UNIT 4 ELECTRICAL PRINCIPLES TECHNOLOGIES Initial Ideas What

















- Slides: 22

UNIT 4: ELECTRICAL PRINCIPLES & TECHNOLOGIES

Initial Ideas What is electricity? Why is knowledge about electricity important? Apollo 13 - “Failure is not an option”

Electrical Safety Don’t use electricity near water Water is a conductor Don’t use power cords that are frayed Can create a ‘short’ circuit Unplug devices before fixing Don’t put anything into sockets that are made for them

Stay away from power lines Can create ‘short’ circuit if contact is made Don’t overload circuits Use fuses or circuit breakers Avoid high places during thunderstorms

Storage of Electrical Energy Static Electricity Some particles in the ATOM are ELECTRICALLY CHARGED PROTONS = POSITIVELY charged ELECTRONS = NEGATIVELY charged

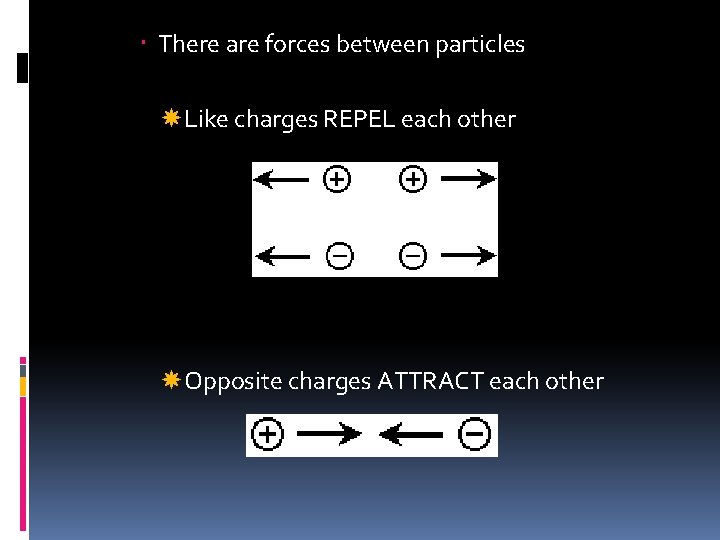
There are forces between particles Like charges REPEL each other Opposite charges ATTRACT each other

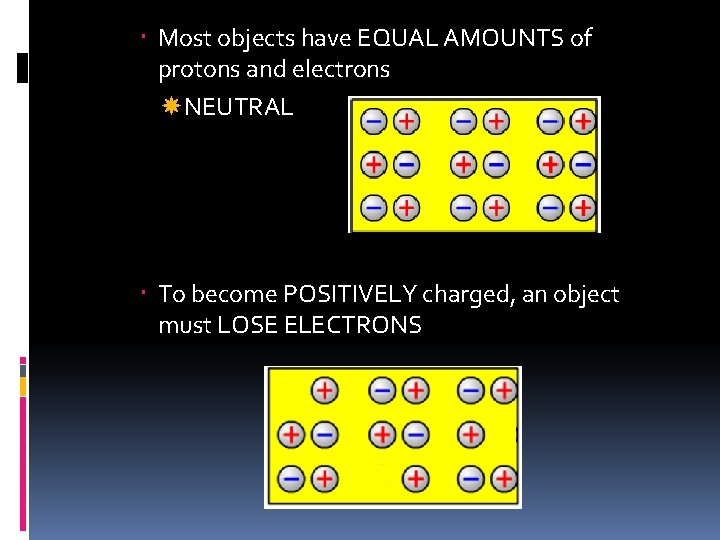
Most objects have EQUAL AMOUNTS of protons and electrons NEUTRAL To become POSITIVELY charged, an object must LOSE ELECTRONS

To become NEGATIVELY charged, an object must GAIN ELECTRONS *Only ELECTRONS are gained or lost by an atom!*

When an object loses or gains electrons, we say the object has built up STATIC CHARGE Static = STATIONARY, NOT MOVING

When NEUTRAL objects are brought near CHARGED objects, they cause CHARGE SEPARATION

Electrical Discharge (Static Shock) Build up of a negative charge that JUMPS to another object Attracted to positive charge Ex. Walking across a carpet and touching a light switch e- are transferred from the carpet to your body You are now negative (compared to the light switch) Electrons JUMP from you to light switch

Static vs. Discharge vs. Current Static = Stationary charge Discharge = Charge jumps Current = Continuously moving charge

Van de Graff Generators Produce a build up of static using friction Rubber belt rubs on a metal sphere Electrons are transferred from the rubber to the metal Electric charge then transfers to you when you touch VDG Sparky Lights

