Unit 4 Periodic Table Have your Periodic Table

Unit 4: Periodic Table Have your Periodic Table out everyday for this unit Chemistry

I. Historical Development of the Table 1860’s A. Dmitri Mendeleev (_____) • Arranged elements into table based on… similarities in the physical and chemical properties • thought similarities were function of atomic mass • Noticed that some elements seemed out of order (Iodine and Tellurium) Mendeleev Video Clip

I. Historical Development of the Table 1913 A. Henry Mosely (_____) • Established that properties of elements were Atomic Number functions of ______ 1. Present table based on Atomic Number ________ 2. Elements’ properties depend on Structure of atom ________ 3. Properties of elements vary Systematically ________ throughout the table
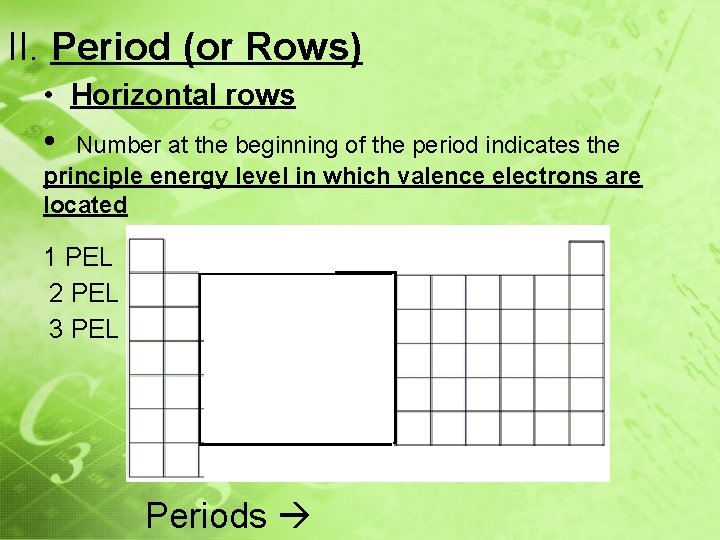
II. Period (or Rows) • Horizontal rows • Number at the beginning of the period indicates the principle energy level in which valence electrons are located 1 PEL 2 PEL 3 PEL Periods

II. Period (or Rows) Example: 4 A) What period is potassium and bromine in? ____ B) Based on the period, how many principal energy levels do potassium and bromine have? ________ 4 Properties change as you move across a period • ______________________ Example: A) What period is potassium and bromine in? ______ 4 B) Based on the period, how do the properties of potassium and bromine compare? ________ Not similar
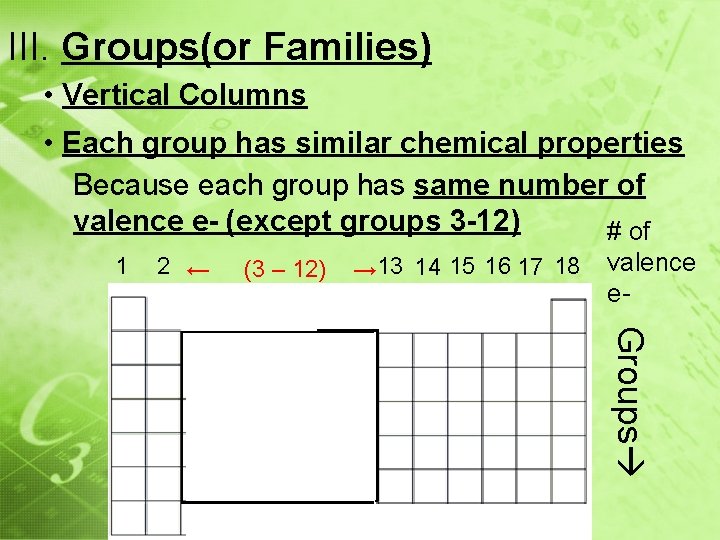
III. Groups(or Families) • Vertical Columns • Each group has similar chemical properties Because each group has same number of valence e- (except groups 3 -12) # of 1 2 ← (3 – 12) → 13 14 15 16 17 18 valence e- Groups

II. Period (or Rows) Example: A) What group is magnesium and calcium in? 2 ____ B) Based on the group, how do the properties of magnesium and calcium compare? ________ Similar (both have 2 valence e-)

III. Groups(or Families) Sample Questions: Which sequence of atomic numbers represents elements which have similar chemical properties? A) 19, 23, 30, 36 C) 9, 16, 33, 50 B) 3, 12, 21, 40 D) 4, 12, 38, 88 Which two elements have the most similar chemical properties? A) Aluminum and Barium C) Nickel and Phosphorous B) Chlorine and Sulfur D) Sodium and Potassium

IV. Metals • Atoms that lose e- and form positive ions (cations) when bonding • Alloy: Mixture of metals by melting them together

