UNIT 5 2 FORMS AND FUNCTION OF COVALENT









- Slides: 10

UNIT 5. 2: FORMS AND FUNCTION OF COVALENT MOLECULES DAY 7: WHERE’S THE FUN, FUNCTIONAL GROUPS, BUILDING MOLECULES

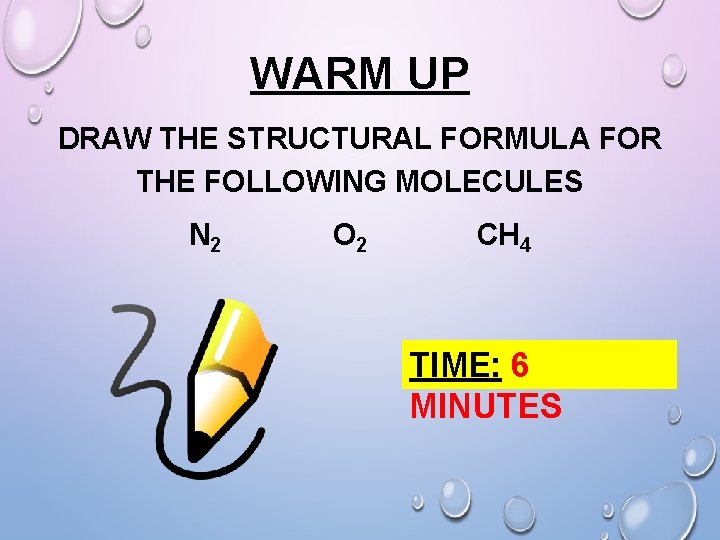
WARM UP DRAW THE STRUCTURAL FORMULA FOR THE FOLLOWING MOLECULES N 2 O 2 CH 4 TIME: 6 MINUTES

AGENDA • DEMO: LIQUID NITROGEN • WHERE’S THE FUN? • IDENTIFY THE FUNCTIONAL GROUP • VSEPR THEORY

LIQUID NITROGEN ØACCORDING TO HONC 1234 RULE, HOW MANY BONDS WILL N 2 MAKE? ØHOW MANY BONDING PAIRS? ØHOW MANY LONE PAIRS? ØBOILS AT -320 F (196 C)

READING: FUNCTIONAL GROUPS DO: ANNOTATE WITH MARGIN NOTES AND HIGHLIGHTING ONE IMPORTANT THING IN EACH PARAGRAPH. THEN: ANSWER ANALYSIS QUESTIONS (ON BACK PAGE) PUT: ANSWERS ON BACK OR ATTACH A SEPARATE SHEET OF PAPER COMPLETE: #1 -5 = 2. 5 #1 -5 & #7 -9 = 3. 0 #1 -9 = 4. 0 HINT: If you are struggling to answer questions, draw out the structures TIME: 36 MINUTES WHEN DONE: BEGIN IDENTIFYING FUNCTIONAL GROUPS

IDENTIFYING FUNCTIONAL GROUPS WITH TABLE PARTNERS: COMPLETE “WHERE’S THE FUN? IDENTIFYING FUNCTIONAL GROUPS” USE: • READING AND QUESTIONS • HIGHLIGHTER TO IDENTIFY GROUPS TIME: 12 MINUTES WHEN DONE: STAPLE TO READING AND QUESTIONS AND TURN INTO

REVIEW – LOOK IN YOUR NOTES! WHAT IS INCORRECT ABOUT EACH OF THE LEWIS H F STRUCTURES BELOW? ? N H A • Nitrogen needs to make 3 bonds • Molecule needs another “H” • Nitrogen will only have 1 F C F B • No lone pairs on Fluorine • Carbon needs to make 4 bonds • Carbon will not have a lone pair O C • Carbon needs to make 4 bonds • Carbon will not have a lone pair • Each Oxygen need 2 bonds

DISCUSSION AND NOTES SET UP: C-NOTE PAGE • TITLE: VSEPR THEORY • ESSENTIAL QUESTION: HOW DO VALENCE ELECTRONS INFLUENCE THE SHAPE OF A MOLECULE? PUT: ON THE NEXT FRESH RIGHT PAGE TIME: 2 MINUTES

VSEPR Theory 3/10 ENDOCRINE DISRUPTORS EQ: How do valence electrons influence the shape of a molecule? EQ: HOW DO ENDOCRINE DISRUPTORS IMPACT Valence Shell Electron Pair Repulsion HUMANS? Theory - Bonding pairs of electrons and lone pairs of electrons on a molecule will repel each other in order to be as far apart from each other as possible in 3 dimensions. - This determines the geometry of molecules and therefore their properties!

BUILDING MOLECULES DO: USE VSEPR THEORY AND THE STRUCTURAL FORMULAS TO HELP YOU DESIGN EACH MOLECULE CORRECTLY. PUT: DIAGRAMS AND DRAWINGS ON ASSIGNMENT SHEET TIME: 42 MINUTES WHEN DONE: WORK ON