Unit 4 Types of Chemical Reactions and Solution





































































- Slides: 70

Unit 4: Types of Chemical Reactions and Solution Stoichiometry Solutions, Predicting Products, and Balancing Oxidation-Reduction Reactions

Unit 4 Topics 4. 1 4. 2 4. 3 4. 4 4. 5 4. 6 4. 7 4. 8 4. 9 4. 10 Water, the Common Solvent The Nature of Aqueous Solutions: Strong and Weak Electrolytes The Composition of Solutions Types of Chemical Reactions Precipitation Reactions Describing Reactions in Solution Stoichiometry of Precipitation Reactions Acid–Base Reactions Oxidation–Reduction Reactions Balancing Oxidation–Reduction Equations

Section 4. 1: Water the Common Solvent Water • One of the most important substances on Earth. • Can dissolve many different substances. • A polar molecule because of its unequal charge distribution. • H 2 O is “bent” or V-shaped (104. 5°C between hydrogens) • H 2 O is a polar molecule because the oxygen atom has a greater attraction for electrons.

Dissolution of a Solid in a Liquid Animation Link: Dissolving Ionic and Molecular Compounds

• Hydration: as ionic solids dissolve in water (break up into individual anions and cations), the + ends of the water attract the anions and the – ends of the water attract the cations.

• The solubility of substances in water depends on two things: 1. The attraction of the ions to each other within the solid. 2. The attraction of the ions to water molecules.

• Though “like dissolves like, ” water dissolves many nonionic substances, such as ethanol (C 2 H 5 OH), which have polar bonds. Thus ionic and polar substances dissolve well in water, which nonpolar substances do not.

Section 4. 2: The Nature of Aqueous Solutions • • A solution is a homogeneous mixture made up of a solute and solvent. Solute – substance being dissolved. Solvent – the substance doing the dissolving. Aqueous solution – solutions in which water is the solvent.

Electrolytes Electrical conductivity is the ability of a solution to conduct electricity. • Strong Electrolytes – conduct current very efficiently (bulb shines brightly). • Weak Electrolytes – conduct only a small current (bulb glows dimly). • Nonelectrolytes – no current flows (bulb remains unlit).

Strong Electrolytes These are completely ionized (completely dissociate into separate ions) when dissolved in water. Three classes: 1. Soluble salts Ex: Na. Cl Na+ + Cl 2. Strong acids – Produce H+ when dissolved in water Ex: HCl H+ + Cl 3. Strong bases – contain OHEx: Na. OH Na+ + OH-

Weak Electrolytes These dissociate only a little, while the majority of the substance does not. Three classes: 1. Slightly soluble salts Ex: Ba. F 2 2. Weak acids Ex: HC 2 H 3 O 2, HF, HCN, HNO 2 3. Weak bases Ex: NH 3

Nonelectrolytes These dissolve in water but do not produce ions. • Ex: C 2 H 5 OH – this substance is dispersed in water but does not break into smaller components. • All molecular substances will act this way.

Electrolyte Behavior Animation Link: Strong vs. Weak Electrolytes

Section 4. 3: Composition of Solutions Concentration is measured in Molarity • Molarity (M) = moles of solute per volume of solution in liters: moles of solute Molarity = liters of solution

Example A 500. 0 -g sample of potassium phosphate is dissolved in enough water to make 1. 50 L of solution. What is the molarity of the solution? 500. 0 g K 3(PO 4) x M= 1 mol K 3(PO 4) 2. 355 mol K (PO ) = 3 4 212. 27 g. K 3(PO 4) 2. 355 mol K 3(PO 4) mol = = 1. 57 M 1. 50 L L

Realize that the solution concentration is given in terms of the form of the solute before it dissolves. It therefore may not accurately reflect the concentration of ions within the solution.

Concentration of Ions For a 0. 25 M Ca. Cl 2 solution: Ca. Cl 2 → Ca 2+ + 2 Cl– Ca 2+: 1 × 0. 25 M = 0. 25 M Ca 2+ Cl–: 2 × 0. 25 M = 0. 50 M Cl–

Example Calculate the concentration of Cl- ions if 0. 70 mol of Zn. Cl 2 are dissolved in 1. 75 L of solution. When Zn. Cl 2 dissolves: Zn. Cl 2 Zn+2 + 2 Cl 0. 70 moles x 2 Cl- = 0. 80 M Cl 1. 75 L soln

Concept Check Which of the following solutions contains the greatest number of ions? a) b) c) d) 400. 0 300. 0 200. 0 800. 0 m. L of of 0. 10 M M Na. Cl. Ca. Cl 2. Fe. Cl 3. sucrose.

