Unit 4 CHEMICAL EQUILIBRIUM K EQ Equilibrium Reactions

Unit 4 - CHEMICAL EQUILIBRIUM - K EQ Equilibrium Reactions, Le Chatelier’s Principle, and Solving Problems in Equilibrium

PRINCIPLE OF EQUILIBRIUM Basic Concepts of Equilibrium: A “closed” system must be present (otherwise it will not truly reach equilibrium). If it isn’t, any chemicals can enter or leave and affect the amounts in the reaction. For any reaction, it is easier to explain or measure what is going on if it is a closed system (constant temperature, pressure, starting concentrations of chemicals) A system where there are constantly changes occurring, like different or opposing reactions (a forward and a reverse reaction) at an equal rate is called a dynamic equilibrium. e. g. A + B C+D

PRINCIPLE OF EQUILIBRIUM: (CONT’D) Any reaction can be written as its forward, or reverse reaction: CO 2 + H 2 O H 2 CO 3 Making H 2 CO 3 is the Forward direction H 2 CO 3 CO 2 + H 2 O Making CO 2 + H 2 O is now Forward Just remember that in the “forward” direction, we must go from left to right, and the reverse direction is going from right to left. CO 2 + H 2 O H 2 CO 3 Making CO 2 + H 2 O is the Reverse direction

PRINCIPLE OF EQUILIBRIUM: (CONT’D) When you have an equilibrium reaction, the reactions do not actually stop. Both the forward and the reverse reactions occur simultaneously. When equilibrium does actually occur, it is both reactions (forward and reverse) that occur at the same rate (so no net change in the concentrations/amounts of the chemicals involved in the reaction). Question: Do both reactions have to occur at exactly the same rate? The answer simply: It depends. It actually depends on the relative concentrations of the reactants and products (i. e. stoichiometry).

TYPES OF EQUILIBRIUM: There are several types of equilibrium: 1. Chemical Reaction Equilibrium This is where reactant(s) change into products and back (the reverse reaction) e. g. H 2 O + CO 2 H 2 CO 3 2. Phase Equilibrium This is where a chemical changes phases, or states, like from a liquid to a solid or a gas and back (the reverse) e. g. H 2 O(l) H 2 O(g) 3. Solubility Equilibrium This is where we have a solid dissolving into a liquid like water or an organic liquid and back (the reverse) e. g. Mg. O(s) Mg. O (aq)

CHEMICAL REACTIONS AT EQUILIBRIUM Reactants going to products, products going to reactants…at the same time.

CHEMICAL REACTIONS AT EQUILIBRIUM Description of Equilibrium Position of Equilibrium No Reaction (NR) <1% (mainly reactants) Reactants are favoured <50% Products are favoured >50% Quantitative (high yield) >99% (mostly product)

A LAW OF CHEMICAL EQUILIBRIUM A relationship between the concentrations of the reactants and products exists, and this can be given as a specific value. This ratio is fixed, at a particular temperature (i. e. it is constant as long as the temperature stays the same). For any balanced chemical reaction that is in equilibrium, we have a ratio (or constant). We call it Keq.

EQUILIBRIUM CONSTANT – KEQ (OR KC)

WRITING A LAW EXPRESSION: 2 2

WRITING A LAW EXPRESSION (CONT’D): 2 2

BACK TO THE EQUILIBRIUM CONSTANT

EQUILIBRIUM CONSTANT (CONT’D): Let us compare 3 different set ups for the same reaction (note that all conditions are the same, aside from the starting concentrations of products and/or reactants): Experiment [H 2] [I 2] [HI] 1 2. 000 mol/L 0 2 0 0 2. 000 mol/L 3 0 0. 010 mol/L 0. 350 mol/L Since this is a reversible reaction, if we start with only product (like in Experiment 2), we will actually form the reactants in the reaction shown on the previous slide.

EQUILIBRIUM CONSTANT (CONT’D): Making Sense of Keq using reaction data: Chemical Reaction Data (note: these are equilibrium concentrations for each chemical in the reaction) [H 2] [I 2] [HI] 0. 442 mol/L 3. 116 mol/L 0. 221 mol/L 1. 560 mol/L 0. 035 mol/L 0. 045 mol/L 0. 280 mol/L Note that we must have equilibrium concentrations for each chemical (not starting concentrations) in order to determine Keq. When we put each of these sets of values into the equilibrium law expression for Keq shown next:
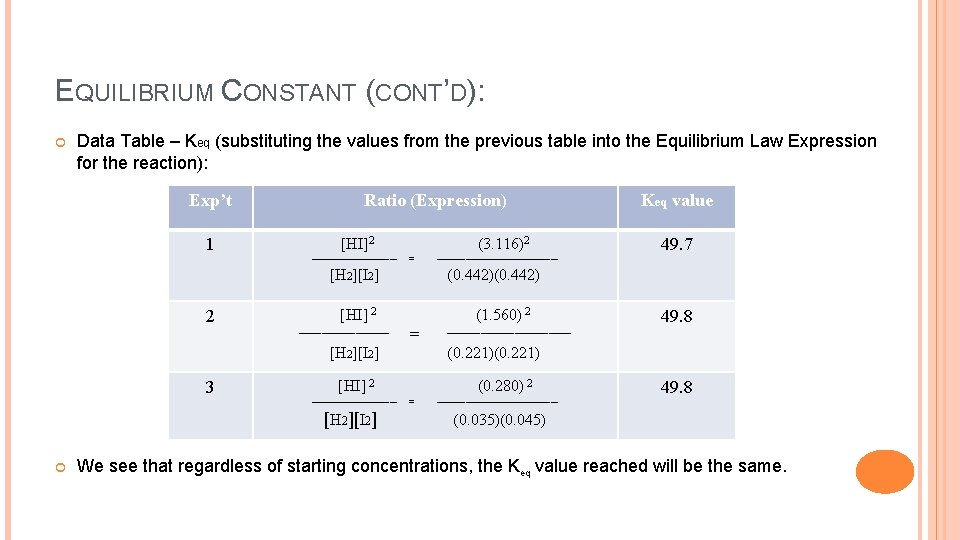
EQUILIBRIUM CONSTANT (CONT’D): Data Table – Keq (substituting the values from the previous table into the Equilibrium Law Expression for the reaction): Exp’t 1 Ratio (Expression) [HI]2 ────── (3. 116)2 = [H 2][I 2] 2 3 [HI] 2 ──── [H 2][I 2] 49. 7 (0. 442) = [HI] 2 ───────────────── Keq value = (1. 560) 2 ────── (0. 221) 49. 8 (0. 280) 2 49. 8 ───────── (0. 035)(0. 045) We see that regardless of starting concentrations, the Keq value reached will be the same.

