UNIT 4 Chemical Names Formulas Rxns Chapter 9


















- Slides: 20

UNIT 4 – Chemical Names, Formulas & Rxns Chapter 9 – Chemical Names and Formulas Chapter 11 – Chemical Reactions Chapter 9 Chemical Names and Formulas Anything in black letters = write it in your notes (‘knowts’)

What is the difference between a chemical name and a chemical formula? Chemical Names Chemical Formulas sodium chloride Na. Cl glucose C 6 H 12 O 11 carbon dioxide CO 2

9. 1 – Naming Ions Monatomic Ions – a single atom with a charge Oxidation Number – the charge of an ion


Usually, a roman numeral is used to show the charge of a variable cation. Copper (I) – Cu+ Copper (II) – Cu 2+ Iron (II) – Fe 2+ Iron (III) – Fe 3+

Classical names can also be used. Copper (I) – Cu+ - cuprous Copper (II) – Cu 2+ - cupric Iron (II) – Fe 2+ - ferrous Iron (III) – Fe 3+ - ferric

Monatomic anions end in –ide Fluoride, chloride, bromide, etc…

Polyatomic Ions – more than one atom with a charge (table on p. 268 or on yellow p. table) These ions often stay together in chemical rxns. Common Polyatomic Ions Charge Formula Name 1– HSO 4– NO 2– Cl. O– Hydrogen sulfate Nitrite Hypochlorite 2– SO 32– SO 42– CO 32– Sulfite Sulfate Carbonate 3– PO 43– Phosphate 1+ NH 4+ Ammonium

Polyatomics to memorize… Nitrate Sulfate Carbonate Phosphate Hydroxide Acetate (ethanoate)

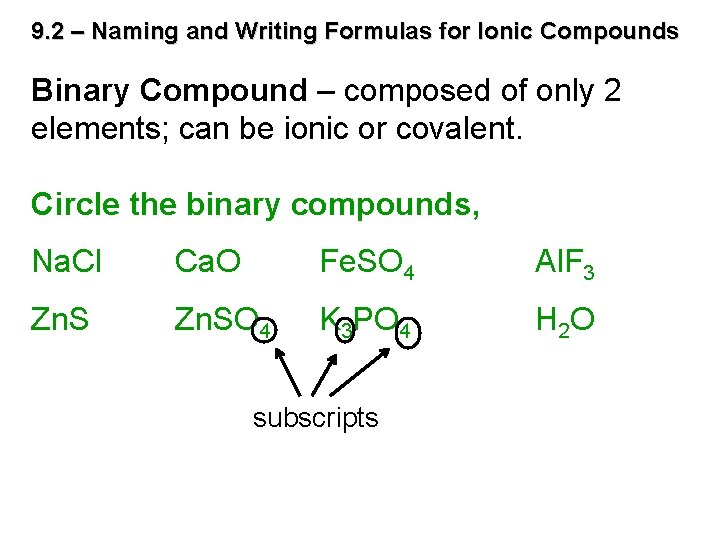
9. 2 – Naming and Writing Formulas for Ionic Compounds Binary Compound – composed of only 2 elements; can be ionic or covalent. Circle the binary compounds, Na. Cl Ca. O Fe. SO 4 Al. F 3 Zn. SO 4 K 3 PO 4 H 2 O subscripts

1. Writing formulas for ionic compounds Write symbols and charges for each ion in the compound; cation first, use crisscross method to balance the ion charges; reduce to lowest ratio

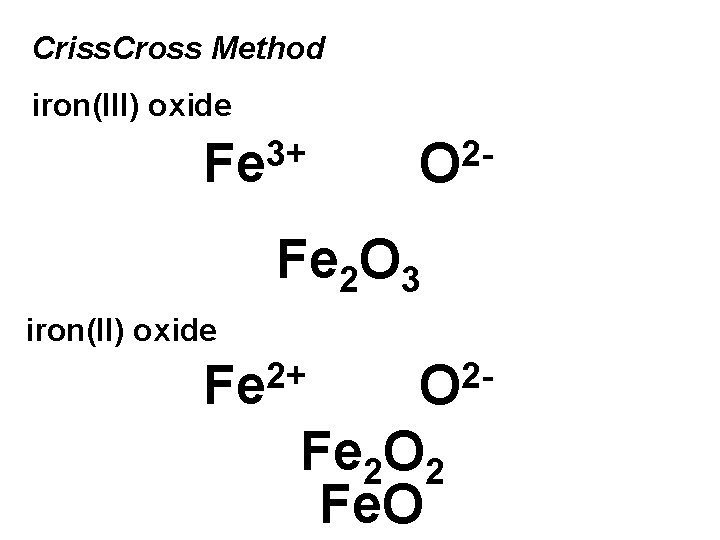
Criss. Cross Method iron(III) oxide 3+ Fe 2 O 3 iron(II) oxide 2+ Fe 2 O 2 Fe. O

Write formulas for these compounds, sodium chloride potassium hydroxide barium chlorate calcium fluoride copper(II) oxide ammonium phosphate

2. Naming ionic compounds Name cation first and then anion, use a roman numeral if metal cation can have more than one charge, end in –ide if binary.

Name these compounds, Na. Cl Al. F 3 Ca. O Fe. SO 4 Zn. S K 3 PO 4 Na. OH

ASSIGNMENT: Chapter 9 #57 -68 (p. 298)

9. 3 – Naming and Writing Formulas for Molecular Compounds Binary molecular compounds contain a prefix to show the number of atoms. Prefixes Used in Naming Binary Molecular Compounds Prefix Number mono- 1 di- 2 tri- 3 tetra- 4 penta- 5 hexa- 6 hepta- 7 octa- 8 nona- 9 deca- 10

Name these compounds, NCl 3 nitrogen trichloride BCl 3 boron trichloride N 2 H 4 dinitrogen tetrahydride NO 2 nitrogen dioxide N 2 O dinitrogen oxide SO 3 sulfur trioxide CO 2 carbon dioxide

Write formulas for these compounds, phosphorus pentachloride PCl 5 iodine heptafluoride IF 7 chlorine trifluoride Cl. F 3 carbon monoxide CO carbon disulfide CS 2 sulfur trioxide SO 3

ASSIGNMENT: Read 9. 3 Chapter 9 #69 -72; 80 -85 (p. 298)