Unit 5 Chemical Reactions Topics Balancing Chemical Reactions

Unit 5 Chemical Reactions Topics ¨ Balancing Chemical Reactions ¨ Types of Chemical Reactions ¨ Enthalpy and Reactions Profiles ¨ Stoichiometry ¨ Limiting Reagents/Excessive Reagents

Chemical Reactions: A process that results in an inter-conversion of chemical species. Reactants Products ¨ Chemical Reactions Obey the following Rules – The number and type of atoms on both sides of the reactions must be the same – The total charge on both sides of the reaction must be the same – Energy is conserved.

Types of Chemical Rx. ’s ¨ Synthesis (or Combination) 2 Elements react to form a single Compound A+B C (General Form) Examples C(S) +O 2(g) CO 2(g) H 2(g) + Cl 2(g) 2 HCl(g) Predicting Products: Assume product is a compound where both elements have a complete octet (least electron negative atom goes first) H 2(g) + F 2(g)

Types of Chemical Rx. ’s ¨ Decomposition: Breaking down a molecule into simpler substances AB A +B Examples 2 Hg. O(s) + Heat 2 Hg(l) + O 2(g) 2 NO(g) + Heat N 2(g) + O 2(g) Predicting Products: assume products are the elements in the original compound 2 H 2 O(l)

Types of Chemical Rx. ’s ¨ Single Replacement: involves replacing one atom in a compound by another atom. A +BX AX +B (General form) Examples Cl 2(aq) +2 KI(aq) 2 KCl(aq) + I 2(aq) Cu. Cl 2(aq) + Fe(s) Fe. Cl 2(aq) +CU(s) Predicting Products: if the reacting element is a metal it trades places with the metal in the compound. If the reacting element is a non-metal it trades places with the non-metal CUI 2 + Br 2

Types of Chemical Rx. ’s ¨ Double Replacement: involves an exchange of atoms or groups between 2 different compounds AB + XY AY +XB (General Form) Examples Na. Cl(aq) + H 2 SO 4(aq) 2 HCl +Na 2 SO 4 Predicting Products: Assume reactants are made of ions. Make products by having positive ions swap negative ions H 3 PO 4 +Ba(OH)2

Types of Chemical Rx. ’s ¨ Combustion: a compound containing Carbon, Hydrogen and possibly Oxygen react with Oxygen to give Carbon Dioxide and Water Cx. Hy. Oz +O 2 CO 2 and H 2 O (General form) Example: C 5 H 12 +8 O 2 5 CO 2 +6 H 2 O

Enthalpy & Reaction Profiles ¨ Enthalpy, H is the Heat contained in a system ¨ DH = Change in enthalpy during a reaction ¨ DH = Hproducts –Hreactants – Hproducts = heat energy contained in products – Hreactatants = heat energy contained in Rg’s. Recall: energy in chemical reactions must be conserved.

Endothermic Reactions ¨ Reactants take heat from Surroundings ¨ Products have more Heat energy than Reactants ¨ DH for Reaction is positive ¨ Heat was added to the system Reactants + Heat Products
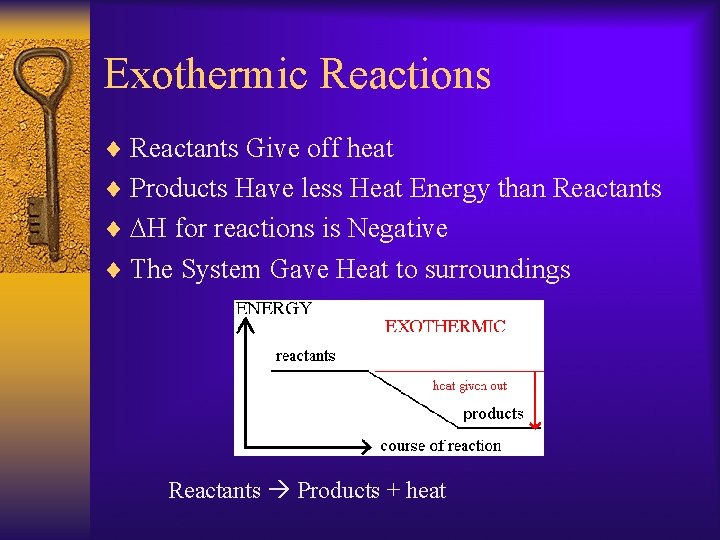
Exothermic Reactions ¨ Reactants Give off heat ¨ Products Have less Heat Energy than Reactants ¨ DH for reactions is Negative ¨ The System Gave Heat to surroundings Reactants Products + heat

Intro to Stoichiometry ¨ Stoichiometry is the calculation of quantities of reactants and products in chemical reactions ¨ Before doing Stoichiometry, you need to know what the balanced reaction is ¨ The Coefficients in the balanced reactions tell you the ratios between the various reactants and products ¨ These ratios allow you to calculate the amounts of all products and reactants if you know the amount of any product or reactant.

N 2 + 3 H 2 2 NH 3 ¨ The balanced Rx shows the simplest whole number ratio between all reactants and products ¨ If we started with 60 molecules of H 2 how many molecules of N 2 would we need? ¨ How many molecules of NH 3 would we produce? ¨ Formula for Calculating Quantities of Reactants or Products: (Stoichiometric formula) Required Qty = Given Qty X Ratio in Balance eqs. Example #N 2 =#H 2 X 1 N 2/ 3 H 2 (ratio from balanced rx) #N 2 =60 X 1/3 #N 2 = 20

General Procedure for Solving Stoichiometric Problems ¨ Step 1: Convert the Given Quantity into Moles. ¨ Step 2: Use the Stiochiometric Formula to find the required quantity. ¨ Step 3: Convert your answer into the required units. ¨ Note: Often Chemists will do all three steps in the same calculation

Limiting Reagents, Theoretical yields & Excessive Quantities ¨ Reactions are often carried out such that one or more of the reactants are present in excessive amounts. ¨ The reactant that is exhausted (total consumed) is called the Limiting reagent. ¨ Theoretical Yield of a product, is the maximum amount of the identified product that can be produced based on the limiting reagent ¨ The Excess quantity is the amount of the excessive reagent left over, after as much of it has reacted as possible as determined by the limiting reagent.

Example C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O If you start with 3 moles of C 3 H 8 and 10 moles of O 2 What is the limiting reagent? What is the Theoretical yield of CO 2 How much of the Excessive reagent is leftover ¨ Example if 20 g of H 2 react with 100 g O 2 to make H 2 O which reactant is present in excessive amounts and by how many grams? Which is the limiting reagent?

Procedure for determining the Limiting Reagent ¨ Step 1: convert all initial quantities into moles ¨ Step 2: Arbitrarily select a product in the Rx. ¨ Step 3: Calculate the amount of product that could be produced for the given quantity(# of moles) of reagent ¨ Step 4: Repeat Step 3 for each given quantity of reagent ¨ Step 5: Determine which reagent produced the least amount of product. This is your Limiting Reagent

Procedure for determining the Amount of Excessive Reagent ¨ Step 1: Use the amount (# of moles) of Limiting Reagent and the Mole ratio from the balanced Rx to determine the actual required amount of the other Reagents. ¨ Step 2: The Excessive Amount = the given amount minus the actual required amount ¨ Step 3: Convert Excessive amount into the units required by the problem
- Slides: 17