Unit 4 Solutions and Kinetics Solubility Solubility the

Unit 4: Solutions and Kinetics Solubility

Solubility: the amount of a substance that dissolves in a given quantity of solvent at a given temp to form a saturated solution Units: grams/100 grams water
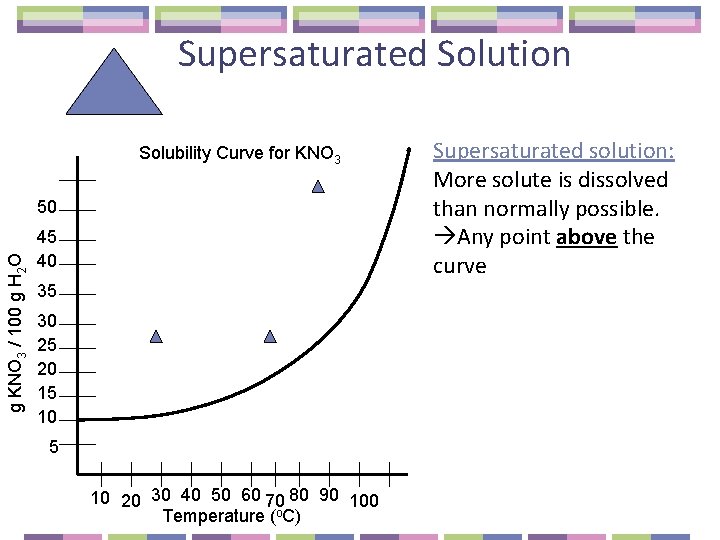
Supersaturated Solution Solubility Curve for KNO 3 g KNO 3 / 100 g H 2 O 50 45 40 35 30 25 20 15 10 20 30 40 50 60 70 80 90 100 Temperature (o. C) Supersaturated solution: More solute is dissolved than normally possible. Any point above the curve
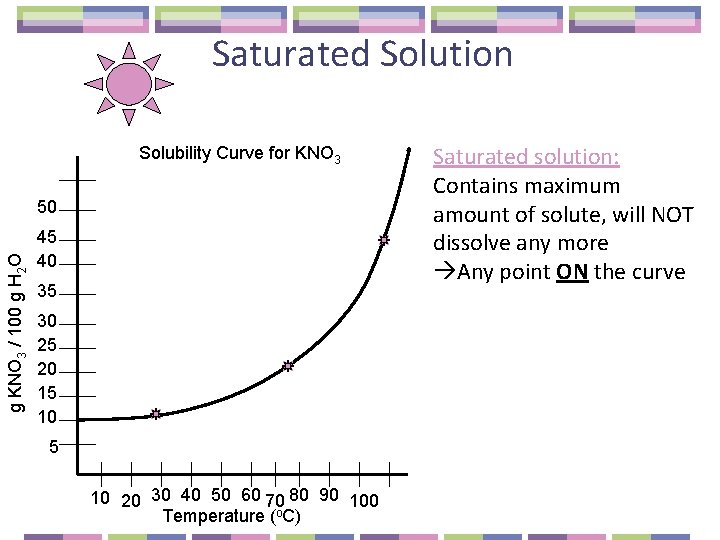
Saturated Solution Solubility Curve for KNO 3 g KNO 3 / 100 g H 2 O 50 45 40 35 30 25 20 15 10 20 30 40 50 60 70 80 90 100 Temperature (o. C) Saturated solution: Contains maximum amount of solute, will NOT dissolve any more Any point ON the curve
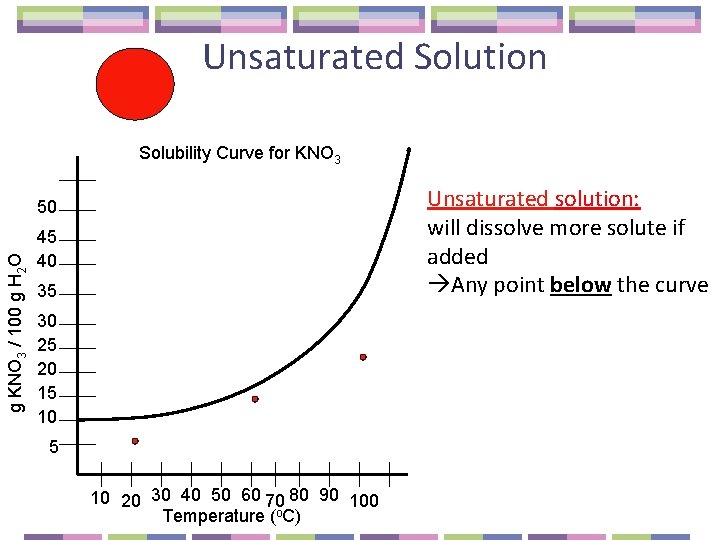
Unsaturated Solution Solubility Curve for KNO 3 Unsaturated solution: will dissolve more solute if added Any point below the curve g KNO 3 / 100 g H 2 O 50 45 40 35 30 25 20 15 10 20 30 40 50 60 70 80 90 100 Temperature (o. C)

