Unit 4 The Periodic Table Review of Periodic

Unit 4: The Periodic Table

Review of Periodic Table Learning Target: Classify elements according to their location on the Periodic Table

Review � Identify the following as being an alkalai metal, alkaline earth metal, transition metal, semi-conductor, halogen, or noble gas: � Germanium � Tungsten � Potassium � Radon � Strontium � Uranium � Silicon � Beryllium � Palladium
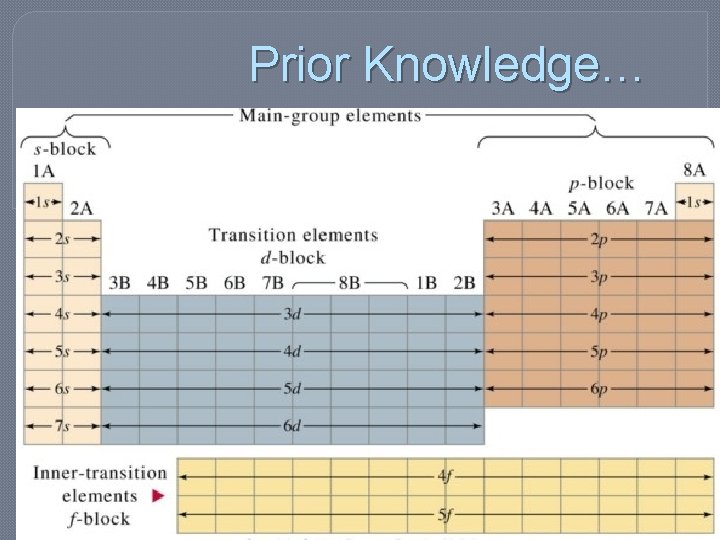
Prior Knowledge…

Review �Which of the following are main-group elements? �Magnesium �Vanadium �Antimony �Lead �Neptunium �Cesium �Rutherfordium �Boron �Indium
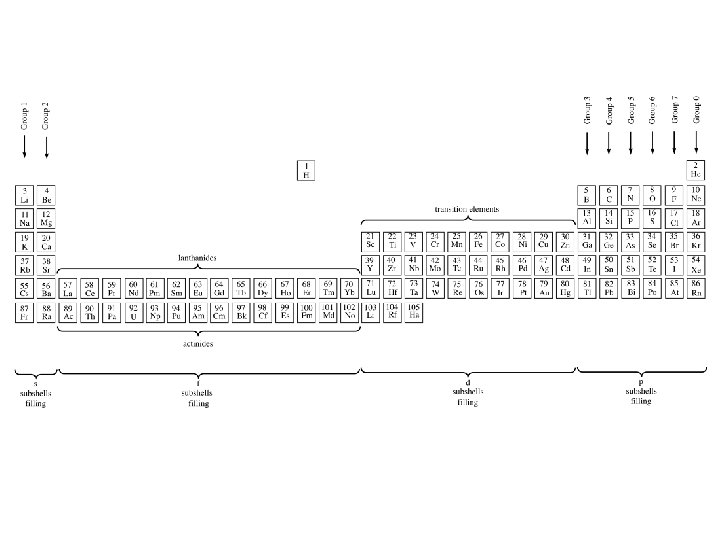

Development of the Periodic Table Section 1: Who’s Who? ? ? Learning Target: Explain the development of Periodic Law

Development of the Periodic Table � 1817 – Dobereiner • Grouped elements in TRIADS… • Three elements with similar properties � 1865 – Newlands • Arranged by increasing atomic MASS. • Law of Octaves = Similar properties over EIGHT elements

Development of the Periodic Table � 1870 – Mendeleev • Created an 8 Column table based on ATOMIC MASS • Elements with similar properties were in the same column

