Unit 5 Ionic Bonding Day 4 Finish Bonding

Unit 5: Ionic Bonding Day 4: Finish Bonding Basics, Toolbox, Ionic Go Fish
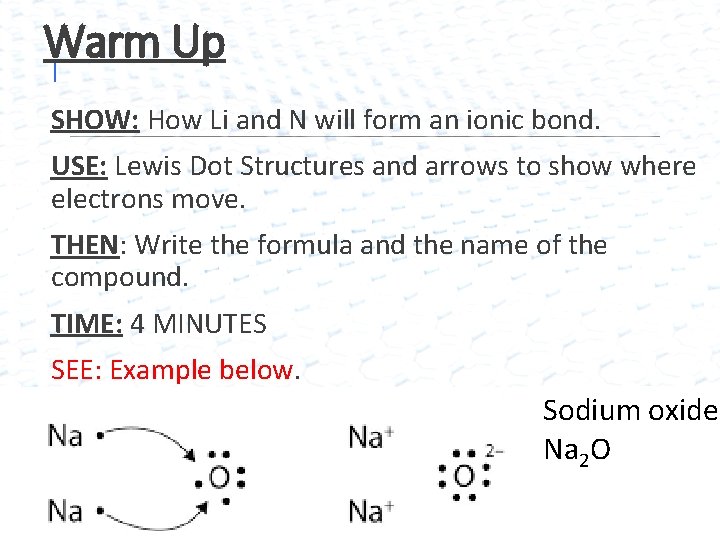
Warm Up I SHOW: How Li and N will form an ionic bond. USE: Lewis Dot Structures and arrows to show where electrons move. THEN: Write the formula and the name of the compound. TIME: 4 MINUTES SEE: Example below. Sodium oxide Na 2 O

Agenda ◦ Learning Check: Teams ◦ Finish Ionic Bonding Practice ◦ Toolbox: Polyatomic Ions ◦ Speed Match ◦ Ionic Go Fish ◦ Progress Reports ◦ IF YOU HAVE NOT COMPLETED THE FLAME TEST LAB (You have until Thu after school) ◦ Come to Office Hours ◦ Mornings Upon Request ◦ Wed (4/5) – 3: 30 -4: 15 ◦ Thu (4/6) – 3: 30 -4: 45

Learning Check: How Ionic Bonds Form DO: On the white board, as a team § Show any alkaline earth metal will bond with any halogen. §Write the name and formula THEN: §Show Aluminum will bond with Sulfur. Also, write the name and formula TIME: 7 minutes WHEN DONE: Take out Ionic Bonding Practice

Learning Check: How Ionic Bonds Form Calcium Chloride Ca. Cl 2 Aluminum Sulfide Al 2 S 3

Ionic Bonding Practice FINISH: Ionic Bonding Practice WORK: Together in Table Groups USE: Manipulatives (if needed) TIME: 6 minutes WHEN DONE: Open to Unit 5 Toolbox

Open to Toolbox Page

UNIT 5 Tool. Box Polyatomic Ions A many-atom ion. A group of non-metal atoms together have a charge. Example 1 Sulfate – In shampoos, soaps, and other cleaning agents -
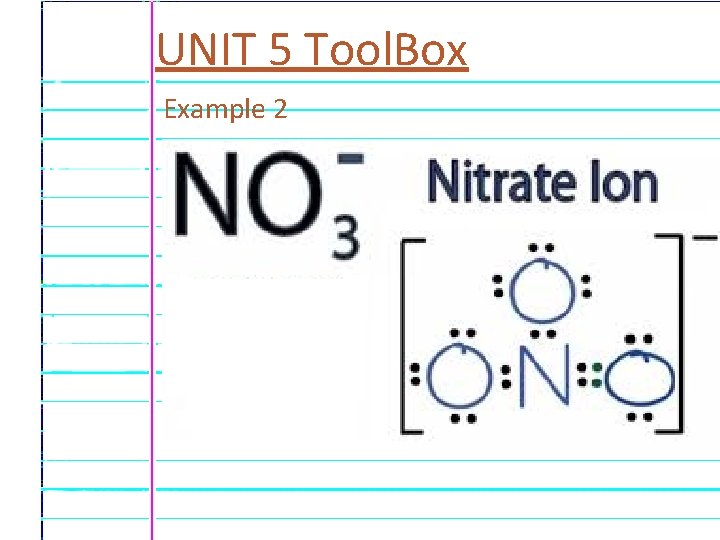
UNIT 5 Tool. Box Example 2