IV. Metals • Properties of Metals: 1. Low ionization energy and electronegativity 2. Good conductors of heat and electricity 3. Exhibit metallic luster (shine) 4. Malleability (can be pounded into thin sheets) 5. Ductility (can be pulled into thin wires) 6. More than 2/3 of the elements are metals 7. High densities 8. Mercury (Hg) is a metal which is a liquid at room temperature Fr (Francium) 9. Most active metal (bottom left corner): ____

IV. Metals CHECKS FOR UNDERSTANDING 1. Which element is malleable and can conduct electricity in the solid phase? A) Iodine B) Phosphorous C) Sulfur D) Tin 2. What happens to metals when they bond? What would happen to their atomic size? Lose electrons; become smaller 3. Where are the most reactive metals located? What about their atomic structure that makes them so reactive? Bottom left corner; the electrons are far away (big atomic radius), so electrons can be easily removed (low ionization energy/electronegativity)

V. Nonmetals • atoms gain e- and form negative ions (anions) when bonding

V. Nonmetals • Properties of Nonmetals: 1. High ionization energy and electronegativity 2. Poor conductors of heat and electricity 3. Brittle and hard 4. Low densities F (Fluorine) (top right corner) 5. Most active nonmetal: ____ Graphite is an allotrope of carbon. It is used in pencils (brittle, soft, and low density)

V. Nonmetals CHECKS FOR UNDERSTANDING 1. Which element is brittle and not a conductor of electricity? A) Sulfur B) Sodium C) Potassium D) Argon 2. What happens to nonmetals when the bond? What happens to their atomic radius when they bond? Gain electrons; become larger 3. Where are the most reactive nonmetals located? What about their atomic structure that makes them so reactive? Top Right corner (Group 17); small atomic radius so they attract electrons (High electronegativity/ ionization energy

VI. Metalloids • Atoms that gain or lose e- and form ions when bonding • Have properties of both metals and nonmetals • Can be located using the “staircase” (see periodic table) • Boron, Silicon, Germanium, Arsenic, Antimony, Tellurium

VI. Metalloids CHECKS FOR UNDERSTANDING 1. Which element has both metallic and nonmetallic properties? A) Rb B) Rn C) Si D) Sr 2. Which list of elements contains 2 metalloids? A) Si, Ge, Po, Pb C) As, Bi, Br, Kr B) Si, P, S, Cl D) Po, Sb, I, Xe
VII. Periodic Properties A. Covalent Atomic Radius: ½ of the distance between adjacent nuclei of the element
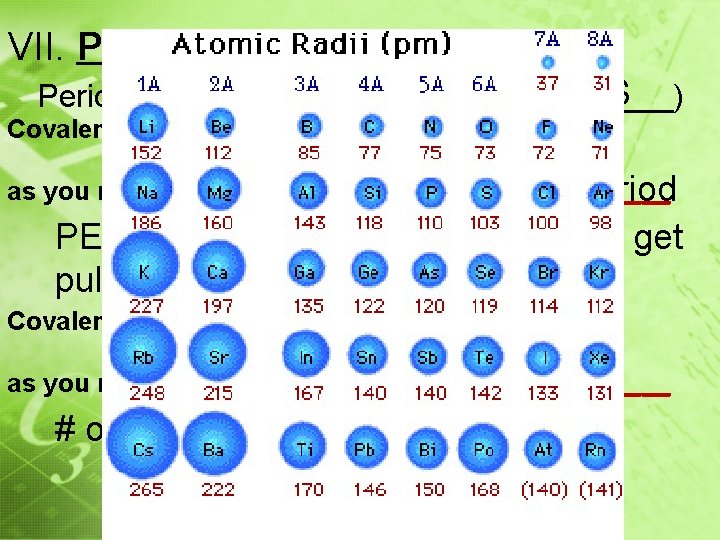
VII. Periodic Properties S Periodic Trend (See Ref Tabs – Table _____) decreases Covalent Atomic Radius ______ as you move from _______________ Left to right across a period PEL remains the same and outer e- get pulled closer to the nucleus Covalent Atomic Radius ______ increases as you move from _______________ down a group # of energy levels increase

VII. Periodic Properties B. Ionic Radius: A loss or gain of e- causes an increase or reduction in atom’s size cations lose • Metals: _____ electrons when they form ions ( _______ ) and ionic radius smaller _____ than neutral atom 2 -8 -1 2 -8

VII. Periodic Properties B. Ionic Radius: anions • Nonmetals: _____ electrons when they form ions ( _____ ) gain and ionic radius _____ than neutral atom bigger 2 -8 -7 2 -8 -8

IV. Periodic Properties Sample Questions: • Which element has an atomic radius that is greater than its ionic radius? A) S C) F B) K D) O • In period 4, the atom with the largest covalent radius is located in group… A) 1 C) 3 B) 13 D) 18

