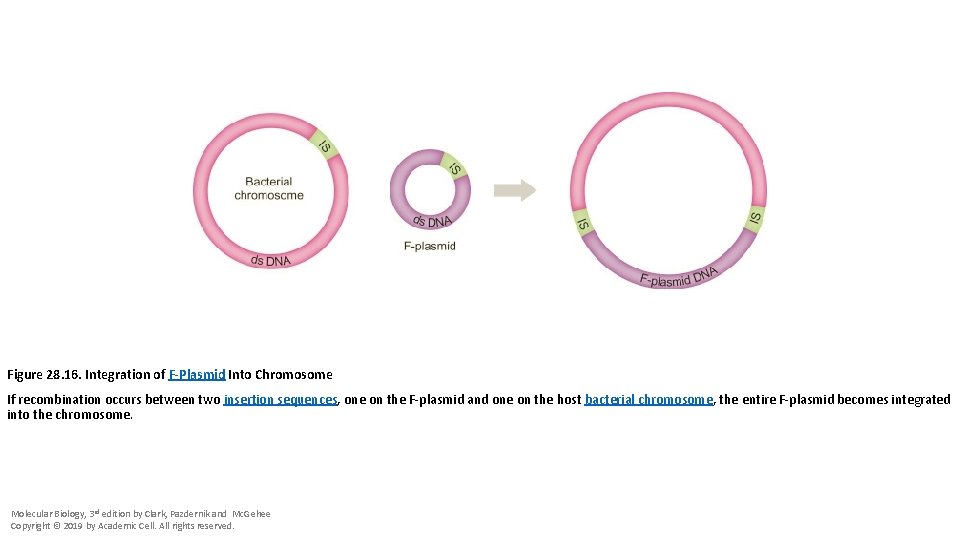
Figure 28. 16. Integration of F-Plasmid Into Chromosome If recombination occurs between two insertion sequences, one on the F-plasmid and one on the host bacterial chromosome, the entire F-plasmid becomes integrated into the chromosome. Molecular Biology, 3 rd edition by Clark, Pazdernik and Mc. Gehee Copyright © 2019 by Academic Cell. All rights reserved.
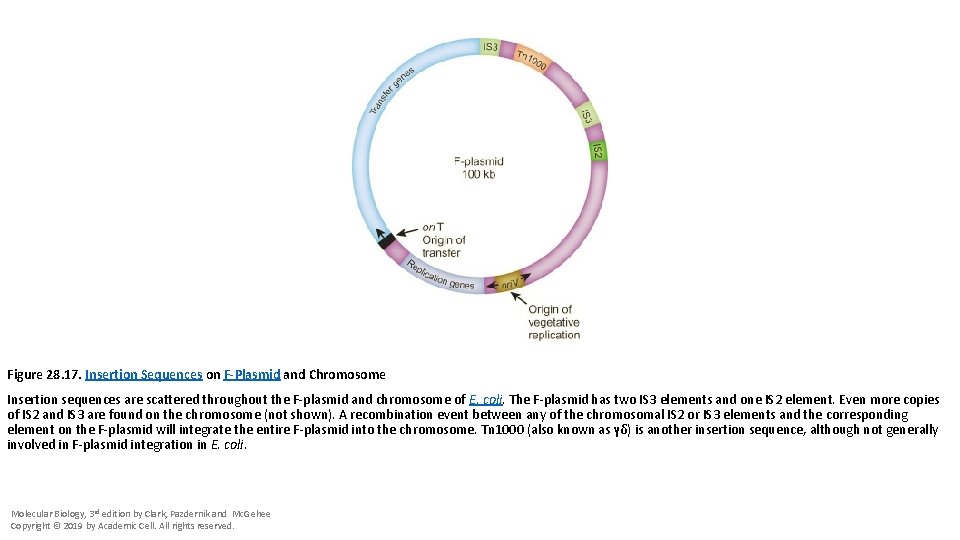
Figure 28. 17. Insertion Sequences on F-Plasmid and Chromosome Insertion sequences are scattered throughout the F-plasmid and chromosome of E. coli. The F-plasmid has two IS 3 elements and one IS 2 element. Even more copies of IS 2 and IS 3 are found on the chromosome (not shown). A recombination event between any of the chromosomal IS 2 or IS 3 elements and the corresponding element on the F-plasmid will integrate the entire F-plasmid into the chromosome. Tn 1000 (also known as γδ) is another insertion sequence, although not generally involved in F-plasmid integration in E. coli. Molecular Biology, 3 rd edition by Clark, Pazdernik and Mc. Gehee Copyright © 2019 by Academic Cell. All rights reserved.