EQUILIBRIUM CONSTANT (CONT’D): You can determine the Keq for the reaction, regardless of the direction we are interested in. So if we wish to have the Keq for the reverse reaction, we find this by writing the reciprocal for the Law Expression: e. g. [HI] [H 2][I 2] Keq = ───── 1/Keq = ────── [H 2][I 2] [HI] We can also do this with the Keq value: 1 Keq = 49. 8 1/Keq = ─── = 2. 01 x 10 -2 49. 8 And hence, we receive the Keq for the reverse reaction of what we started with. Therefore for: HI ↔ H 2 + I 2 Keq = 2. 01 x 10 -2

BIGGER Keq? WHAT DOES THIS MEAN? The value of the Keq does tell us something about the reaction: For example: Value of Keq Result of Reaction Keq >> 1 Reaction will go towards completion or concentration of products will be much greater than the reactants at equilibrium. Given the reaction: H 2 O(l) ↔ H 2(g) + O 2(g) Keq = 7. 3 x 10 -10 Since Keq is so small, the reaction will favour the reactant (much higher H 20 concentration than the products). Keq = 1 Keq << 1 Concentrations of reactants and products are about the same at equilibrium. Very small amount of products will form, reactant concentration much greater at equilibrium

LE CHATELIER’S PRINCIPLE The Balancing Game

LE CHATELIER’S PRINCIPLE We have studied in the past that different conditions in which a reaction occurs can play a role in how the reaction proceeds. Any equilibrium reaction (just like any other chemical reaction) is affected by conditions like: Temperature Pressure Presence of Catalysts Concentrations of the reactants and products.

1. CONCENTRATION EFFECT Generally, when the concentrations of either the reactants or products change, the system will “react”: A. With the addition of either to the system, the equilibrium will shift in order to adjust to the addition B. Make more product when reactant is added, or product is removed C. Change product back to reactant if reactant is removed or product(s) are added These are dictated by the Keq.

2. TEMPERATURE EFFECT When the other conditions change (like T), usually the rate of the chemical reactions change. This is contingent on whether energy is either produced or used in the reaction. The system will adjust itself accordingly. How? This is based on the type of reaction (thermodynamically), endothermic or exothermic. Exothermic – if you have an exothermic reaction, adding heat will cause the reaction to shift to the reactant side (heat is formed on the product side) Endothermic – if you have an endothermic reaction, adding heat will cause the reaction to shift to the product side (heat is required to form product)

3. PRESSURE EFFECT When volume changes (hence the pressure), only the reactions where gases are formed or are acting as reactants will experience any shift in the equilibrium. Since it is a closed system, increasing gas pressure (decreasing volume) will cause an increase in concentration of the gaseous chemicals causing the equilibrium shift. Gaseous Reactants – if there are only gases on the reactant side, increasing the pressure will cause a shift to the product side. Gaseous Products – if there are only gases on the product side, increasing the pressure will cause a shift to the reactant side.

4. CATALYSTS AND EQUILIBRIUM Remember that when a catalyst is added, only the activation energy for the reaction is lowered. Since the activation energy is lowered, the speed at which the reaction completes will be faster. There is no net change in the final concentrations for the reactants and for the products, so when equilibrium is reached, the concentrations are still the same as when there was no catalyst. The reaction equilibrium in the presence of catalyst only occurs much sooner than without.

EQUILIBRIUM PROBLEM-SOLVING Mathematical Treatment of Equilibrium Reactions

QUANTIFYING CHANGES IN EQUILIBRIUM SYSTEMS During any equilibrium reaction, we can measure the concentrations for each of the reactants and products. Knowing these, we can determine whether the reaction has reached its equilibrium. To see in which direction a reaction will proceed, we calculate a value called a Reaction Quotient (Q). In general: Looks familiar, doesn’t it?

