Unit 5 Thermochemistry Entropy and Gibbs Free Energy

Unit 5: Thermochemistry Entropy and Gibbs Free Energy

First Law of Thermodynamics l Energy can be converted from one form to another, but cannot be created or destroyed l Energy of the universe is constant Reference: Chemistry 7 th ed. , Chang, Chemistry 7 th ed. , Zumdahl

Second Law of Thermodynamics l The entropy of the universe increases in a spontaneous process and remains unchanged in an equilibrium process l the entropy of the universe is increasing Reference: Chemistry 7 th ed. , Chang, Chemistry 7 th ed. , Zumdahl

Third Law of Thermodynamics l The entropy of a perfect crystalline substance is zero at absolute zero
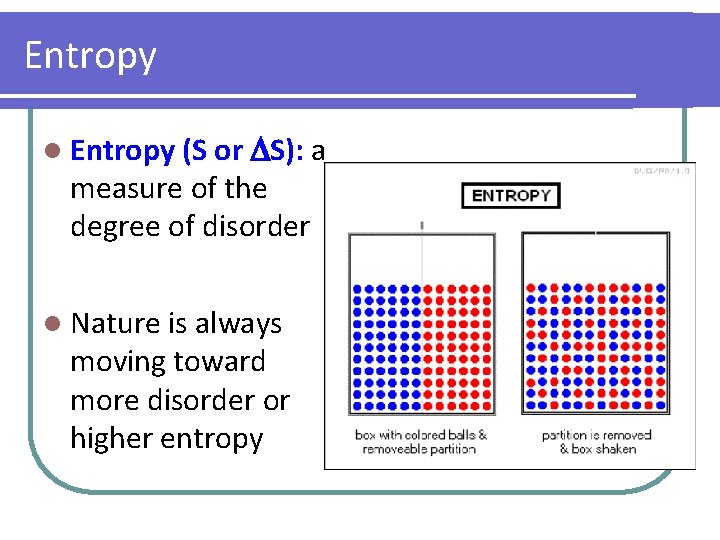
Entropy l Entropy (S or S): a measure of the degree of disorder l Nature is always moving toward more disorder or higher entropy

Molecular Entropy High S Low S Gas Liquid/Aq. Solid Example: Will ΔS increase or decrease for the following reaction? CH 4(g) + 2 O 2(g) → CO 2(g) + 2 H 2 O(l)

Calculating Entropy l Just like we did with H… S = ΣSproducts – ΣSreactants l An increase in entropy promotes spontaneous reactions l Units:
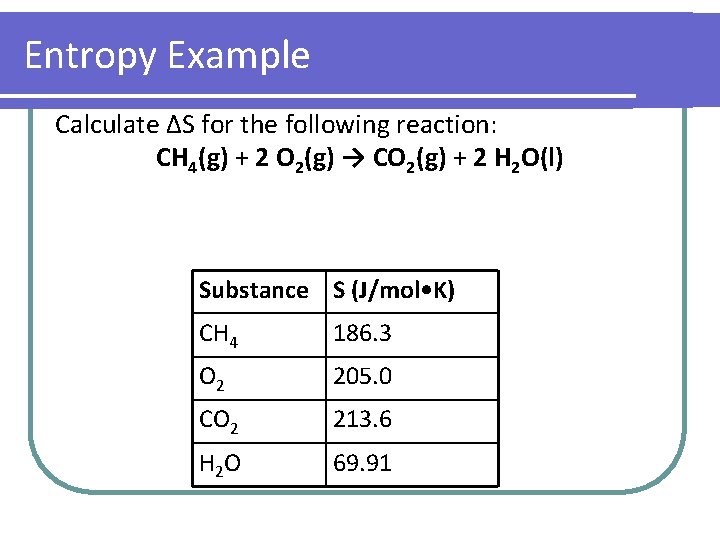
Entropy Example Calculate ΔS for the following reaction: CH 4(g) + 2 O 2(g) → CO 2(g) + 2 H 2 O(l) Substance S (J/mol • K) CH 4 186. 3 O 2 205. 0 CO 2 213. 6 H 2 O 69. 91
Gibbs Free Energy l Free energy: capacity of a system to do work l Useful in predicting if a reaction is spontaneous

What is spontaneity? l spontaneous reaction: a chemical reaction that occurs without any outside energy l Example: l At room temperature: Spontaneous: ice → liquid water l Not spontaneous: liquid water → ice l

Why do we care about spontaneity? l It is almost impossible to know details about every chemical reaction and determine if a reaction will occur on its own l Examples: l l Does Na. OH decompose spontaneously? Can you store it in the chemical store room with other chemicals or does it need a special environment? Why is hydrogen peroxide (H 2 O 2) stored in a brown opaque bottle? Could it be that when exposed to light H 2 O 2 spontaneously decomposes? Spontaneity can be predicted using the Gibbs Free Energy equation

Gibbs’ Free Energy Equation G = H – T S G = Gibbs Free Energy H = heat or enthalpy T = temperature (in KELVIN: K = C + 273. 15) S = entropy G > 0 reaction is NOT spontaneous G < 0 reaction is spontaneous l Note: Units for H are usually k. J and units for S are usually J – be sure to convert!

Gibbs Free Energy Example Calculate the free energy change for the formation of NO(g) from N 2(g) and O 2(g) at 298 K given H=180. 7 k. J and S=24. 7 J/K. Is the reaction spontaneous under these circumstances? N 2 (g) + O 2 (g) 2 NO (g)

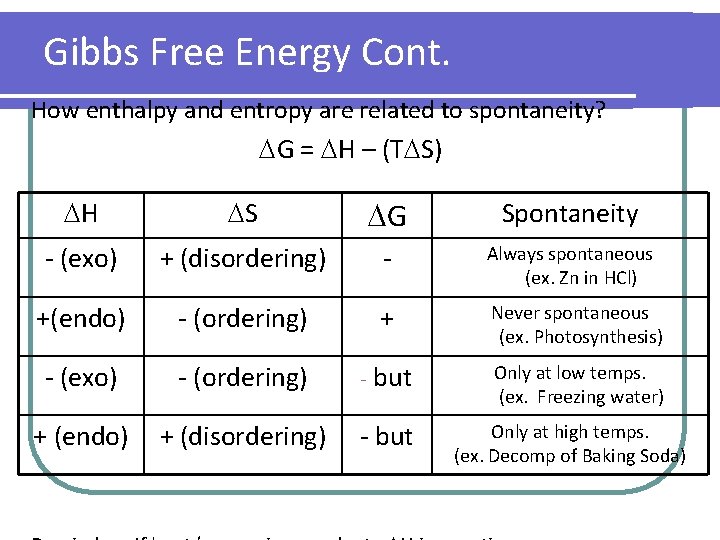
Gibbs Free Energy Cont. How enthalpy and entropy are related to spontaneity? G = H – (T S) H S G Spontaneity - (exo) + (disordering) - Always spontaneous (ex. Zn in HCl) +(endo) - (ordering) + Never spontaneous (ex. Photosynthesis) - (exo) - (ordering) - but Only at low temps. (ex. Freezing water) + (endo) + (disordering) - but Only at high temps. (ex. Decomp of Baking Soda)
- Slides: 15