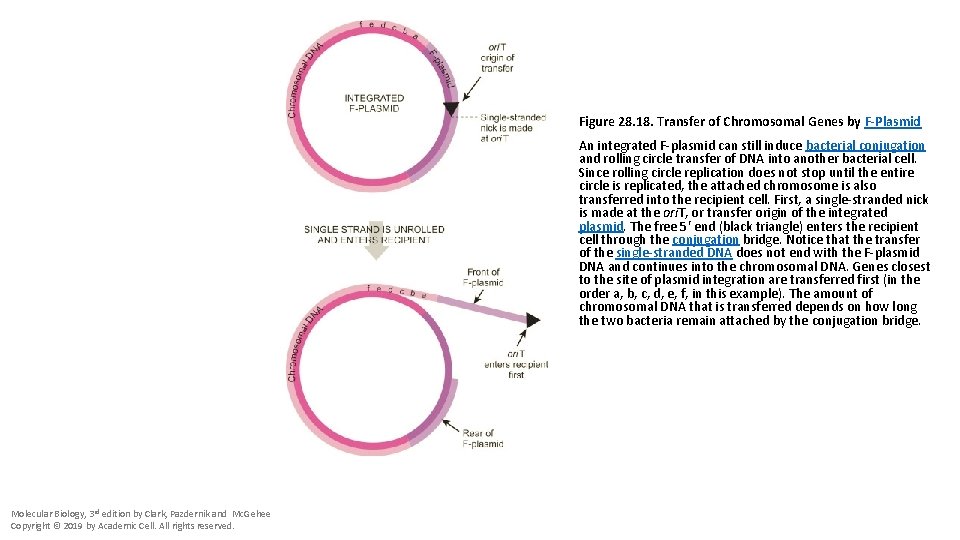
Figure 28. 18. Transfer of Chromosomal Genes by F-Plasmid An integrated F-plasmid can still induce bacterial conjugation and rolling circle transfer of DNA into another bacterial cell. Since rolling circle replication does not stop until the entire circle is replicated, the attached chromosome is also transferred into the recipient cell. First, a single-stranded nick is made at the ori. T, or transfer origin of the integrated plasmid. The free 5′ end (black triangle) enters the recipient cell through the conjugation bridge. Notice that the transfer of the single-stranded DNA does not end with the F-plasmid DNA and continues into the chromosomal DNA. Genes closest to the site of plasmid integration are transferred first (in the order a, b, c, d, e, f, in this example). The amount of chromosomal DNA that is transferred depends on how long the two bacteria remain attached by the conjugation bridge. Molecular Biology, 3 rd edition by Clark, Pazdernik and Mc. Gehee Copyright © 2019 by Academic Cell. All rights reserved.
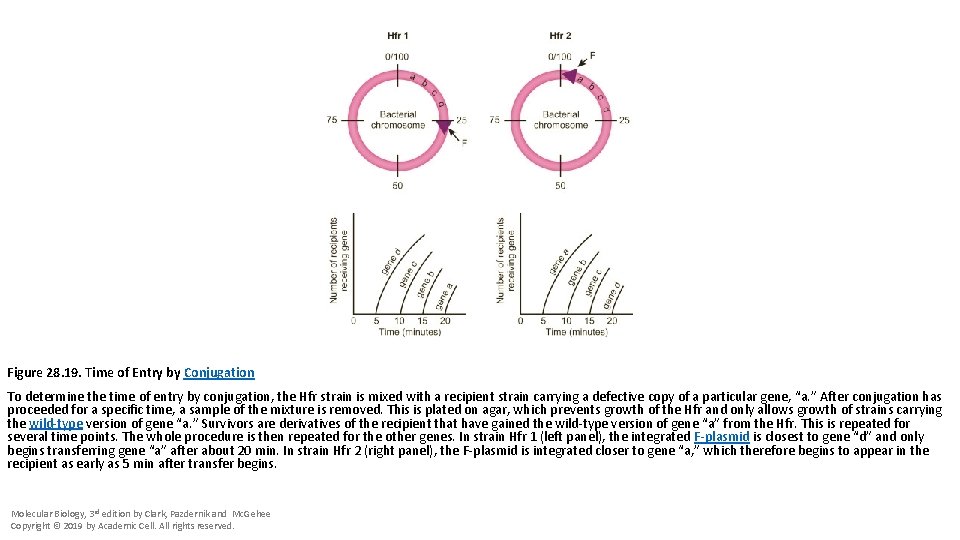
Figure 28. 19. Time of Entry by Conjugation To determine the time of entry by conjugation, the Hfr strain is mixed with a recipient strain carrying a defective copy of a particular gene, “a. ” After conjugation has proceeded for a specific time, a sample of the mixture is removed. This is plated on agar, which prevents growth of the Hfr and only allows growth of strains carrying the wild-type version of gene “a. ” Survivors are derivatives of the recipient that have gained the wild-type version of gene “a” from the Hfr. This is repeated for several time points. The whole procedure is then repeated for the other genes. In strain Hfr 1 (left panel), the integrated F-plasmid is closest to gene “d” and only begins transferring gene “a” after about 20 min. In strain Hfr 2 (right panel), the F-plasmid is integrated closer to gene “a, ” which therefore begins to appear in the recipient as early as 5 min after transfer begins. Molecular Biology, 3 rd edition by Clark, Pazdernik and Mc. Gehee Copyright © 2019 by Academic Cell. All rights reserved.