Development of the Periodic Table • Problems w/ Mendeleev’s table: 1. Some elements did not fit the order. Ex: Tellurium (Te) and Iodine (I) 2. There were gaps in the table • BUT he was able to predict the properties and masses of the unknown elements very accurately.
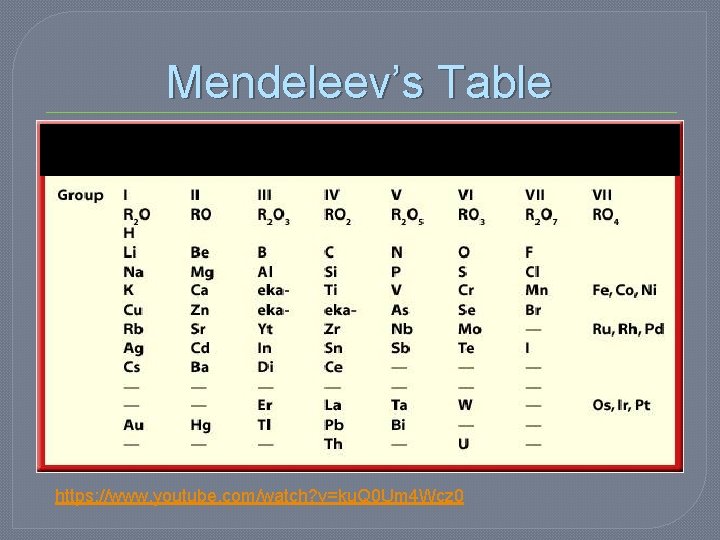
Mendeleev’s Table https: //www. youtube. com/watch? v=ku. Q 0 Um 4 Wcz 0

Development of the Periodic Table � 1913 – Mosley • Studied X-ray spectra of elements and noticed they correlated with atomic number • Arranged elements with increasing ATOMIC NUMBER � PERIODIC LAW: • Properties of the elements are a (periodic) function of their atomic number

Development of the Periodic Table � 1944 �Last –Glenn Seaborg rearrangement of the periodic table – created the actinide series

Who’s Who Timeline 1817 1865 1870 1913 1944 Dobereiner Newlands Mendeleev Mosley Seaborg Triads Law of Octaves First 8 Periodic Law column table Actinide Series

