Unit 5 Bonding and Nomenclature 5 1 Nomenclature

Unit 5: Bonding and Nomenclature 5. 1 Nomenclature • • Rules for nomenclature (naming) compounds Formula Writing A. Ionic B. Covalent C. Diatomic 5. 2 Bonding • Ionic and Covalent molecules D. Acids

Unit 5: Terminology List Nomenclature Acids Metals Bases Non-Metals Ionic Combining Capacity (C. C. ) Charges Covalent Bond Polyatomic Ions Roman numerals Diatomics prefixes

Part D. Acids Involves “H” hydrogen at the front of a chemical formula Recall our “Family of One” notes from Unit 3: Periodic Table and Trends Hydrogen acts likes a metal Becomes a H+ ion in solutions

I. Binary Acids Made up of two types of atoms HX H = metal X = non-metal For example: HCl becomes 1. hydrogen = hydro 2. Write “-ic” instead of “-ide” 3. acid goes on the end hydrochloric acid

LEARNING CHECK - Binary Acids 1. HI = 2. HBr = 3. hydrofluoric acid = 4. hydrosulphuric acid =

II. Ternary Acids Made up of MORE THAN two types of atoms H = metal HX#Y# X = non-metal Y = non-metal For example: HNO 2 becomes nitrous acid 1. drop hydrogen from the name 2. Write “-ic” instead of “-ate” 3. Write “-ous” instead of “-ite” 4. acid goes on the end I ate it, it was icy

LEARNING CHECK - Ternary Acids 1. HNO 2 = 2. HNO 3 = 3. sulphuric acid= 4. sulphurous acid =

II. Ternary Acids – Special Names There a lot of special names as well You don´t need to memorize them all! Examples: CH 3 COOH = H 2 CO 3 = H 2 C 2 O 4 =


Lecture. PLUS Timberlake Learning Check Match each set with the correct name: A. B. Na 2 CO 3 1) magnesium sulfite Mg. SO 3 2) magnesium sulfate Mg. SO 4 3) sodium carbonate Ca(HCO 3)2 Ca. CO 3 Ca 3(PO 4)2 1) calcium carbonate 2) calcium phosphate 3) calcium bicarbonate 10

Lecture. PLUS Timberlake Solution A. B. Na 2 CO 3 3) sodium carbonate Mg. SO 3 1) magnesium sulfite Mg. SO 4 2) magnesium sulfate Ca(HCO 3)2 3) calcium bicarbonate Ca. CO 3 1) calcium carbonate Ca 3(PO 4)2 phosphate 2) calcium 11

Lecture. PLUS Timberlake Learning Check A. aluminum nitrate 1) Al. NO 3 2) Al(NO)3 3) Al(NO 3)3 B. copper(II) nitrate 1) Cu. NO 3 2) Cu(NO 3)2 3) Cu 2(NO 3) C. iron (III) hydroxide 1) Fe. OH 2) Fe 3 OH 3) Fe(OH)3 D. tin(IV) hydroxide 1) Sn(OH)4 2) Sn(OH)2 3) Sn 4(OH) 12

Lecture. PLUS Timberlake Solution A. aluminum nitrate 3) Al(NO 3)3 B. copper(II) nitrate 2) Cu(NO 3)2 C. Iron (III) hydroxide 3) Fe(OH)3 D. Tin(IV) hydroxide 1) Sn(OH)4 13
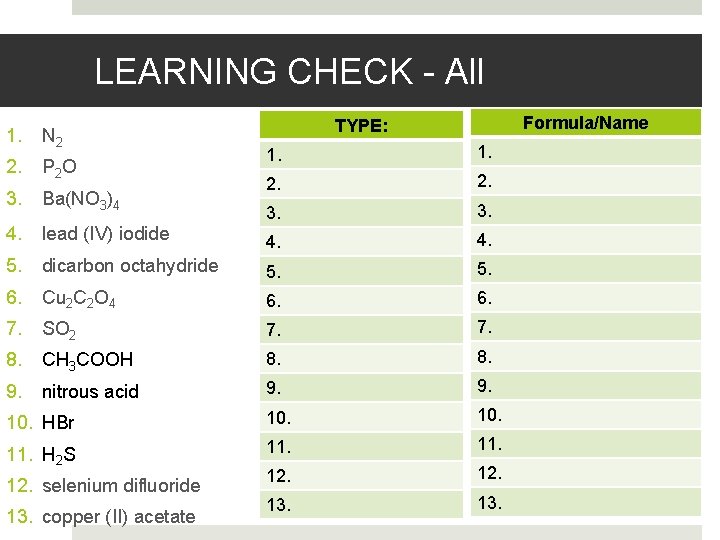
LEARNING CHECK - All 1. N 2 2. P 2 O 3. Ba(NO 3)4 4. lead (IV) iodide 5. Formula/Name TYPE: 1. 2. 3. 4. dicarbon octahydride 5. 6. Cu 2 C 2 O 4 6. 7. SO 2 7. 8. CH 3 COOH 8. 9. nitrous acid 9. 10. HBr 10. 11. H 2 S 11. 12. 13. 12. selenium difluoride 13. copper (II) acetate
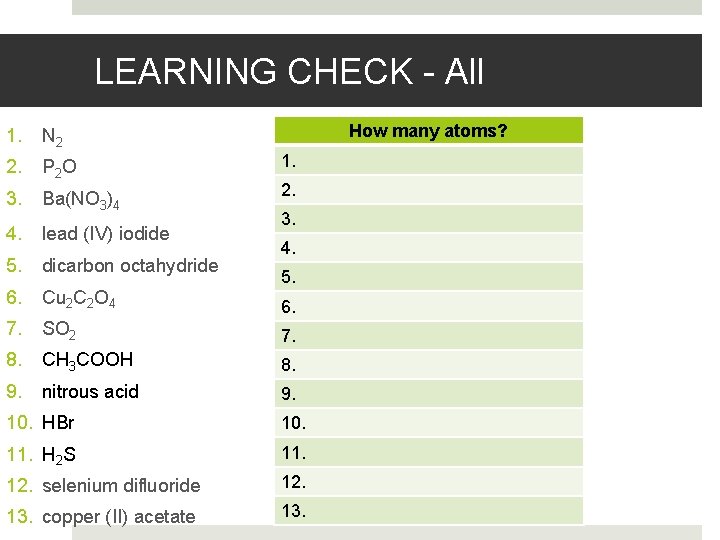
LEARNING CHECK - All How many atoms? 1. N 2 2. P 2 O 3. Ba(NO 3)4 4. lead (IV) iodide 5. dicarbon octahydride 6. Cu 2 C 2 O 4 7. SO 2 7. 8. CH 3 COOH 8. 9. nitrous acid 9. 1. 2. 3. 4. 5. 6. 10. HBr 10. 11. H 2 S 11. 12. selenium difluoride 12. 13. copper (II) acetate 13.

SPACE RACE STYLE QUIZ – ALL 20+ Questions (all sections) Take your time, it counts for your mark! For unreliable internet, copy your answers (just the letter) on paper
- Slides: 16