VII. Periodic Properties C. Ionization Energy: Amount of energy needed to remove the most loosely bound e - (valence e -)

VII. Periodic Properties S Periodic Trend (See Ref Tabs – Table _____) Ionization Energy ______ increases as you move from _______________ Left to right across a period Number of protons and valence e- increases and size decreases, so e- are held tighter Ionization Energy ______ decreases as you move from _______________ down a group e- are farther away, so easier to remove e- and valence e- are shielded by inner e-

VII. Periodic Properties D. Electronegativity: Measure of atom’s attraction for electrons when bonded S • Periodic Trend (See Reference Tables – Table _____): increases Electronegativity _______ as you move from _______________ Left to right across a period decreases as you move from Electronegativity ______________________ down a group

IV. Periodic Properties Check for Understandings • What are the electronegativities for group 18 elements? Why do you think they have “this” electronegativity? They do not have electronegativity numbers. Why? Because they already have their 8 valence electrons (octet) • Which element attracts electrons the most when bonding? A) Ca C) Br B) Sr D) I • Which element gives off its valence electrons the easiest? A) Ca C) Br B) Sr D) I

VIII. Groups within Groups Alkali Metals A. Group 1: ________ • All soluble (dissolves) in water and strongly basic • Hydrogen is the exception on the periodic table – it belongs nowhere : ( Brainiac – Alkali Metals MYTHBUSTERS – ALKALI METALS Part 1 Part 2

VIII. Groups within Groups Alkaline Earth Metals B. Group 2: ___________ • Have 2 valence electrons • Form +2 charge when they bond In General for Groups 1 and 2: As you move down, they become more reactive

VIII. Groups within Groups CHECKS FOR UNDERSTANDING 1. As you move down groups 1 and 2 elements become more reactive. Explain why this happens based on atomic size and ionization energy/electronegativity. Size increases, ionization energy decreases (easier to remove e-) so they become more reactive 2. What will be the charge on a Ca ion when it bonds? +2 3. Based on your answer to question 2, what happens to the atomic size once it becomes an ion? Smaller (loses last energy level)

VIII. Groups within Groups Metals C. Groups 3 -12: Transition ________ • Dense Metals and high melting points • Form multiple ions and less reactive than Groups 1 and 2 • Form ions that have color

VIII. Groups within Groups CHECKS FOR UNDERSTANDING 4. Identify the metal that has multiple oxidation states. A) K B) Ba C) Be D) Pd 5. Which compound forms a green aqueous solution? A) Rb. Cl B) Ca. Cl 2 C) Ni. Cl 2 D) Be. Cl 2 6. Which set of properties is most characteristic of transition elements? A) Colorless ions in solutions, multiple positive oxidation states B) Colorless ions in solutions, multiple negative oxidation states C) Colored ions in solutions, multiple negative oxidation states D) Colored ions in solutions, multiple positive oxidation states

VIII. Groups within Groups Halogens D. Group 17: ________ • Have 7 valence e-, become -1 charge ion • Mostly nonmetallic elements

VIII. Groups within Groups Noble Gases E. Group 18: ________ • All energy levels are full (2 or 8 valence electrons) • Do not combine with other elements to form molecules (exist as monatomic in normal room conditions) • They are stable • All other elements want to be like them

VIII. Groups within Groups CHECKS FOR UNDERSTANDING 7. Which halogen is most reactive? Explain why, based on atomic size, electronegative/ionization energy. F: smallest in size so attraction for electrons is very high (electronegativity) 8. Explain why noble gases don’t typically react with other elements? Their outer energy level is completely full. They do not want to gain anymore e-

IX. Phases of Matter • All of the information on the Periodic Table is given at STP • STP - Standard Temperature and Pressure ( 0 o. C and 1 atm/101. 3 k. Pa (sea level pressure)) • Gases - H, N, O, F, Cl and Noble Gases • Liquids - Br, Hg • Solids - Everything else

X. Other Categories • Diatomics Molecule containing 2 identical atoms • Remember them: I, Br, Cl, F, O, N, H (7 -H club) • Radioactive An element that ALL its isotopes are unstable and break down spontaneously • Remember them: Atomic # greater than 84 and 43 (Technitium), 61 (Promethium)

X. Other Categories • Synthetic Man-made elements (all are radioactive as well) • Remember them: Atomic # greater than 93 and 43 (Technitium), 61 (Promethium)

X. Other Categories • Allotropes 2 or more different structural forms of an element in the same phase • Remember them: Examples: C (s) – Diamond, graphite, coal, and Buckminster O (g) – O 2 – Dioxygen- what we breath - O 3 – Ozone - Toxic Diamond Graphite Coal Buckminster
- Slides: 37