Unit 4 AcidBase Theory Lesson 3 BrnstedLowry Theory

Unit 4: Acid-Base Theory Lesson 3: Brønsted-Lowry Theory

Brønsted-Lowry Theory • This theory expands on Arrhenius theory and gives new definitions for acids and bases: • An acid is a substance that loses a proton to another substance. • A base is a substance that gains a proton from another substance.
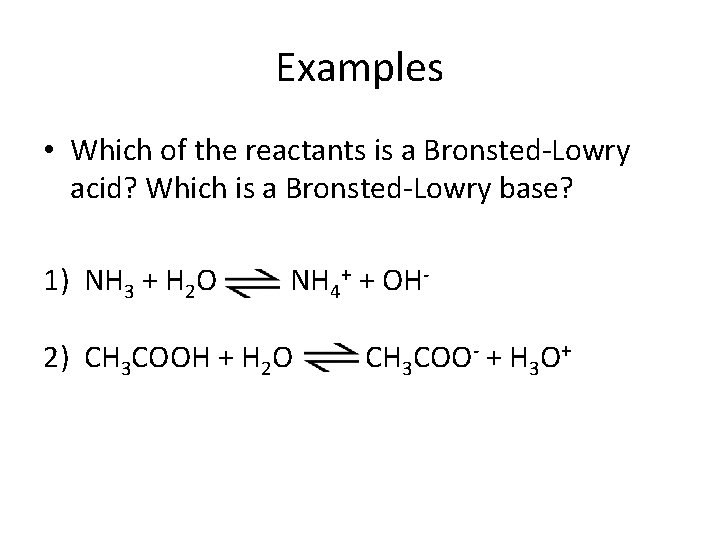
Examples • Which of the reactants is a Bronsted-Lowry acid? Which is a Bronsted-Lowry base? 1) NH 3 + H 2 O NH 4+ + OH- 2) CH 3 COOH + H 2 O CH 3 COO- + H 3 O+

Brain Break: Spot 5 Differences

Hold up, wait a minute. . . • In our earlier examples, H 2 O was an acid in one reaction, and a base in the other! • Substances that can act as a Bronsted-Lowry acid OR base are said to be amphiprotic.

Amphiprotic Substances • Aside from H 2 O, a substance will be amphiprotic if: a) it possesses a negative charge, AND b) it has an easily removable hydrogen. • Examples include H 2 PO 4 - and HS-

Demo: The Many Faces of Aluminum • Predict: What will happen as Na. OH is added to the solution of [Al(H 2 O)6]3+? • Observe: What changes occur? • Explain: As OH- is added, the [Al(H 2 O)6]3+ acts as an acid and loses a proton, then another, and so on! It has seven possible forms, all of which are aqueous EXCEPT the neutral [Al(H 2 O)3(OH)3], which appears as a white precipitate.

Acid-Base Equilibrium • Bronsted-Lowry acid-base reactions are at equilibrium, so they proceed in both the forward and reverse directions. • Thus, there is an acid and a base on both sides of the equation!
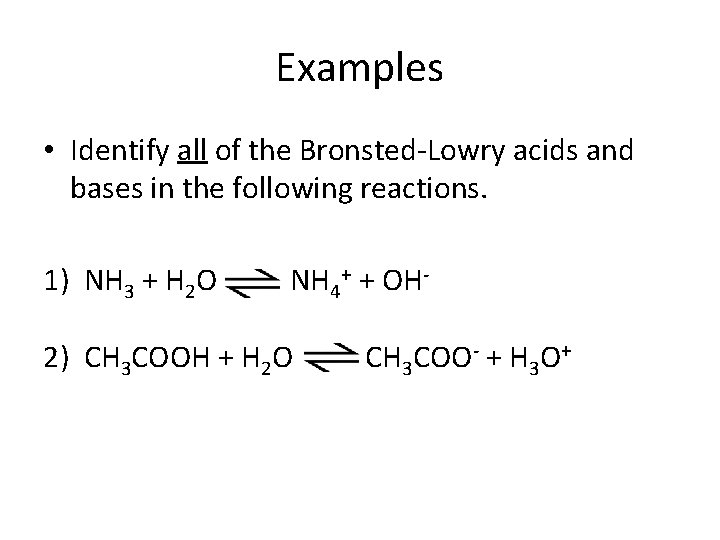
Examples • Identify all of the Bronsted-Lowry acids and bases in the following reactions. 1) NH 3 + H 2 O NH 4+ + OH- 2) CH 3 COOH + H 2 O CH 3 COO- + H 3 O+

Practice: • Pg. 117 #11 • Pg. 119 #13 and 14
- Slides: 10