g/100 g H 2 O Solubility curves use concentrations in: g solute/100 g H 2 O To calculate: mass solute (in grams) x 100 g H 2 O mass H 2 O (in grams)
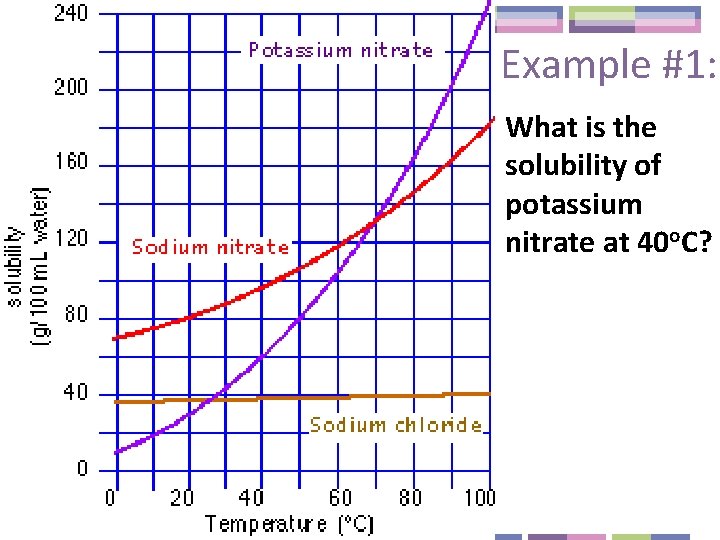
Example #1: What is the solubility of potassium nitrate at 40 o. C?
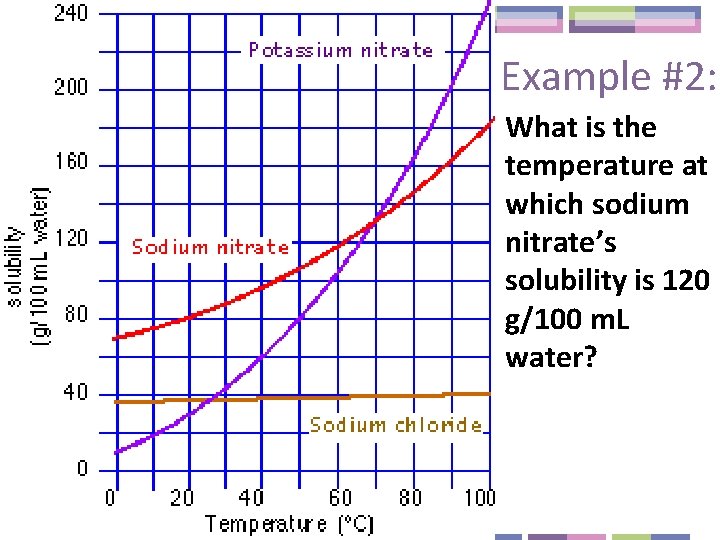
Example #2: What is the temperature at which sodium nitrate’s solubility is 120 g/100 m. L water?
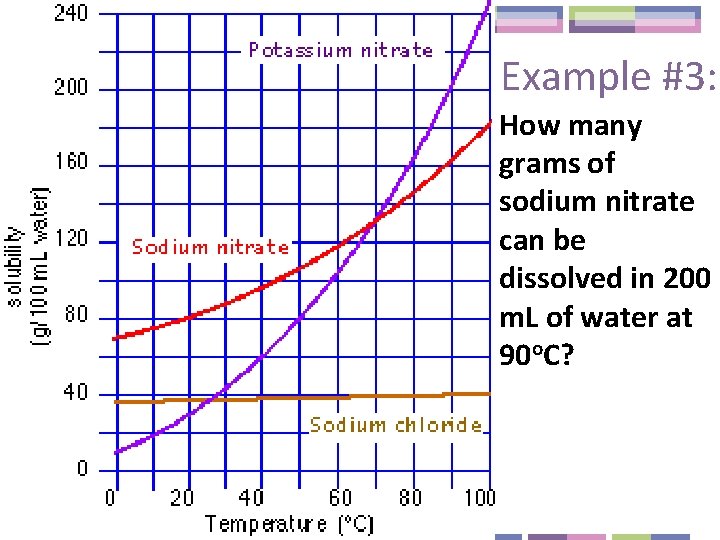
Example #3: How many grams of sodium nitrate can be dissolved in 200 m. L of water at 90 o. C?
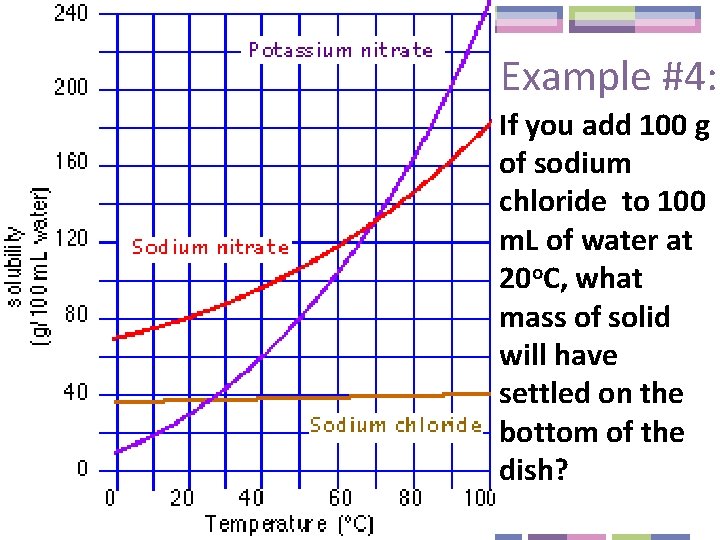
Example #4: If you add 100 g of sodium chloride to 100 m. L of water at 20 o. C, what mass of solid will have settled on the bottom of the dish?
- Slides: 10