QUANTIFYING (CONT’D)

INTERPRETING Q When you perform the calculation for a reaction, if the result is: Q = Keq Reaction is at equilibrium Q > Keq Reaction must shift towards the reactants (product to reactant ratio is too high) Q < Keq Reaction must shift towards the products (product to reactant ratio is too low)

SOLVING EQUILIBRIUM PROBLEMS ICE Table Method

Solving Equilibrium Reactions Question: When hydrogen reacts with fluorine, hydrogen fluoride is formed according to the equation: H 2(g) + F 2(g) 2 HF(g) The equilibrium constant is Keq = 1. 15 x 10 at STP. 2 Calculate the concentrations for all chemicals if 4. 00 moles each of H 2(g) and F 2(g) and 6. 00 moles of HF(g) are initially placed in a 2. 00 L reaction vessel.

SOLVING METHOD Step 1 – Find the Initial Concentrations for All Chemicals in the Reaction: [H 2] = [F 2] = 4. 00 mol = 2. 00 mol/L 2. 00 L [HF] = 6. 00 mol = 3. 00 mol/L 2. 00 L

Solving Equilibrium Reactions (cont’d): Step 2 – Determine direction of reaction (find Q) 2 Q = [HF] = (3. 00) = 2. 25 [H 2][F 2] (2. 00) 2 Keq = 1. 15 x 10 2 Since Q < Keq, reaction is not at equilibrium, it must shift to the right (forming more product) in order to achieve equilibrium.
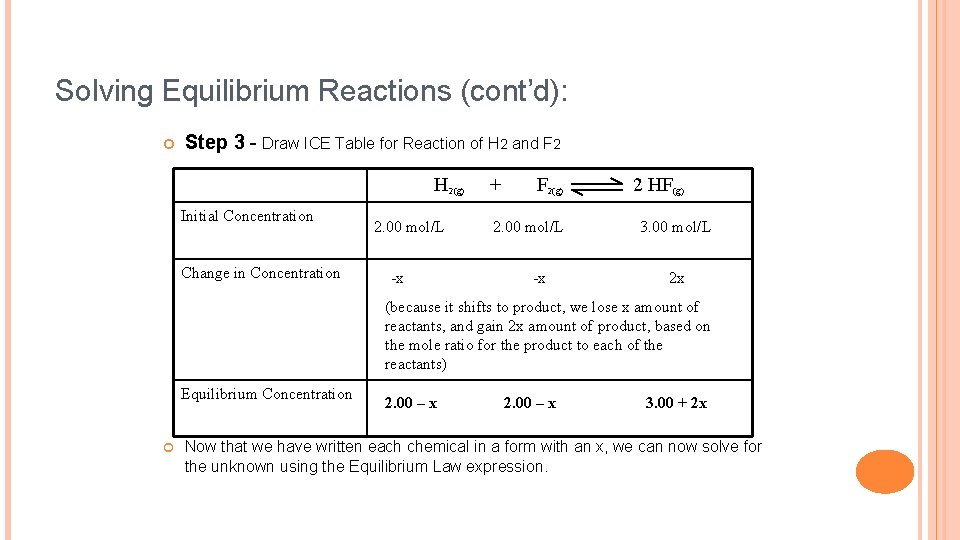
Solving Equilibrium Reactions (cont’d): Step 3 - Draw ICE Table for Reaction of H 2 and F 2 H 2(g) Initial Concentration Change in Concentration 2. 00 mol/L -x + F 2(g) 2. 00 mol/L -x 2 HF(g) 3. 00 mol/L 2 x (because it shifts to product, we lose x amount of reactants, and gain 2 x amount of product, based on the mole ratio for the product to each of the reactants) Equilibrium Concentration 2. 00 – x 3. 00 + 2 x Now that we have written each chemical in a form with an x, we can now solve for the unknown using the Equilibrium Law expression.
![Solving Equilibrium Reactions (cont’d): Step 4 – Once we have achieved equilibrium: 2 [HF] Solving Equilibrium Reactions (cont’d): Step 4 – Once we have achieved equilibrium: 2 [HF]](http://slidetodoc.com/presentation_image/2deebcd3f22915e60316c2318021b969/image-33.jpg)
Solving Equilibrium Reactions (cont’d): Step 4 – Once we have achieved equilibrium: 2 [HF] = K eq [H 2][F 2] Substitute in the expressions from the ICE Table so that we can solve for x: 2 (3. 00 + 2 x) = 1. 15 x 10 2 (2. 00 – x)(2. 00 – x) (3. 00 + 2 x) = 10. 724 (2. 00 – x) 3. 00 + 2 x = 10. 724(2. 00 – x) 3. 00 + 2 x = 21. 448 – 10. 724 x 12. 724 x = 18. 448 x = 1. 450 mol/L

Solving Equilibrium Reactions (cont’d): Step 5 – Substitute into Equilibrium Concentration expressions from Step 3 to find actual concentration values: [H 2] = 2. 00 mol/L – x = 2. 00 mol/L – 1. 450 mol/L = 0. 550 mol/L [F 2] = 2. 00 mol/L – x = 2. 00 mol/L – 1. 450 mol/L = 0. 550 mol/L [HF] = 3. 00 mol/L + 2 x Q = [HF]2 [H 2][F 2] = (5. 900)2 (0. 550)2 = 1. 15 x 102 = 3. 00 mol/L + 2(1. 450 mol/L) = 3. 00 mol/L + 2. 900 mol/L = 5. 900 mol/L You can check these values with the Equilibrium Law expression and you will find that substituting these values will give you the Keq for that reaction.