Modern Periodic Table Section 2
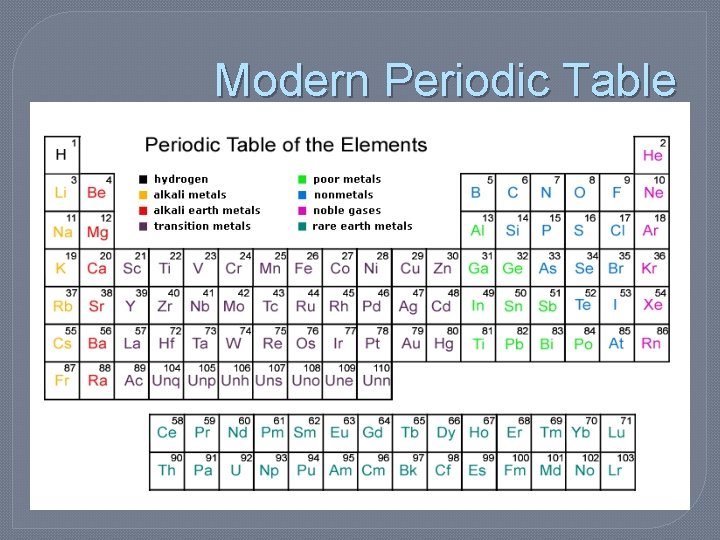
Modern Periodic Table
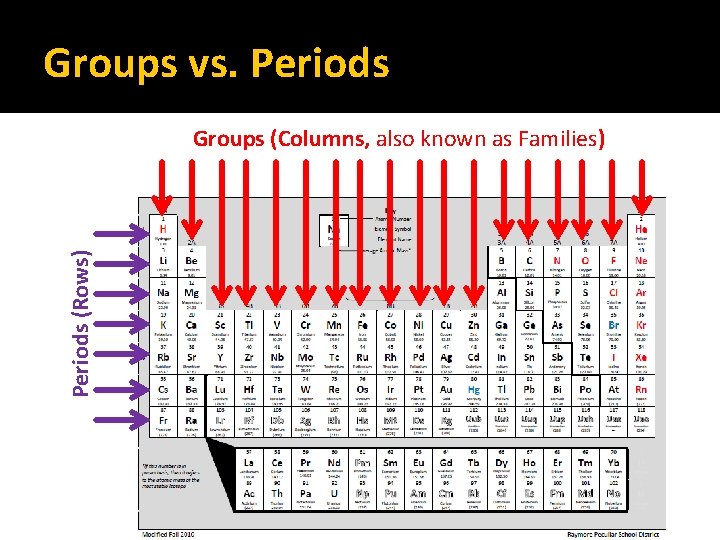
Groups vs. Periods (Rows) Groups (Columns, also known as Families)
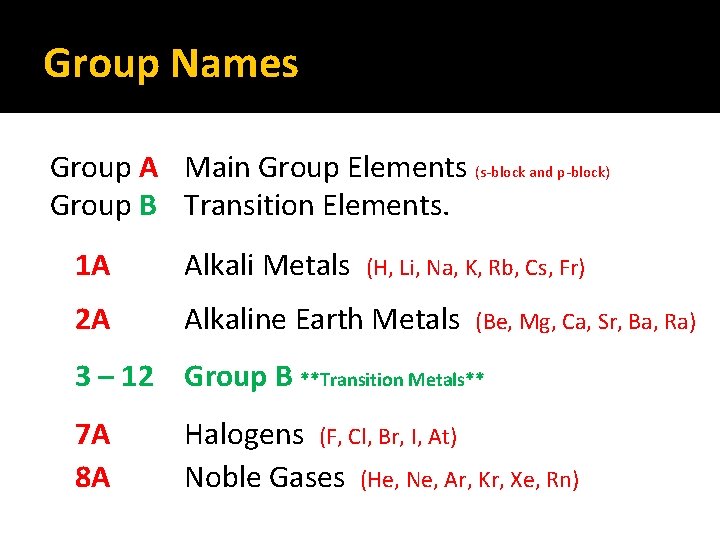
Group Names Group A Main Group Elements (s-block and p-block) Group B Transition Elements. 1 A Alkali Metals 2 A Alkaline Earth Metals (H, Li, Na, K, Rb, Cs, Fr) (Be, Mg, Ca, Sr, Ba, Ra) 3 – 12 Group B **Transition Metals** 7 A 8 A Halogens (F, Cl, Br, I, At) Noble Gases (He, Ne, Ar, Kr, Xe, Rn)
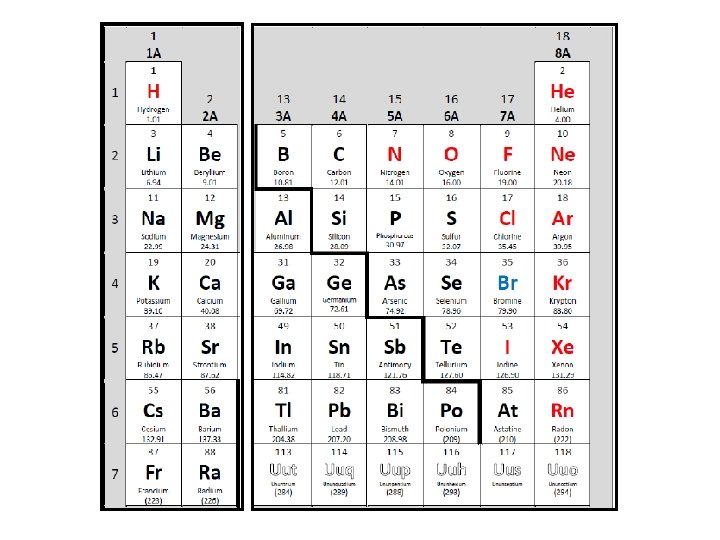