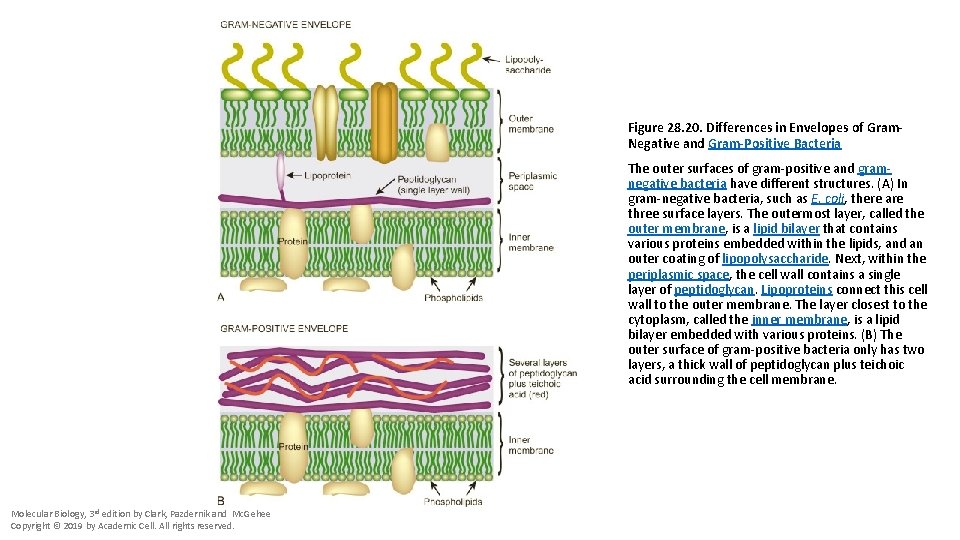
Figure 28. 20. Differences in Envelopes of Gram. Negative and Gram-Positive Bacteria The outer surfaces of gram-positive and gramnegative bacteria have different structures. (A) In gram-negative bacteria, such as E. coli, there are three surface layers. The outermost layer, called the outer membrane, is a lipid bilayer that contains various proteins embedded within the lipids, and an outer coating of lipopolysaccharide. Next, within the periplasmic space, the cell wall contains a single layer of peptidoglycan. Lipoproteins connect this cell wall to the outer membrane. The layer closest to the cytoplasm, called the inner membrane, is a lipid bilayer embedded with various proteins. (B) The outer surface of gram-positive bacteria only has two layers, a thick wall of peptidoglycan plus teichoic acid surrounding the cell membrane. Molecular Biology, 3 rd edition by Clark, Pazdernik and Mc. Gehee Copyright © 2019 by Academic Cell. All rights reserved.