TRY THIS QUESTION: Reaction: I 2(g) + Br 2(g) IBr(g) Reaction is in a 2. 0 L container Keq = 1. 2 x 102 Starting with 4. 00 moles each of I 2 and Br 2

EQUILIBRIUM PROBLEMS: (CONT’D) Solving equilibrium problems are pretty easy once you get the idea, if you can always use the square root to solve it. If, however, you have problems which make solving using the root difficult, for example: Cu 2+(aq) + 4 Cl-(aq) So we would have the expression: 2 -] [Cu. Cl 4 Keq = ________ [Cu 2+][Cl-]4 Cu. Cl 42 -(aq) Regardless of where the unknown (x) is, we will not be able to square root it.

What can we do? Get angry? NO! As fun as that may be, we have some options.

EQUILIBRIUM PROBLEMS When You Do Not Have Perfect Squares

AN EQUILIBRIUM WITHOUT PERFECT SQUARES Solving Equilibrium problems becomes more difficult when we do not have perfect squares in both the numerator and denominator of the Equilibrium Law expression. There a couple of tricks that we can use in order to help us simplify the process. The following methods are methods that will be covered: The “Hundred Rule” (simplify method) Using the Quadratic Equation

THE HUNDRED RULE The Hundred Rule goes like this: If the Keq value is very small and the starting concentration(s) of the chemicals is large enough that dividing the [chemical] by the K value gives a number 100 or larger, we can assume that the [chemical]initial – x will still be ~[chemical]initial In that case, we can ignore the –x part of the equation and attempt to solve without it. Example: Carbon dioxide reacts to form carbon monoxide and oxygen gas according to: 2 CO 2(g) 2 CO(g) + O 2(g)

HUNDRED RULE Starting with 0. 250 moles of CO 2(g) with a Keq of 6. 40 x 10 -7. The volume for the container is 1. 00 L. Solve the above equilibrium using the standard ICE table method. Keq = [CO]2[O 2] [CO 2]2 Initial Change Equilibrium 2 CO 2 2 CO + O 2 0. 250 0. 00 -2 x +x 0. 25 - 2 x 2 x x

HUNDRED RULE: (CONT’D) Substituting in: Keq = (2 x)2(x) = (0. 25 – 2 x)2 4 x 3 (0. 25 – 2 x)2 We will have a cubic equation which is difficult to solve. We attempt then to assume to ignore – x. x = 2. 15 x 10 -3
![HUNDRED RULE: (CONT’D) Therefore, we have a [CO 2] at equilibrium of 0. 250 HUNDRED RULE: (CONT’D) Therefore, we have a [CO 2] at equilibrium of 0. 250](http://slidetodoc.com/presentation_image/2deebcd3f22915e60316c2318021b969/image-43.jpg)
HUNDRED RULE: (CONT’D) Therefore, we have a [CO 2] at equilibrium of 0. 250 – 2 (2. 15 x 10 -3) = 0. 247 ≈ 0. 250 depending on rounding. We can try the same calculation, but with the assumption of ignoring the x for the CO 2.

EQUILIBRIUM PROBLEMS: (CONT’D) We now have one tool to handle non-perfect square problems, however, what if we do not get a test value of over 100 (Hundred Rule)? We now have to go to our weapon of “last resort”, using the Quadratic Equation.

SOLVING BY USING THE QUADRATIC EQUATION A seemingly last resort method…

SOLVING USING THE QUADRATIC EQUATION There are some equilibrium problems where the equilibrium equation (law expression) does not allow a simple square root in order to remove the powers (eg. x 2) In order to solve these problems (assuming you cannot use a cube root for both the numerator and denominator), is to use the Quadratic Equation.

SOLVING AN EQUILIBRIUM PROBLEM USING THE QUADRATIC EQUATION Example: 0. 50 moles of N 2 O 4 is placed into a 1. 0 L closed container. The reaction proceeds at 150 o. C. What are the concentrations of both N 2 O 4 and NO 2 (the product) after equilibrium? Keq is 4. 50 at 150 o. C. The balanced equation: [N 2 O 4] = 0. 50 moles/L N 2 O 4(g) 2 NO 2(g) [NO 2] = 0. 00 moles/L Q = [NO 2]2 = 0. 00 [N 2 O 4]
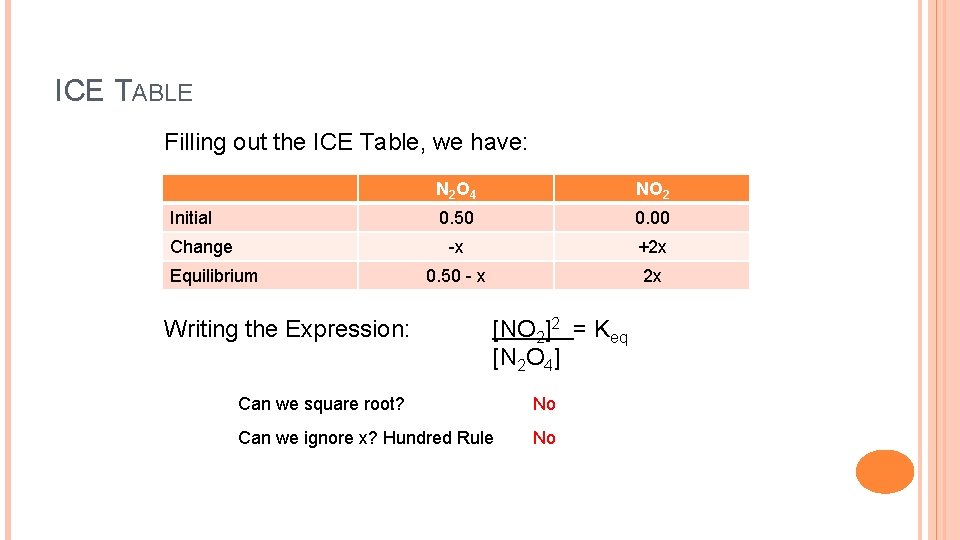
ICE TABLE Filling out the ICE Table, we have: Initial Change Equilibrium Writing the Expression: N 2 O 4 NO 2 0. 50 0. 00 -x +2 x 0. 50 - x 2 x [NO 2]2 = Keq [N 2 O 4] Can we square root? No Can we ignore x? Hundred Rule No