Basis of the Periodic Table �Atomic properties are determined by ELECTRON CONFIGURATION. � Ex. 1 s 2 2 p 6 3 s 2 3 p 3 �Therefore…the table is designed on the basis of electron configuration…

Valence Electrons VALENCE ELECTRONS Electrons in the OUTERMOST SHELL Highest Energy Level (n = 1, 2, 3…)

Valence Electrons �Valence Electrons: Electrons that are found in an atom’s outer most shell �Determines chemical properties of an atom �These are the electrons that can be gained, lost or shared when forming compounds
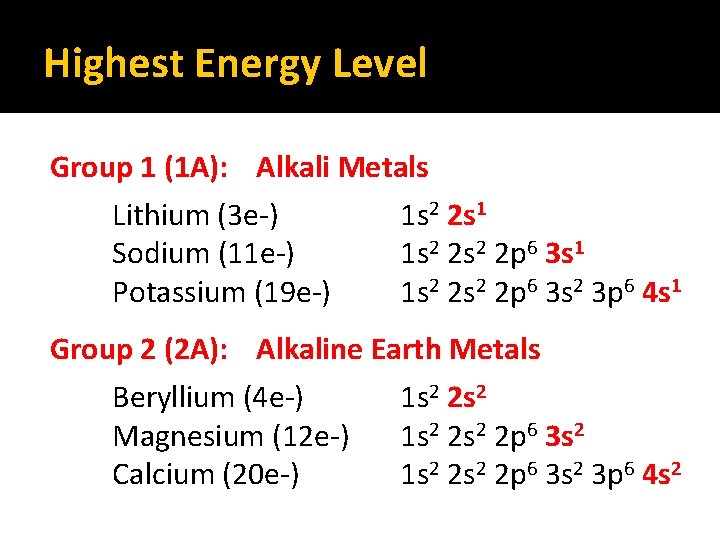
Highest Energy Level Group 1 (1 A): Alkali Metals Lithium (3 e-) Sodium (11 e-) Potassium (19 e-) 1 s 2 2 s 1 1 s 2 2 s 2 2 p 6 3 s 2 3 p 6 4 s 1 Group 2 (2 A): Alkaline Earth Metals Beryllium (4 e-) 1 s 2 2 s 2 Magnesium (12 e-) 1 s 2 2 p 6 3 s 2 Calcium (20 e-) 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2

Highest Energy Level Group 17 (7 A): Halogens Fluorine (9 e-) Chlorine (17 e-) Bromine (35 e-) 1 s 2 2 p 5 1 s 2 2 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 10 4 p 5 Group 18 (8 A): Noble Gases Neon (10 e-) 1 s 2 2 p 6 Argon (18 e-) 1 s 2 2 s 2 3 p 6 Krypton (36 e-) 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 10 4 p 6
Modern Periodic Table

Main-Group Elements: Valence Electrons/ Lewis Dot Diagrams

Modern Periodic Table OCTET RULE 8 valence electrons make the atom UNREACTIVE. Unreactive = Very Stable Atoms react with other atoms to fill their octet. 1. Gain electrons. 2. Lose electrons. 3. Share electrons. Ionic Bonding Covalent Bonding