Cells & Batteries Electrochemical cell Package of chemical designed to produce small amounts of electricity Electricity is produced from chemical reactions Electrons travel FROM negative (-) terminal, through the device and BACK to the positive (+) terminal

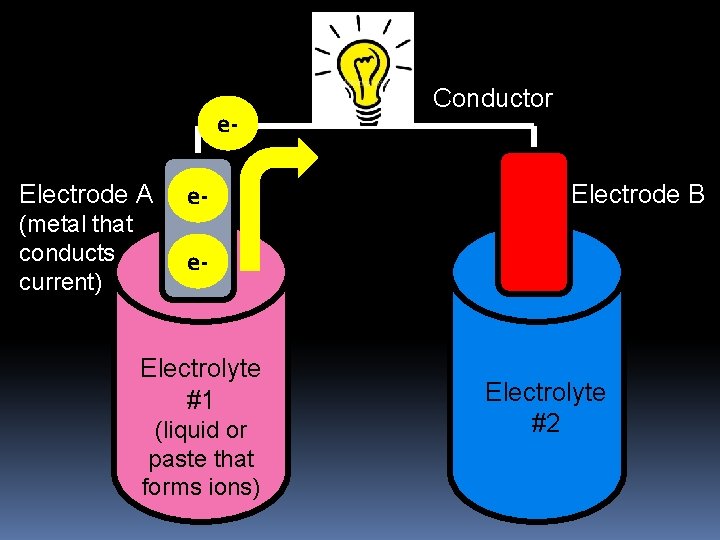
e. Electrode A (metal that conducts current) e- Conductor Electrode B e- Electrolyte #1 (liquid or paste that forms ions) Electrolyte #2

An electrochemical cell consists of: 1) Electrodes 2 DIFFERENT metals through which electric current enters or leaves 2) Electrolyte Paste or liquid that forms ions One electrode becomes loses electrons (-); other gains electrons (+) Results in a ‘potential difference’ Voltage

Types of electrochemical cells 1. Primary Cells Produces electricity from chemical reactions that CAN NOT be reversed a) Dry cells Chemicals are in a paste & are sealed Good combination of cost, output, shelf life, reliability & leak resistance b) Wet cells Uses a liquid electrolyte (acid) Easier to make than dry cells Danger in spilling electrolyte (corrosive)

2. Secondary Cells Rechargeable cells Chemical reaction CAN be reversed by using an external electrical source to run electricity back through the cell Reversed flow of electrons restores the reactants Most common reaction – between nickel oxide and cadmium (Ni-Cd)

Battery When more than one cell is connected together Most are sealed into cases with only 2 terminals Why would you connect more than one cell together? Produces more voltage = more power

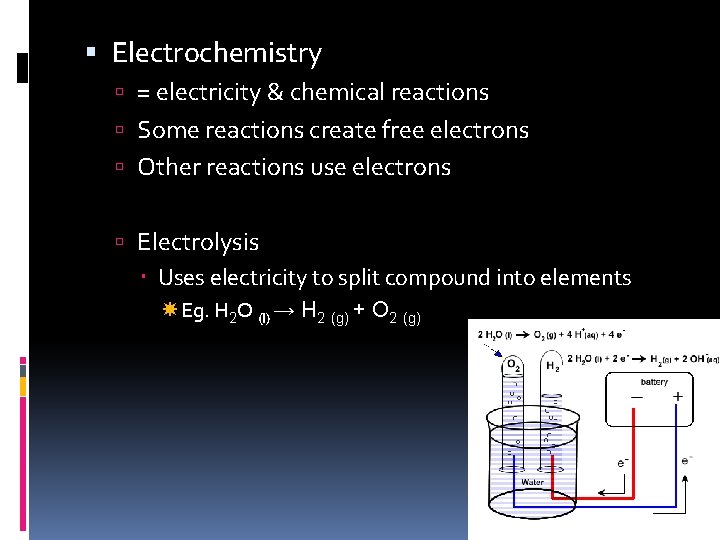
Electrochemistry = electricity & chemical reactions Some reactions create free electrons Other reactions use electrons Electrolysis Uses electricity to split compound into elements Eg. H 2 O (l) → H 2 (g) + O 2 (g)

Electroplating Use of electricity to coat an object with a thin layer of metal Flow of electrons deposits atoms of (+) charged metal onto (–) object Used to protect objects from corrosion

Anodizing Coat aluminum objects with aluminum oxide Makes object harder Electrorefining Used to remove impurities