Notice The solution with the greatest number of ions is not necessarily the one in which: • the volume of the solution is the largest. • the formula unit has the greatest number of ions.

Making Solutions: Terminology Standard solution: Solution whose concentration is accurately known. Stock solution: Solutions in concentrated forms (used for dilutions of different concentrations). Dilution: The process of adding water to stock solutions to attain desired concentration.

Dilution • Dilution with water does not alter the numbers of moles of solute present. • Moles of solute before dilution = moles of solute after dilution M 1 V 1 = M 2 V 2

Example What volume of 16 M sulfuric acid must be used to prepare 1. 5 L of a 0. 10 M H 2 SO 4 solution? M 1 V 1=M 2 V 2 (16 M)(V 1) = (0. 10 M)(1. 5 L) V 1=0. 0094 L x 1000 m. L = 9. 4 m. L 1 L

Concept Check A 0. 50 M solution of sodium chloride in an open beaker sits on a lab bench. Which of the following would decrease the concentration of the salt solution? a) Add water to the solution. b) Pour some of the solution down the sink drain. c) Add more sodium chloride to the solution. d) Let the solution sit out in the open air for a couple of days. e) At least two of the above would decrease the concentration of the salt solution.

Section 4. 4: Types of Chemical Reactions • • Synthesis (review) Decomposition (review) Not in your Single replacement (review) textbook Double replacement (review) Combustion (review) Precipitation Reactions Acid–Base Reactions Oxidation–Reduction Reactions

Synthesis Elements combine with other elements to form a compound. sodium + chlorine sodium chloride magnesium + phosphorus magnesium phosphide sulfur + aluminum sulfide

Decomposition Compounds break down into elements and/or smaller compounds. calcium sulfide calcium + sulfur potassium chlorate potassium chloride + oxygen sodium carbonate sodium oxide + carbon dioxide magnesium hydroxide magnesium oxide + water hydrogen peroxide water + oxygen

Single Replacement A more active element will replace a less active element within a compound. 1. Active metals can replace less active metals lithium + copper(II) chloride copper + lithium chloride 2. Active metals can replace hydrogen in water sodium + water sodium hydroxide + hydrogen 3. Active metals can replace hydrogen in acid potassium + nitric acid potassium nitrate + hydrogen 4. Active nonmetals can replace less active nonmetals fluorine + aluminum chloride aluminum fluoride + chlorine

Gas-producing Certain combinations of ions will produce gases. 1. NH 4+ + OH- produces NH 3 + H 2 O ammonium chloride + sodium hydroxide ammonia + water + sodium chloride 2. H+ + CO 3 -2 produces CO 2 + H 2 O hydrochloric acid + calcium carbonate carbon dioxide + water + calcium chloride 3. H+ + SO 3 -2 produces SO 2 + H 2 O acetic acid + lithium sulfite sulfur dioxide + water + lithium acetate 4. H+ + S-2 produces H 2 S phosphoric acid + silver sulfide hydrogen sulfide + silver phosphate

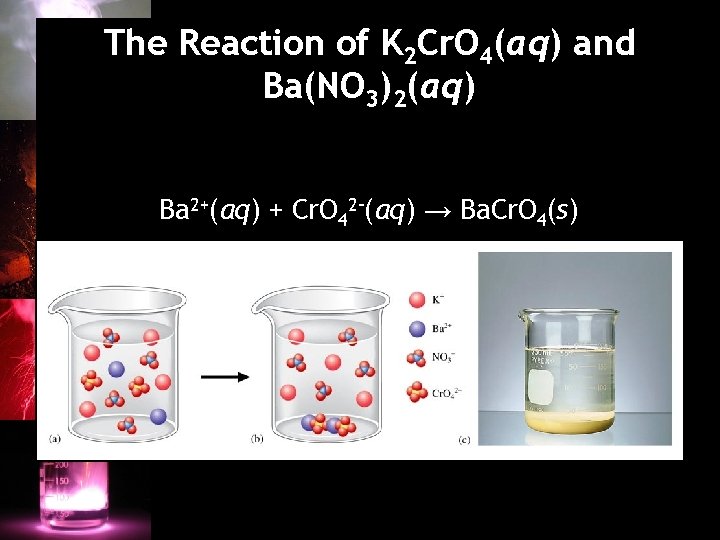
Section 4. 5: Precipitation Reactions Precipitation Reaction A double displacement reaction in which a solid forms and separates from the solution. • When ionic compounds dissolve in water, the resulting solution contains the separated ions. • Precipitate – the solid that forms.