Stability of the Periodic Table � OCTET RULE… • 8 electrons in the outer energy level renders an atom UNREACTIVE • Unreactive = Very Stable • So…which elements are considered unreactive?
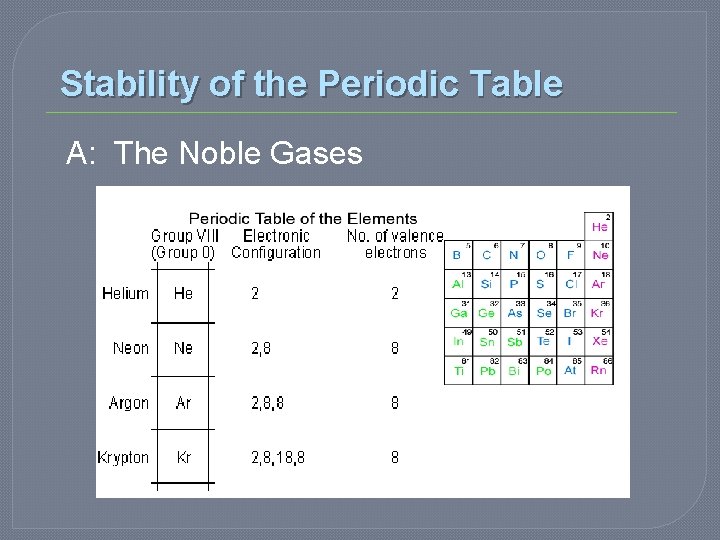
Stability of the Periodic Table A: The Noble Gases

How do other atoms become stable? �Atoms react with other atoms so all atoms end up with FULL OUTER ENERGY LEVELS (i. e. compounds) 1. They may add electrons 2. They may lose electrons 3. They may share electrons

Crash Course Chemistry The Periodic Table https: //www. youtube. com/watch? v=0 RRVV 4 Diomg

Metals vs. Nonmetals Section 3 Learning Target: Identify properties of metals, nonmetals and metalloids

Color-Coded Periodic Table �Don’t forget your color-coded periodic table… �It identifies the… • Groups • Periods • Main Group Elements

Color-Coded Periodic Table � It also classifies elements as… • Metals: �Alkali metals �Alkaline Earth metals �Transition metals �Lanthanide Series �Actinide Series �Other Metals • Nonmetals: �Noble gases �Halogens �Other nonmetals • Metalloids: �semiconductors
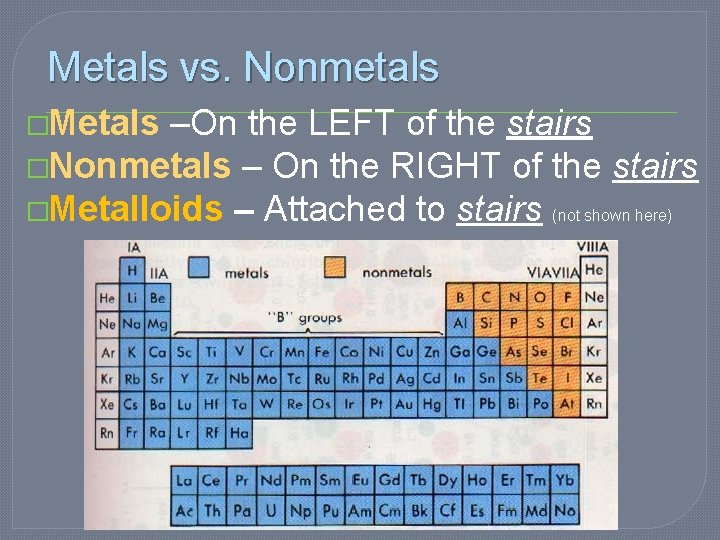
Metals vs. Nonmetals �Metals –On the LEFT of the stairs �Nonmetals – On the RIGHT of the stairs �Metalloids – Attached to stairs (not shown here)
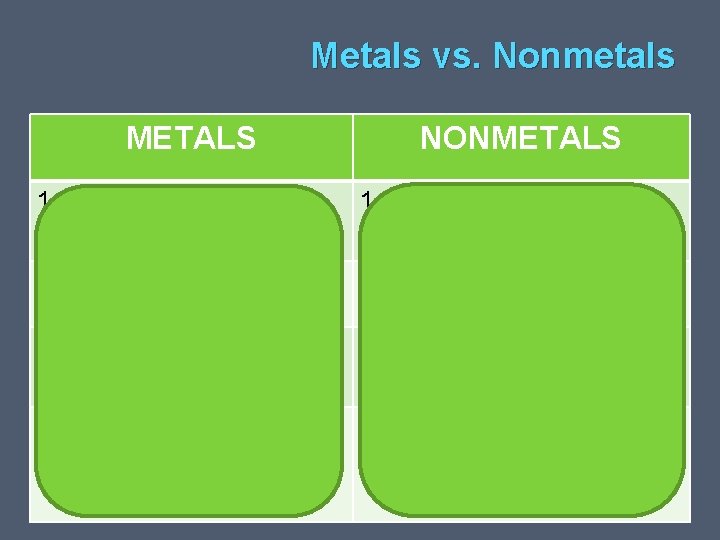
Metals vs. Nonmetals METALS 1. NONMETALS Metals vs. Nonmetals Typically Solids 1. Gasses or BRITTLE solids 2. Shiny 2. Dull 3. Conduct Heat and Electricity 3. Insulators 4. Tendency when forming compounds to lose outer compounds to GAIN or electrons SHARE electrons