USING THE QUADRATIC Since we cannot use what we have employed before, we have to use the Quadratic. (2 x)2 = 4. 50 (0. 50 – x) 4 x 2 = 4. 50(0. 50 – x) 4 x 2 = 2. 25 – 4. 50 x 4 x 2 + 4. 50 x – 2. 25 = 0 Now substitute into the Quadratic Equation

SOLUTION(S) After solving and receiving 2 possible answers: X = -1. 50 X = 0. 375 We cannot use the – 1. 50 value, since that would result in a negative concentration for NO 2 Substitute the 0. 375 into equilibrium concentration expressions: [NO 2] = 2(0. 375) = 0. 75 moles/L [N 2 O 4] = 0. 50 – 0. 375 = 0. 125 moles/L Test these values to compare with Keq

EQUILIBRIUM – WHAT NEXT? We have now gone through solving various equilibrium systems, using a number of techniques: Square Root Hundred Rule (simplify by ignoring) Quadratic Equation We have now completed working with chemical reaction systems. We will now look at solubility.

SOLUBILITY An Application for Equilibrium

SOLUTIONS AND SOLUBILITY Recall that a solution is a homogeneous mixture of a chemical (the solute) dissolved into a solvent (mainly it is water, but can be other liquids too). The term, solubility, refers to the ability of a chemical to dissolve into a solvent. Different chemicals, of course, have different abilities to go into a solution. As it turns out, we can apply a version of the ideas that we have learned previously in equilibrium systems here.

PREDICTING THE SOLUBILITY OF CHEMICALS The ability for a chemical to be dissolved may seem obvious. We can put a chemical into a solvent and see if it dissolves, but that would be a waste of a chemical. How can we predict whether a chemical can/will dissolve? To do this, we need to study what determines whether a chemical will be soluble. Our experiences with chemicals has helped us make predictions, by making the Solubility Rules.

THE SOLUBILITY RULES: Studies on many chemicals have determined that certain chemicals have specific abilities to dissolve, or not to dissolve. These rules are based on Ionic compounds. This can be seen based largely on the group that an element is found in, or what type of chemical it is. What follows is a list of Rules for Solubility of Chemicals: 1. All Group 1 Compounds are Soluble 2. All Nitrates are Soluble 3. All common Halides (F, Cl, Br, I) are soluble (with exceptions)

SOLUBILITY RULES: (CONT’D) 4. Most Sulfates are soluble (exceptions here) 5. All Chlorates are soluble 6. All Perchlorates are soluble 7. All Acetates are soluble 8. All Phosphates are insoluble (except Gr. I and NH 4+) 9. All Carbonates are insoluble (except Gr. I and NH 4+) 10. All Hydroxides are insoluble (exceptions here) 11. All Oxides are insoluble (except Gr. I) 12. All Oxalates are insoluble (except Gr. I and NH 4+) 13. All Sulfides are insoluble (exceptions here)

PREDICTING SOLUBILITY: PRACTICE Using the Table provided for you, predict the solubility for the following: Na 3 PO 4 Mg(OH)2 Fe(Cl. O 4)3 KCH 3 COO K 2 S Soluble Insoluble Soluble Zn. S Insoluble Ba. SO 4 Insoluble

KSP – THE SOLUBILITY PRODUCT CONSTANT Measuring the Solubility of a Chemical

EQUILIBRIUM – SOLUBILITY PRODUCT Similar to Chemical Equilibrium systems, we have an equilibrium for Solubility. Recall: Mg. O(s) Mg. O(aq) We can write this as an expression like Keq: Keq = [Mg. O(aq)] [Mg. O(s)] However, something that is solid cannot be represented as a concentration, since a concentration implies it is in solution, so we write it differently for K in solubility.

KSP – SOLUBILITY EQUILIBRIUM CONSTANT So instead of writing it as an “equilibrium” constant, solubility expressions are written as such: KKspeq = [Mg. O(aq)] Where the reactant side (the solid) is ignored in the expression. We write it as the chemical dissolving since it is a measure of solubility, not formation of the solid. We now use a constant, Ksp (instead of Keq) and it is called the Solubility Product Constant.