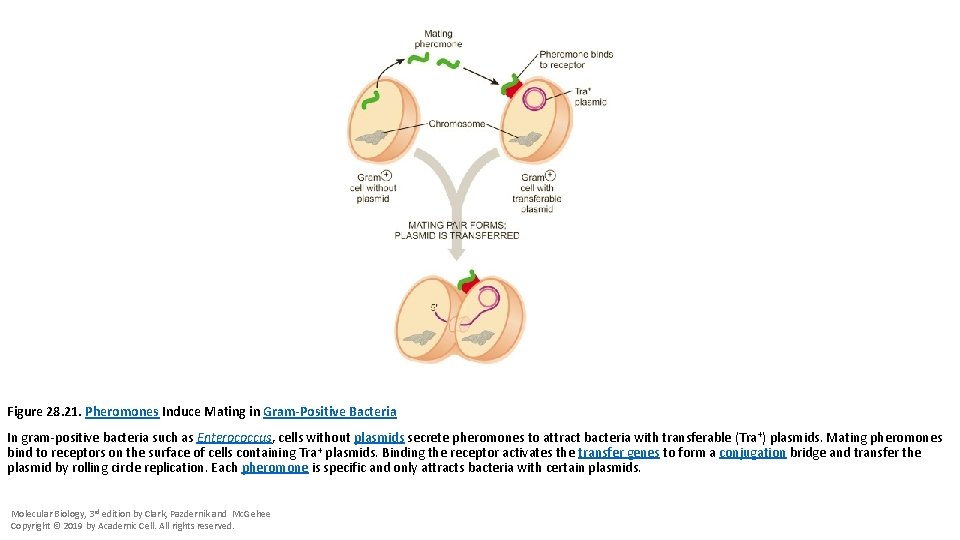
Figure 28. 21. Pheromones Induce Mating in Gram-Positive Bacteria In gram-positive bacteria such as Enterococcus, cells without plasmids secrete pheromones to attract bacteria with transferable (Tra+) plasmids. Mating pheromones bind to receptors on the surface of cells containing Tra+ plasmids. Binding the receptor activates the transfer genes to form a conjugation bridge and transfer the plasmid by rolling circle replication. Each pheromone is specific and only attracts bacteria with certain plasmids. Molecular Biology, 3 rd edition by Clark, Pazdernik and Mc. Gehee Copyright © 2019 by Academic Cell. All rights reserved.
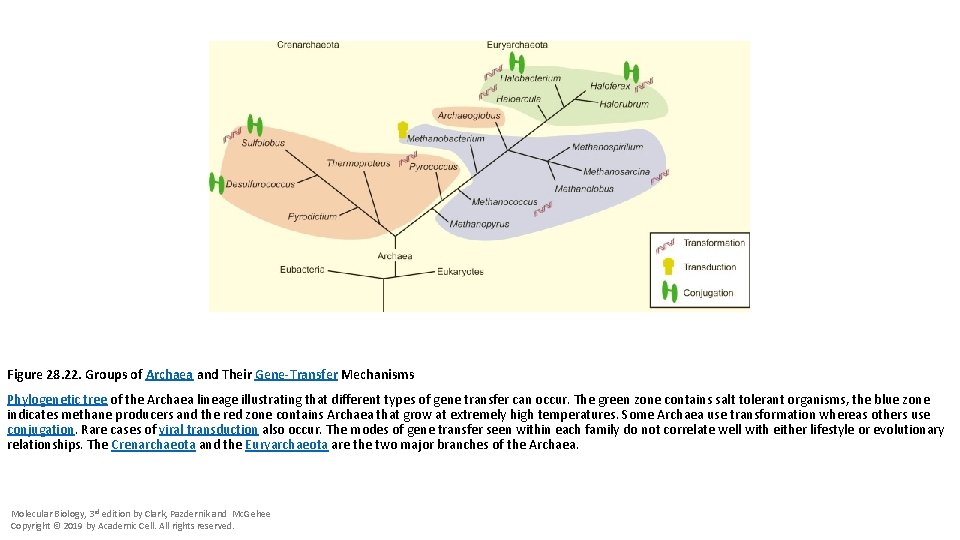
Figure 28. 22. Groups of Archaea and Their Gene-Transfer Mechanisms Phylogenetic tree of the Archaea lineage illustrating that different types of gene transfer can occur. The green zone contains salt tolerant organisms, the blue zone indicates methane producers and the red zone contains Archaea that grow at extremely high temperatures. Some Archaea use transformation whereas others use conjugation. Rare cases of viral transduction also occur. The modes of gene transfer seen within each family do not correlate well with either lifestyle or evolutionary relationships. The Crenarchaeota and the Euryarchaeota are the two major branches of the Archaea. Molecular Biology, 3 rd edition by Clark, Pazdernik and Mc. Gehee Copyright © 2019 by Academic Cell. All rights reserved.