The Reaction of K 2 Cr. O 4(aq) and Ba(NO 3)2(aq) Ba 2+(aq) + Cr. O 42–(aq) → Ba. Cr. O 4(s)

Precipitation of Silver Chloride

Precipitates • Soluble – solid dissolves in solution; (aq) is used in reaction. • Insoluble – solid does not dissolve in solution; (s) is used in reaction. • Insoluble and slightly soluble are often used interchangeably.

Simple Rules for Solubility

Concept Check Which of the following ions form compounds with Pb 2+ that are generally soluble in water? a) b) c) d) e) S 2– Cl– NO 3– SO 42– Na+

Section 4. 6: Describing Reactions in Solution Formula Equation (Molecular Equation) • Gives the overall reaction stoichiometry but not necessarily the actual forms of the reactants and products in solution. • Reactants and products generally shown as compounds. • Use solubility rules to determine which compounds are aqueous and which compounds are solids. Example: Ag. NO 3(aq) + Na. Cl(aq) Ag. Cl(s) + Na. NO 3(aq)

Complete Ionic Equation • Represents as ions all reactants and products that are strong electrolytes. Example: Ag+(aq) + NO 3 -(aq) + Na+(aq) + Cl-(aq) Ag. Cl(s) + Na+(aq) + NO 3 -(aq)

Net Ionic Equation • Includes only those solution components undergoing a change. • Show only components that actually react. Example: Ag+(aq) + Cl-(aq) Ag. Cl(s) • Spectator ions are not included (ions that do not participate directly in the reaction). • Na+ and NO 3 - are spectator ions.

Concept Check Write the correct formula equation, complete ionic equation, and net ionic equation for the reaction between cobalt(II) chloride and sodium hydroxide. Formula Equation: Co. Cl 2(aq) + 2 Na. OH(aq) Co(OH)2(s) + 2 Na. Cl(aq) Complete Ionic Equation: Co 2+(aq) + 2 Cl (aq) + 2 Na+(aq) + 2 OH (aq) Co(OH)2(s) + 2 Na+(aq) + 2 Cl (aq) Net Ionic Equation: Co 2+(aq) + 2 OH (aq) Co(OH)2(s)

Section 4. 7: Solution Stoichiometry Solving problems involving precipitates from solution makes use of: • MOLARITY • SOLUBILITY RULES • BALANCING EQUATIONS • LIMITING REACTANT CALCULATIONS

Solving Stoichiometry Problems for Reactions in Solution: 1. Identify the species present in the combined solution, and determine what reaction if any occurs. 2. Write the balanced net ionic equation for the reaction. 3. Calculate the moles of reactants. 4. Determine which reactant is limiting. 5. Calculate the moles of product(s), as required. 6. Convert to grams or other units, as required.

Example When aqueous solutions of Na. Br and Pb(NO 3)2 are mixed, Pb. Br 2 precipitates. Calculate the mass of Pb. Br 2 formed when 1. 25 L of 0. 0500 M Pb(NO 3)2 and 2. 00 L of 0. 0250 M Na. Br are mixed. 1. When Na. Br(aq) (Na+ and Br- ions) are mixed with Pb(NO 3)2(aq) (Pb+2 and NO 3 ions) the resulting solution will have the ions Na+, Br-, Pb 2+ and NO 3 -. Pb. Br 2 is insoluble and will precipitate. 2. Write the balanced ionic equation. Pb 2+ (aq) + 2 Br-(aq) Pb. Br 2(s) 3. Find moles of reactants. 0. 0500 M Pb(NO 3)2 contains 0. 0500 M Pb 2+ ions. Convert to moles. 1. 25 L x (0. 0500 mol Pb 2+) = 0. 0625 mol Pb 2+ / 1 = 0. 0625 L 0. 0250 M Na. Br contains 0. 0250 M Br- ions. Convert to moles. 2. 00 L x (0. 0250 mol Br-) = 0. 0500 mol Br- / 2 = 0. 0250 L

Example (cont. ) When aqueous solutions of Na. Br and Pb(NO 3)2 are mixed, Pb. Br 2 precipitates. Calculate the mass of Pb. Br 2 formed when 1. 25 L of 0. 0500 M Pb(NO 3)2 and 2. 00 L of 0. 0250 M Na. Br are mixed. 4. Find limiting reactant. Br- will be limiting. 5. Calculate moles of product. Pb+2(aq) + 2 Br-(aq) Pb. Br 2(s) 0. 0500 mol. Br- x 1 mol Pb. Br 2 = 0. 0250 mol. Pb. Br 2 2 mol Br 6. Convert to grams. 0. 0250 mol. Pb. Br 2 x 367. 00 g. Pb. Br 2 = 9. 18 g. Pb. Br 2 1 mol Pb. Br 2