Metals vs. Nonmetals RULE of THUMB � Metals: • 3 or fewer outer energy level electrons � Nonmetals: • 5 or more outer energy level electrons � Metalloids: • Properties of both Metals and Nonmetals • Semiconductors

Periodic Trends Section 4 Learning Target: Describe element trends on the Periodic Table

Periodic Trends �What is a trend? �A predictable change in a particular direction �(FOCUS ON MAIN GROUP ELEMENTS)

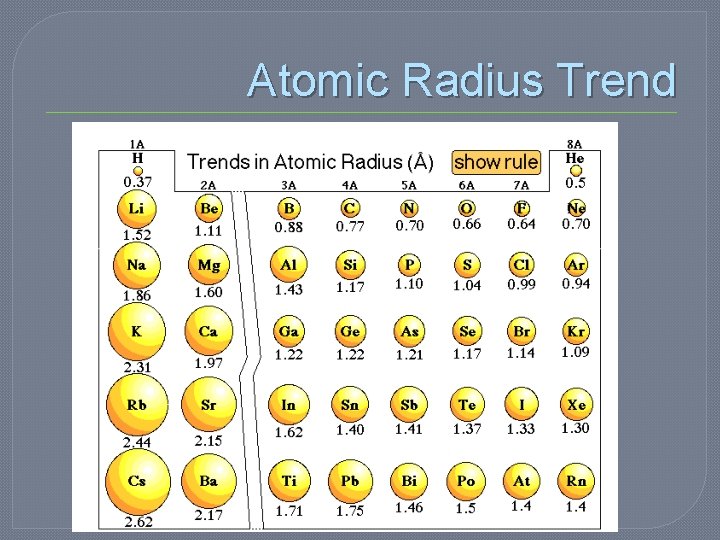
Periodic Trends: Atomic Radii � ½ the distance between the nuclei of identical atoms that are bonded together � Atomic Radii increase from top to bottom and from right to left on the periodic table Radius = d/2
Periodic Trends – Atomic Radii Increases Periodic Table
Atomic Radius Trend

Atomic Radius Trends � Increases as you move down due to electron shielding • electrons in the inner energy levels are between the nucleus and the outer electrons and therefore are shielded from being pulled away

Atomic Radius Trends � Decrease as you go right due to increasing charge of the nucleus (means increases as you go left due to decreasing charge of nucleus) • although more electrons are also present they are at the same distance from the nucleus b/c they are in the same energy level

Which has a larger atomic radius? �Gallium �Thallium �Yttrium �Strontium �Helium �Neon �Hafnium �Vanadium

Periodic Trends: Ionization Energy Ions � Atom or group of atoms that has a positive or negative charge • Positive charge occurs when atom loses electrons • Ex: Na+: Sodium that has lost an electron, and now has 10 e • Negative charge occurs when atom gains electrons • Ex: Cl-: Chlorine that has gained an electron, and now has 18 e- � This process that results in an ion requires energy—this process is referred to as ionization