WRITING KSP EXPRESSIONS: Similar to equilibrium expressions, we write them according to the balanced equation. We also change the reaction a bit by writing the products as the ions in the chemical, not as a complete compound (since the compound will form ions in solution). Example: Write the solubility expression for the following. This would give us: Ba. SO 4(s) Ba 2+(aq) + SO 42 -(aq) Ksp = [Ba 2+][SO 42 -]

WRITING KSP EXPRESSIONS: (CONT’D) Example: Write the following equation as an expression for solubility: Pb. Cl 2(s) Pb 2+(aq) + 2 Cl-(aq) Note that there will be 2 Cl- ions formed, so we must have the coefficient of 2 shown for that ion. We can now write the Ksp expression: Ksp = [Pb 2+][Cl-]2

PRACTICE: For the following solubility equations, balance and write the proper Ksp expression: Sr. CO 3(s) Sr 2+(aq) + CO 32 -(aq) Ba. Cl 2(s) Ba 2+(aq) + Cl-(aq) Ca 3(PO 4)2(s) 33 Ca 2+(aq) + PO 4 (aq) 2 Ksp = [Sr 2+][CO 32 -] 2 Ksp = [Ba 2+][Cl-]2 Ksp = [Ca 2+]3[PO 43 -]2 Assignment: Solubility worksheet (Solubility Rules and Solubility Expressions)

KSP VALUES &SOLUBILITY PROBLEMS Applying Ksp

KSP VALUES AND SOLVING PROBLEMS Now that we have a mathematical relationship for the ions that are formed from the dissolution of a chemical, we can now investigate and apply this Ksp. First of all, this Ksp value. How do we find it? All chemicals that are only soluble to a small degree have a known Ksp value. For example (all values are at 25 o. C ): Ba. CO 3 has a Ksp of 1. 6 x 10 -9 Hg 2 Cl 2 has a Ksp of 1. 1 x 10 -18 Looking at these values, it means that barium carbonate is slightly more soluble than mercury chloride. A higher negative means less soluble.

USING KSP: There a number of problems that we can address: Finding a Ksp given the soluble ion concentrations. Solving for an ion concentration given the Ksp Predicting the formation of a Precipitate Determining the effect of a “common ion” We will look at the first two ideas, which are simple algebraic problems. 1. Calculating Ksp. This is a simple write an expression and solve for Ksp when given the ion concentration values.

1. CALCULATING KSP: The steps are simple for performing this: 1. Balance the ionic solubility equation 2. Write the Ksp equilibrium expression 3. Substitute the provided values for concentration Example: The concentrations for both Ag+ and Br- is 7. 07 x 10 -7 M. What is the Ksp value for the dissolution of Ag. Br? Solution – the ionic equation would be written as: Ag. Br(s) Ag+(aq) + Br-(aq) So the solubility expression will be: Ksp = [Ag+][Br-]

CALCULATING KSP: (CONT’D) We can now substitute into the equation the data we have: Ksp = (7. 07 x 10 -7 M) Ksp = 4. 998 x 10 -13 or Ksp = 5. 00 x 10 -13 Try this one: What is the Ksp for Fe(OH)3 if the concentration for Fe 3+ is 1. 39 x 10 -10 M and OH- is 4. 16 x 10 -10 M? Step 1. Write ionic equation: Fe(OH)3(s) Fe 3+(aq) + 3 OH-(aq)
![CALCULATING KSP: (CONT’D) Step 2. Write solubility expression. Ksp = [Fe 3+][OH-]3 Step 3. CALCULATING KSP: (CONT’D) Step 2. Write solubility expression. Ksp = [Fe 3+][OH-]3 Step 3.](http://slidetodoc.com/presentation_image/2deebcd3f22915e60316c2318021b969/image-69.jpg)
CALCULATING KSP: (CONT’D) Step 2. Write solubility expression. Ksp = [Fe 3+][OH-]3 Step 3. Substitute in the given values. Ksp = (1. 39 x 10 -10 M)(4. 16 x 10 -10)3 Ksp = 1. 00 x 10 -38 Calculating a Ksp gave us a chance to write and work with the Ksp expression. We now will investigate by using a Ksp, what would the soluble ion concentrations would be for a particular salt.

CALCULATING KSP: (CONT’D) Other things to watch for in problems involving finding a Ksp: When you are given a “dissolved” chemical concentration, in order to find the correct dissolved ion concentrations, you must remember the formula and how many of each ion there is. Example: If the dissolved concentration for Ca. CO 3 is 6. 86 x 10 -5 M, what is the Ksp? The equation is: Ca. CO 3(s) So the Ksp expression is: Ca 2+(aq) + CO 32 -(aq) Ksp = [Ca 2+][CO 32 -]
![CALCULATING KSP: (CONT’D) How do we find [Ca 2+] and [CO 32 -]? We CALCULATING KSP: (CONT’D) How do we find [Ca 2+] and [CO 32 -]? We](http://slidetodoc.com/presentation_image/2deebcd3f22915e60316c2318021b969/image-71.jpg)
CALCULATING KSP: (CONT’D) How do we find [Ca 2+] and [CO 32 -]? We have the dissolved [Ca. CO 3], that will tell us both of these concentrations. Since the only source for these ions is this chemical, the [Ca 2+] and [CO 32 -] should be the same as the dissolved [Ca. CO 3]. Hence: [Ca 2+] = 6. 86 x 10 -5 M, and [CO 32 -] = 6. 86 x 10 -5 M So, plugging in: Ksp = [Ca 2+][CO 32 -] = (6. 86 x 10 -5 M) = 4. 7 x 10 -9

SOLUBLE ION CONCENTRATIONS What the Ksp can tell us… or help us solve.