Section 4. 8: Acid-Base Reactions Neutralization of a Strong Acid by a Strong Base

Performing Calculations for Acid–Base Reactions 1. List the species present in the combined solution before any reaction occurs, and decide what reaction will occur. 2. Write the balanced net ionic equation for this reaction. (*For the reaction of a strong acid with a strong base, the net ionic equation is always: H+ + OH- H 2 O(l)) 3. Calculate moles of reactants. (For reactions in solution, use molarity and volume of original solutions. ) 4. Determine the limiting reactant, where appropriate. 5. Calculate the moles of the required reactant or product. 6. Convert to grams or volume (of solution), as required.

Example What volume of 0. 100 M HCl solution is required in order to neutralize 25. 0 m. L of 0. 350 M Na. OH? 1. List species present in combined solution, decide reaction. H+, Cl-, Na+, OH 2. Write the balanced net ionic equation. H+(aq) + OH-(aq) H 2 O(l) 3. Find moles of reactants. 25. 0 m. L Na. OH x 1 L 1000 m. L x 0. 350 mol OH- = 8. 75 x 10 -3 mol OHL Na. OH 4. Find limiting reactant. There is no need to determine the LR because the problem requires the addition of just enough H+ ions to react exactly with the OH- ions present.

Example (cont. ) What volume of 0. 100 M HCl solution is required in order to neutralize 25. 0 m. L of 0. 350 M Na. OH? 5. Calculate moles of reactant. H+ and OH- react in a 1: 1 ratio, so 8. 75 x 10 -3 mol H+ ions is required to neutralize OH- ions. 6. Convert to needed volume. V x (0. 100 mol. H+/L) = 8. 75 x 10 -3 mol. H+ V = 8. 75 x 10 -2 L of 0. 100 MHCl required to neutralize 25. 0 m. L of 0. 350 M Na. OH

• An acid-base reaction is also known as a neutralization reaction. • Once the exact amount of base is added to react with an acid in solution, the acid is said to have been neutralized.

Acid–Base Titrations • Volumetric analysis – a method to determine the concentration of a particular substance by doing a titration. • Titration – delivery of a measured volume of a solution of known concentration (the titrant) into a solution containing the substance being analyzed (the analyte). • Equivalence point – enough titrant added to react exactly with the analyte. • Endpoint – the indicator changes color so you can tell the equivalence point has been reached. 49

For a Titration to be Successful: 1. The exact reaction with titrant and analyte has to be known. 2. The equivalence point must be marked accurately. 3. The volume of titrant needed to react equivalence point must be known accurately.

Section 4. 9: Redox Reactions • Reactions in which one or more electrons are transferred.

Video Link: Reaction of Sodium and Chlorine

Oxidation States • Provide a way to keep track of electrons in oxidation-reduction reactions (especially those with covalent substances) • Oxidation states of atoms in covalent compounds are obtained arbitrarily by assigning the electrons to particular atoms.

Rules for Assigning Oxidation Numbers 1. Each atom in a pure element has an oxidation number of zero (0). Example: I 2, Cu, Al, Na, O 2, P 4, S 8 all have zero oxidation states.

Rules for Assigning Oxidation Numbers 2. Monatomic ions have oxidation numbers equal to the charge on the ion. Example: Mg 2+ = +2, H 1+ = +1

Rules for Assigning Oxidation Numbers 3. Group 1 elements have an oxidation number of +1, group 2 have +2, and group thirteen have +3. Example: Na. Cl, Na = +1 Ca. SO 4, Ca = +2 Li. OH, Li = +1

Rules for Assigning Oxidation Numbers 4. Fluorine always has an oxidation number of -1 in compounds with other elements. -1 -1 Example: HF, BF 3

Rules for Assigning Oxidation Numbers 5. The oxidation number of H is +1 and for O is -2 in most compounds. a) Exception #1: When H forms a compound with a metal, it will have an oxidation number of -1. +1 -1 Example: Li. H

Rules for Assigning Oxidation Numbers 5. The oxidation number of H is +1 and for O is -2 in most compounds. b) Exception #2: When O forms a peroxide it will have a oxidation number of -1. +1 -1 Example: H 2 O 2

Rules for Assigning Oxidation Numbers 6. The sum of oxidation numbers must be zero (0) for a neutral compound, and for a polyatomic ion it must be equal to the ion charge. +1 -2 Example: Cl. O 1 -