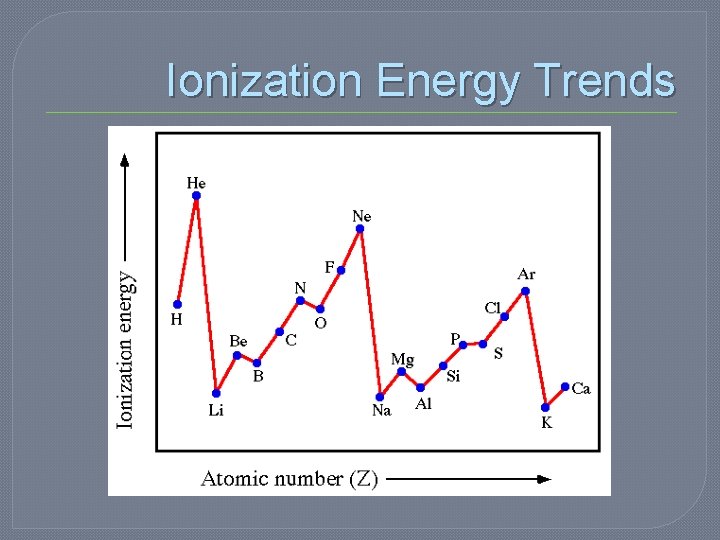
Periodic Trends: Ionization Energy �Energy required to remove one electron from a neutral atom of an element • This happens when compounds form � Ionization energy tends to increase from bottom to top and left to right
Periodic Trends – Ionization Energy Increases Periodic Table
Ionization Energy Trends

Ionization Energy Trends �As you move across, the nucleus gets larger and holds the electrons more tightly THEREFORE harder to remove �Requires MORE energy (higher ionization energy)

Ionization Energy Trends �As you move down, the outer most electrons are farther away from the nucleus THEREFORE easier to remove �Requires LESS energy (lower ionization energy)

Which has a higher ionization energy? �Iron �Cobalt �Cesium �Rhenium �Sulfur �Tin �Iodine �Bismuth

Periodic Trends: Electron Affinity �Energy change that occurs when an electron is acquired by a neutral atom (The ability to attract and hold an electron. ) � Electron affinity follows the same trend as Ionization Energy (Bottom to Top/Left to Right generally more exothermic)
Periodic Trends – Electron Affinity Increases Periodic Table

Which has a higher electron affinity? �Gold �Osmium �Platinum �Cadmium �Barium �Beryllium �Fluorine �Aluminum
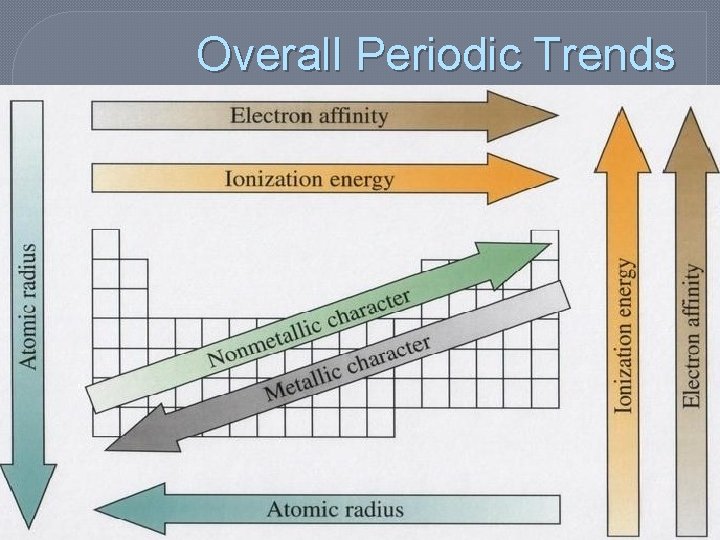
Overall Periodic Trends
- Slides: 56