SOLUBLE ION CONCENTRATIONS: The Ksp for a slightly soluble salts does not just tell us the relative abilities for different salts to dissolve. We can use the Ksp to determine the actual concentration of the dissolved ions that formed from the slightly soluble salt. How we do this requires us to: A. Have a Ksp value for the slightly soluble salt. B. Write the Ksp expression for that salt, and using the chemical formula for that salt, and C. Determine the relative concentrations for each soluble component in that salt.

SOLUBLE ION CONCENTRATIONS: (CONT’D) Example: Let’s find the soluble ion concentrations for the slightly soluble salt, Pb. I 2. Step 1 – Find the Ksp for Pb. I 2 Ksp = 8. 3 x 10 -9 Step 2 – Write the ionic equation and Ksp expression for the dissolution of Pb. I 2(s) Pb 2+(aq) + 2 I-(aq) Ksp = [Pb 2+][I-]2 Step 3 – Since the concentrations for both ions are linked (they come from the same source), we can equate them. Let us use x to represent the concentration of the dissolved chemical. Let [Pb. I 2(aq)] = x, then [Pb 2+(aq)] = x; [I-(aq)] = 2 x

SOLUBLE ION CONCENTRATIONS: (CONT’D)

PRACTICE:

PRECIPITATION REACTIONS Predicting Precipitation and the Effect of Common Ions

USING KSP TO PREDICT PRECIPITATES The value of Ksp becomes clear when we deal with the formation of precipitates. Precipitate – a chemical which will appear out of a solution as a solid. We can use the Ksp to determine whether we will form a precipitate or not.

PREDICTING PRECIPITATES Remember that the value of Ksp can help us determine the soluble ion concentration for any of the ions that form the compound. Those ion concentrations also dictate at what concentrations can the ions be (at most) before a precipitate will form. If you understand what all that meant, you should see that we can now Ksp = [A]a[B]b predict precipitate formation as long as we know the ion concentration values.

PREDICTING PRECIPITATES: (CONT’D) The deciding factor is Ksp, so we do something like Q for Equilibrium Systems, do a test Ksp: If the concentrations give a test Ksp value less than the reported Ksp, a precipitate will NOT form. If the concentrations give a test Ksp value larger than the reported Ksp, a precipitate WILL form. Precipitate Formation Problems: Mixing Solutions 1. Determining what the concentrations for the ions are, and 2. Finally calculating the test Ksp to see if a precipitate forms.

REVIEW: DILUTIONS In order to properly calculate the test Ksp value, we need to review dilution calculations: Dilutions: (from Solutions Unit in Grade 11) The important thing to understand is that if you mix two different solutions together, the strengths of the chemicals in each solution will change (get weaker). - This depends on the volume of the original solution and the final volume for the mixture of solutions. - We use the formula: M 1 V 1 = M 2 V 2 - We just solve for M 2, given that we start with M 1 (starting solution concentration), V 1 (the volume of that solution), and , V 2 (the sum of the two solution volumes). -

PREDICTING PRECIPITATES: (CONT’D) Example: You have a 250. 0 m. L solution of 1. 75 x 10 -3 M of Sr 2+ and a 250. 0 m. L solution of 1. 50 x 10 -3 M of SO 42 -. Will a precipitate form if you mix them together? The Ksp is 7. 6 x 10 -7 for Sr. SO 4. Step 1 – Determine the final concentrations of both ions. M 1 V 1 = M 2 V 2 For Sr 2+: (1. 75 x 10 -3 M)(0. 250 L) = M 2(0. 5000 L) M 2 = (1. 75 x 10 -3 M)(0. 250 L) (0. 5000 L) = 8. 75 x 10 -4 M

PREDICTING PRECIPITATES: (CONT’D) For SO 42 -: (1. 50 x 10 -3 M)(0. 250 L) = M 2(0. 5000 L) M 2 = (1. 50 x 10 -3 M)(0. 250 L) (0. 5000 L) = 7. 50 x 10 -4 M Step 2 – Write the Ksp expression, and check to see if the test value is higher or lower than the known Ksp. Write the ionic equation and Ksp expression. Sr. SO 4(s) Sr 2+(aq) + SO 42 -(aq) Ksp = [Sr 2+][SO 42 -] Test: Ksp = (8. 75 x 10 -4 M)(7. 50 x 10 -4 M) = 6. 56 x 10 -7 Since the test Ksp is lower or smaller than the Ksp, a precipitate will not form.

PRACTICE: You have 350. 0 m. L of a 0. 035 M solution of Pb(NO 3)2 and you mix it with 250. 0 m. L of a 0. 080 M solution of KCl. Will a precipitate of Pb. Cl 2 form? The Ksp for Pb. Cl 2 is 1. 7 x 10 -5. Step 1 – Final Ion Concentrations M 1 V 1 = M 2 V 2 For Pb 2+: (0. 035 M)(0. 3500 L) = M 2(0. 6000 L) M 2 = (0. 035 M)(0. 3500 L) (0. 6000 L) = 0. 02042 M For Cl-: (0. 080 M)(0. 2500 L) = M 2(0. 6000 L) M 2 = (0. 080 M)(0. 2500 L) = 0. 03333 M (0. 6000 L)

PRACTICE: (CONT’D) Step 2 – Determine test Ksp. Write the ionic equation for Pb. Cl 2 dissociation and the Ksp expression. Pb. Cl 2(s) Pb 2+(aq) + 2 Cl-(aq) Ksp = [Pb 2+][Cl-]2 Test: Ksp = (0. 02042 M)(0. 03333 M)2 = 2. 27 x 10 -5 Looking at our Ksp value: 1. 7 x 10 -5 Our test value is higher, so a precipitate will indeed form.