Rules for Assigning Oxidation Numbers 7. Oxidation states do not have to be integers. Example: I 31 -, each I is = -1/3 Fe 3 O 4, each O is -2, then each Fe must be + 8/3

Examples Predict the oxidation states for each element in the following compounds. +4 -2 a. CO 2 +6 -2 b. Cr 2 O 7 -2 +1 +6 -2 c. H 2 SO 4 +5 -2 d. NO 2+1

Redox Characteristics • • Transfer of electrons Transfer may occur to form ions Oxidation – increase in oxidation state (loss of electrons); reducing agent Reduction – decrease in oxidation state (gain of electrons); oxidizing agent Le. O the lion says Ge. R! Lose electrons Oxidize Gain electrons Reduced

Redox Characteristics • The oxidizing agent (electron acceptor): causes another substance to be oxidized and therefore is itself reduced. • The reducing agent (electron donor): causes another substance to be reduced and therefore is itself oxidized.

Concept Check Which of the following are oxidationreduction reactions? a)Zn(s) + 2 HCl(aq) Zn. Cl 2(aq) + H 2(g) b)Cr 2 O 72 -(aq) + 2 OH-(aq) 2 Cr. O 42 -(aq) + H 2 O(l) c)2 Cu. Cl(aq) Cu. Cl 2(aq) + Cu(s)

Section 4. 10: Balancing Redox Reactions Balancing Oxidation–Reduction Reactions by Half Reactions To balance redox reactions, we use the halfreaction method. Oxidation states are assigned to each element. Elements being oxidized or reduced are identified. We then separate the reaction into two half-reactions: one involving oxidation and the other dealing with reduction. The method for balancing half-reactions varies depending on whether the reactions are taking place in acidic or basic (alkaline) solution.

The Half-Reaction Method for Balancing Redox Reactions in Acidic Solution 1. Assign oxidation numbers to identify which element is oxidized and which is reduced. 2. Write separate equations each for the reduction and oxidation half reactions. 3. For each half reaction: a. Balance first for the element changing oxidation states b. Balance for all other elements except hydrogen and oxygen. i. Balance oxygen with the use of H 2 O ii. Balance hydrogen using H+ c. Add electrons to balance for charge 4. If needed, multiply one or both half reactions by an integer to make the electrons gained equal the electrons lost. 5. Combine half-reactions, cancel identical species that appear on either side. 6. Make sure elements and charges are balanced.

Example: Balance the following reaction in acidic solution: +2 +7 -2 oxd +3 +2 Mn. O 4 -1 + Fe+2 Fe+3 + Mn+2 red [ Fe+2 Fe+3 + 1 e- ] x 5 Oxd ½ rxn: Red ½ rxn: 5 e- + 8 H+1 + Mn. O 4 -1 Mn+2 + 4 H 2 O 5 Fe+2 5 e- + 8 H+1 + Mn. O 4 -1 + 5 Fe+3 + 5 e Mn+2 + 4 H 2 O 5 Fe+2 5 Fe+3 + Mn+2 + 4 H 2 O

The Half-Reaction Method for Redox Reactions in Basic Solution 1. Use half-reaction method as specified for solutions that are acidic to obtain balanced half reactions as if H+ were present. 2. For each half reaction: a. Balance first for the element changing oxidation states b. Balance for all other elements except hydrogen and oxygen. i. Balance oxygen with the use of H 2 O ii. Balance hydrogen using H+ iii. *Add OH- to BOTH sides of the equation for each H+ you added iv. *Combine H+ and OH- on the same side to make H 2 O v. *Cancel/Reduce H 2 O c. Add electrons to balance for charge 3. If needed multiply one or both half reactions by an integer to make the electrons gained equal the electrons lost. 4. Combine half reactions, cancel identical species that appear on either side. 5. Make sure elements and charges are balanced.

Example: Balance the following redox reaction in basic solution: -2 +1 HS-1 Oxd ½ rxn: Red ½ rxn: 6 e- + oxd + [ OH-1 + HS-1 6 H 2 O + 6 H+1 + Cl. O 3 -1 6 OH-1 3 OH-1 + 6 e- + 6 H 2 O +5 -2 Cl. O 3 0 -1 -1 red. S + Cl-1 H 2 O S + H+1 + OH-1 + 2 e- ] x 3 Cl-1 + 3 H 2 O + 6 OH-1 + 3 H 2 O + 6 e- 3 HS-1 3 S + Cl. O 3 -1 Cl-1 + 3 H 2 O + 3 6 OH-1 3 HS-1 + Cl. O 3 -1 3 S + Cl-1 + 3 OH-1