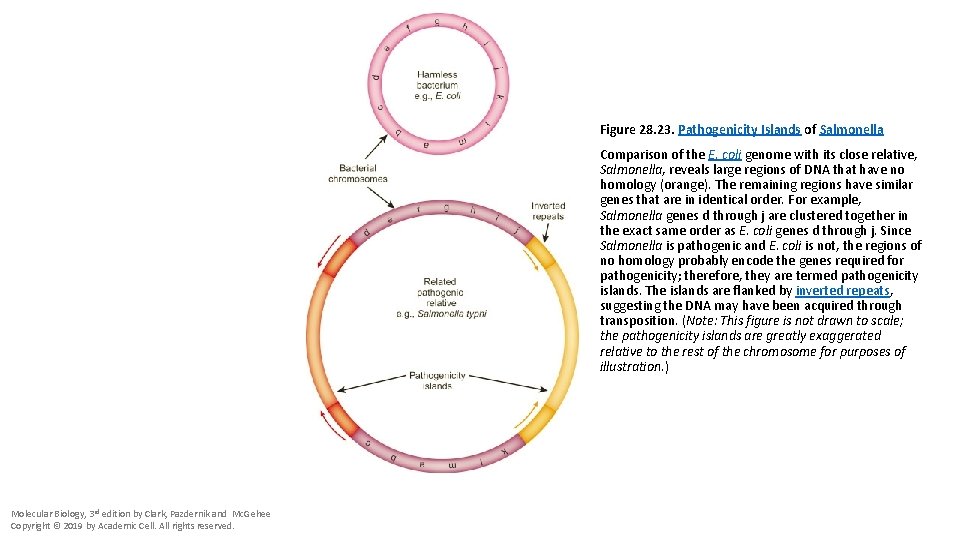
Figure 28. 23. Pathogenicity Islands of Salmonella Comparison of the E. coli genome with its close relative, Salmonella, reveals large regions of DNA that have no homology (orange). The remaining regions have similar genes that are in identical order. For example, Salmonella genes d through j are clustered together in the exact same order as E. coli genes d through j. Since Salmonella is pathogenic and E. coli is not, the regions of no homology probably encode the genes required for pathogenicity; therefore, they are termed pathogenicity islands. The islands are flanked by inverted repeats, suggesting the DNA may have been acquired through transposition. (Note: This figure is not drawn to scale; the pathogenicity islands are greatly exaggerated relative to the rest of the chromosome for purposes of illustration. ) Molecular Biology, 3 rd edition by Clark, Pazdernik and Mc. Gehee Copyright © 2019 by Academic Cell. All rights reserved.
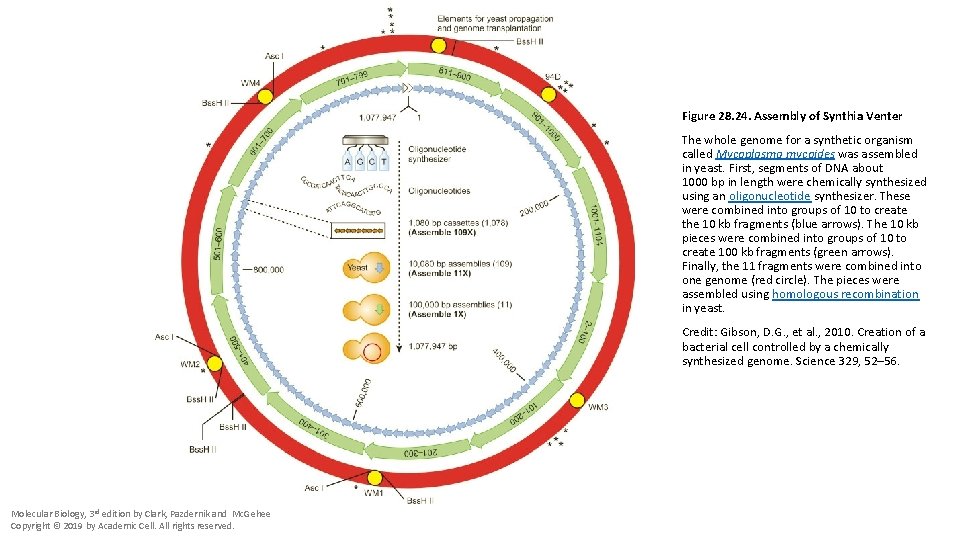
Figure 28. 24. Assembly of Synthia Venter The whole genome for a synthetic organism called Mycoplasma mycoides was assembled in yeast. First, segments of DNA about 1000 bp in length were chemically synthesized using an oligonucleotide synthesizer. These were combined into groups of 10 to create the 10 kb fragments (blue arrows). The 10 kb pieces were combined into groups of 10 to create 100 kb fragments (green arrows). Finally, the 11 fragments were combined into one genome (red circle). The pieces were assembled using homologous recombination in yeast. Credit: Gibson, D. G. , et al. , 2010. Creation of a bacterial cell controlled by a chemically synthesized genome. Science 329, 52– 56. Molecular Biology, 3 rd edition by Clark, Pazdernik and Mc. Gehee Copyright © 2019 by Academic Cell. All rights reserved.
- Slides: 25