UNIT 5 Tool. Box Polyatomic Ions Example 3

UNIT 5 Tool. Box Rules for Formulas and Names of Polyatomic Ions Calcium Sulphate Ca. SO 4 Rule 1: Don’t write charge in formula Potassium Nitrate: KNO 3 Rule 2: Don’t write “ 1” in subscript of formula Ammonium Oxide (NH 4)2 O Rule 3: Use parentheses when there is more than one polyatomic ion
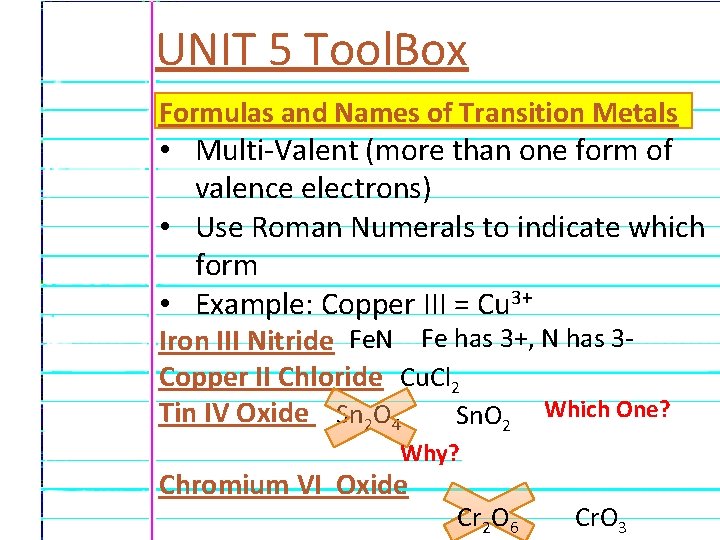
UNIT 5 Tool. Box Formulas and Names of Transition Metals • Multi-Valent (more than one form of valence electrons) • Use Roman Numerals to indicate which form • Example: Copper III = Cu 3+ Iron III Nitride Fe. N Fe has 3+, N has 3 Copper II Chloride Cu. Cl 2 Tin IV Oxide Sn 2 O 4 Sn. O 2 Which One? Why? Least Common Multiple Chromium VI Oxide Cr 2 O 6 Cr. O 3

UNIT 5 Tool. Box WRITE SOMEWHERE AT THE TOP OF YOUR PAGE: AND HIGHLIGHT IT! Vocabulary: Cation = Positively Charged Ion Anion = Negatively Charged Ion ONE MORE THING!

Match n’ Make Compounds Speed Round DO: Match ALL CARDS without any left over. The point of this part of the lesson is for you to become confident in how to match up cations (+) and anions (-). PUT: Matches on desk TIME: 9 minutes This is not the WHEN DONE: Show me game yet!

Go Fish! (for Ions!) Basic Directions (more on handout) Deal 7 cards each First player is to the right of dealer FIRST TURN: Everyone Lays down ALL MATCHES. KEEP: 7 cards in your hand at all times ASK: One person for a card you want. That person must you ALL of that card. You may play any matches you get. ◦ If that person doesn’t have it, that person says “go fish!” You draw a card and your turn is over. Tally up your points, fill out your score sheet as you go. GAME OVER: When one person plays all their cards OR when deck is gone. HOWEVER, if you can play all 7 cards in your hand, the game is NOT over, and you get 10 points, and have to draw 7 more cards.

Exit Ticket ON A BLANK PIECE OF PAPER: Write your name and answer the following questions - 1. How do ionic bonds form? 2. Do you want more practice with ionic bonds formation? 3. On a scale of 1 -10 (10 being highest), how comfortable are you with the concept of ionic bonding? TIME: 9 minutes WHEN DONE: Turn it into class box!

Exit Ticket: - Write the formula for the names given. Write the names of the formulas given. Copper II phosphate Iron II sulfate Iron III oxide Lithium nitrate Aluminum sulfide K 2 O Mg 3(PO 4)2 Al 2(CO 3)3
- Slides: 17