Unit 5 Fresh and Salt Water Systems Topic




















- Slides: 21

Unit #5 Fresh and Salt Water Systems

Topic #1 A World of Water

Topic #1 – A World of Water n n n Water is the key to life – scientists believe that life cannot exist without it! More species of organisms inhabit the water than any other place on Earth Whether you realize it or not, you depend on water every moment of your life

n n n Salt water – water such as ocean water and seawater that has a high salt content 97% of the earth’s water is salt water – not drinkable (potable) Fresh water – water that most organisms can drink or use for life functions such as lake water, river water, pond water or well water.

How do you depend on water? n n n n About 65% of your body mass is water In comparison, an apple is about 84% water and a watermelon is about 98% water Every breath you exhale has water vapour in it Every cellular process uses a tiny fraction of your body’s water To maintain a normal body temperature of 37 degrees Celsius you need to sweat and release water You lose water every time you eliminate wastes as well You need to consume about 2. 5 L of water each day

How else do we depend on water? n

How else do we depend on water? n Swimming and other recreational activities

How else do we depend on water? n n Swimming and other recreation Watering lawns/flowers/etc

How else do we depend on water? n n n Swimming and other recreation Watering lawns/flowers/etc Putting out fires

How else do we depend on water? n n Swimming and other recreation Watering lawns/flowers/etc Putting out fires Cooling vehicles/running machinery

How else do we depend on water? n n n Swimming and other recreation Watering lawns/flowers/etc Putting out fires Cooling vehicles/running machinery Hydro-electric power

The Water Cycle n n n Ever wondered when it rains, “Where does all that water go? ” Some runs off the land into streams and rivers and then flows into oceans Some soaks, seeps and flows underground Some goes into wetlands and lakes Some just appears to vanish into the atmosphere

n n The ability of water to appear and disappear is the result of 2 previously learned changes of state: evaporation and condensation The water cycle controls the distribution of Earth’s water as it evaporates from bodies of water, condenses, precipitates and returns to those bodies of water The Sun’s energy drives the water cycle About 520, 000 km 2 of water is evaporated each year on Earth

n n n Water vapour does not remain in the atmosphere for very long On average there is only enough water vapour in the atmosphere for a 10 day supply of precipitation worldwide About 78% of this precipitation falls into the oceans

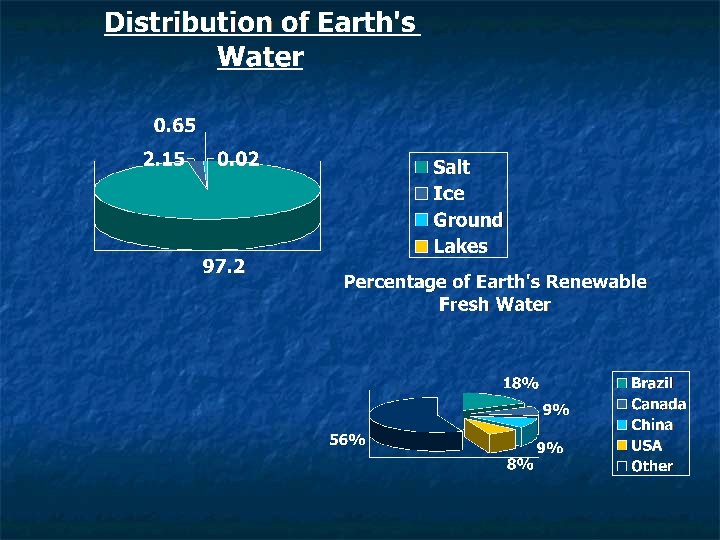
Distribution of Water on Earth n Take the WATER QUIZ 1) How much of the Earth’s water is salt water? a) 50% b) 75% c) 93% d) 97%

2) How much of Earth’s remaining 3% fresh water is frozen in glaciers and ice sheets? a) 0. 50% b) 1% c) 2. 15% d) 2. 85% 3) How much does this leave humans for liquid fresh water? a) 2% b) 1% c) 0. 5% d) 0. 1%

Distribution of Water on Earth n n Only 2. 8% of Earth’s water is fresh water Of this 2. 8%, 2. 15% is frozen in glaciers and ice sheets Of the remaining 0. 65%, 0. 63% is groundwater This leaves on 0. 02% of Earth’s water in rivers, ponds, lakes, etc!!!!!!



Concerns? ? ? n n n Water quantity – the amount of water Water quality – the characteristics of a water resource that make it suitable or unsuitable for various uses Canada’s rivers and lakes we need to manage properly

Your task…. n Complete investigation 5 -A on page 365 a) identify and record all the ways your family uses water b) Talk to your parents and locate the water metre in your house if possible c) Over the course of one week, estimate how much water your family uses every day d) Record this data in a table as shown in the Investigation e) Complete all parts to this activity (questions, tables, etc) f) graph the results in some form (ie. pie graph)