THE COMMON-ION EFFECT What happens to ion concentrations when we mix in another solution containing one of the same ions?

COMMON-ION EFFECT What happens when we add to a solution containing a slightly soluble salt, another solution that has a very soluble salt, but one of the ions is identical to one of the ions found in the insoluble salt? Consider the following: Pb. Cl 2 is a slightly soluble salt (Ksp = 1. 7 x 10 -5). We can now determine the soluble ion concentrations for the ions, Pb 2+ and Cl-. What happens to those ion concentrations if we add a solution of Na. Cl (a very soluble salt)? Two approaches: Solution contains both salts (i. e. same volume) Different solutions are mixed together (dilution effect)

COMMON-ION EFFECT: (CONT’D) The first possibility gives us a simpler situation to resolve: Example: We have a solution containing Pb. CO 3 (Ksp = 1. 5 x 10 -15). What is the soluble ion concentration for both Pb 2+ and CO 32 -? If in the same solution volume, we also have 0. 500 M Na 2 CO 3, what will the Pb 2+ concentration now be? Step 1 – Soluble Ion Concentrations For Pb. CO 3, the ionic equation and the Ksp expression is: Pb. CO 3(s) Pb 2+(aq) + CO 32 -(aq) Ksp = [Pb 2+][CO 32 -] Let x = [Pb 2+] = [CO 32 -]

COMMON-ION EFFECT: (CONT’D)
![COMMON-ION EFFECT: (CONT’D) [Pb 2+] = 1. 5 x 10 -15 (0. 500 M) COMMON-ION EFFECT: (CONT’D) [Pb 2+] = 1. 5 x 10 -15 (0. 500 M)](http://slidetodoc.com/presentation_image/2deebcd3f22915e60316c2318021b969/image-90.jpg)
COMMON-ION EFFECT: (CONT’D) [Pb 2+] = 1. 5 x 10 -15 (0. 500 M) [Pb 2+] = 3. 0 x 10 -15 M So we can see that if a common-ion is added into a mixture of soluble ions, we can precipitate more. This is of practical use when attempting to recover metals of economic importance. What about when we have two solutions that we are mixing together? How would we solve that problem?

COMMON-ION EFFECT Mixing two solutions

COMMON-ION EFFECT: (CONT’D) How do we approach a problem where we have two solutions being mixed together, and a common is involved? Example: We have a 500. 0 m. L solution containing Mg. CO 3. We also have a 50. 0 m. L solution of 0. 500 M Na 2 CO 3. (Ksp for Mg. CO 3 is 1. 0 x 10 -15) A. What is the soluble ion concentration for Mg 2+ and CO 32 - in the Mg. CO 3 solution? B. What is the Mg 2+ concentration after you mix the Na 2 CO 3 solution together with the Mg. CO 3 solution? A. Soluble Ion Concentrations: Mg. CO 3(s) Mg 2+(aq) + CO 32 -(aq) Ksp = [Mg 2+][CO 32 -]

COMMON-ION EFFECT: (CONT’D)

COMMON-ION EFFECT: (CONT’D) We have the Ksp, all we now have to figure out is the concentration for the CO 32 -. In order to determine what you should use, we need to look first at the soluble [CO 32 -] value to consider if we need to include it. Generally, if the value is large relative to the final concentration of the added ion, we will have to include it, otherwise, we can ignore it. Dilution of Addition: M 1 V 1 = M 2 V 2 (0. 500 M)(0. 050 L) = M 2(0. 550 L) M 2 = (0. 500 M)(0. 050 L) (0. 550 L) = 4. 55 x 10 -2 M

COMMON-ION EFFECT: (CONT’D) Since the concentration of the diluted addition is much higher than the soluble ion concentration for that same ion in the first part: 3. 16 x 10 -8 M << 4. 55 x 10 -2 M We can ignore it in our next step. We now can substitute our current value for [CO 32 -], and use our Ksp value to solve for [Mg 2+]: Ksp = [Mg 2+][CO 32 -] 1. 00 x 10 -15 = [Mg 2+](4. 55 x 10 -2 M) [Mg 2+] = 1. 00 x 10 -15 4. 55 x 10 -2 M [Mg 2+] = 2. 20 x 10 -14 M

COMMON-ION EFFECT: (CONT’D) We can see now that the concentration of the Mg 2+ is lowered by a large amount: From 3. 16 x 10 -8 M down to 2. 20 x 10 -14 M. This can also be used to recover as much of an ion as possible (precipitate it out by adding more of the counter-ion).

UNIT REVIEW: 1. Equilibrium Systems and Types of Equilibrium Open and Closed systems Chemical, Phase, and Solubility Forward and Reverse Reactions 2. Equilibrium Expression (Keq) and Q 3. Le Chatelier’s Principle Temperature, Pressure, Catalysts 4. Equilibrium Problems ICE Table Square Root, Hundred Rule, and Quadratic Equation 5. Solubility Equilibrium

UNIT REVIEW: (CONT’D) 6. Rules of Solubility Ionic compounds, Rule Table 7. Solubility Expression (Ksp) 8. Soluble Ion Concentrations Finding concentrations for each ion (solve for x) 9. Predicting Precipitates Does a precipitate form, given ion concentrations 10. Common-Ion Effect How does the addition of one ion affect the soluble concentration for the other?
- Slides: 98