UNIT 3 PERIODIC TRENDS ELECTRON CONFIGURATIONS AND BONDING

UNIT 3 PERIODIC TRENDS, ELECTRON CONFIGURATIONS, AND BONDING

Periodic Table Trends Periodic Law “When arranged by increasing atomic number, the chemical elements display a regular and repeating pattern of chemical and physical properties. ” Atoms with similar properties appear in groups or families (vertical columns) on the periodic table. all have the same number of valence (outer shell) electrons, which governs their chemical behavior.

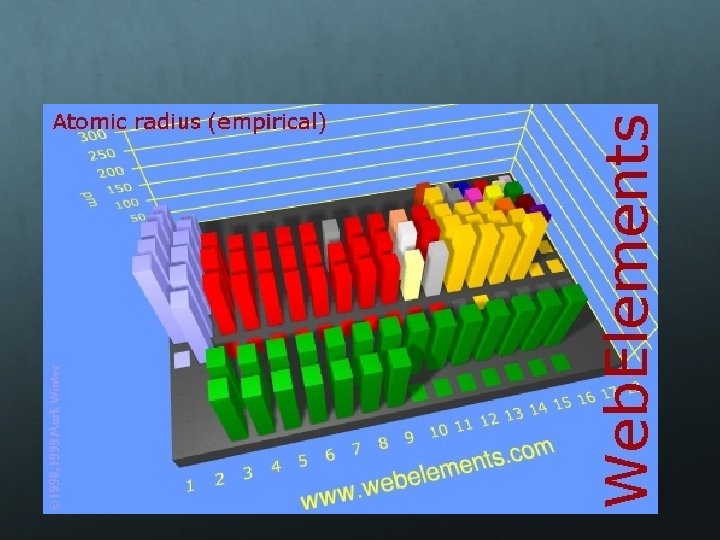
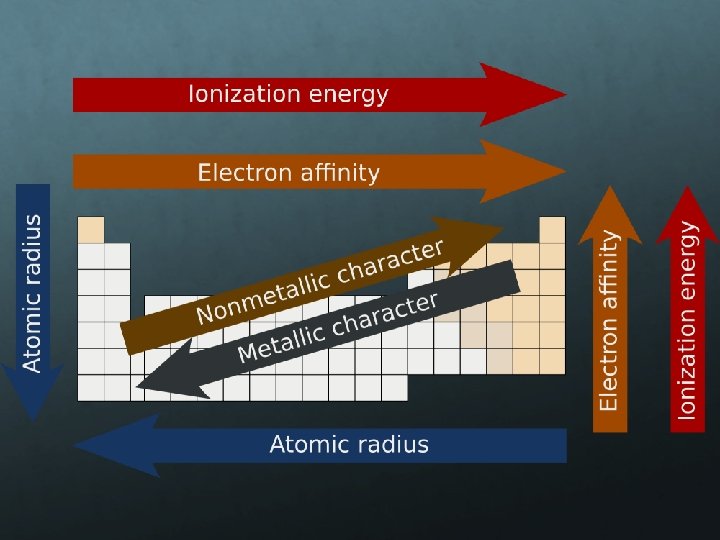
Atomic Radius is the distance from the center of the nucleus to the “edge” of the electron cloud. Since a cloud’s edge is difficult to define, scientists use covalent radius, or half the distance between the nuclei of 2 bonded atoms. Atomic radii are usually measured in picometers (pm) or angstroms (Å). An angstrom is 1 x 10 -10 m. Ex. Two Br atoms bonded together are 2. 86 angstroms apart. So, the radius of each atom is 1. 43 Å. 2. 86 Å 1. 43 Å

The trend for atomic radius smaller at the top to larger at the bottom. add an entirely new energy level to the electron cloud, making the atoms larger with each step. The trend across a horizontal period is less obvious. Each step from left to right adds a proton and an electron (and 1 or 2 neutrons) and electrons are added to existing energy levels. The effect is that the more positive nucleus has a greater pull on the electron cloud. The nucleus is more positive and the electron cloud is more negative. The increased attraction pulls the cloud in, making atoms smaller as we move from left to right across a period.

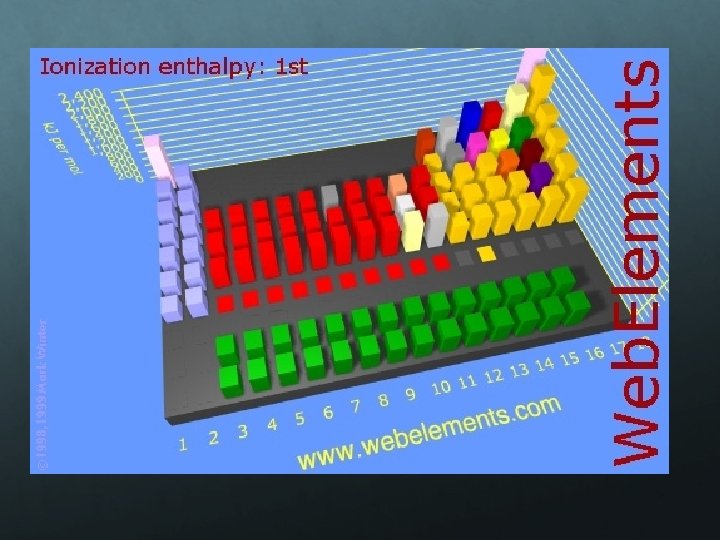
Ionization Energy If an electron is given enough energy to overcome the forces holding it in the cloud, it can leave the atom completely. The atom has been “ionized” or charged. The number of protons and electrons is no longer equal. The energy required to remove an electron from an atom is ionization energy. (measured in kilojoules, k. J) The larger the atom is, the easier its electrons are to remove. Ionization energy and atomic radius are inversely proportional.

Electron Affinity Electron affinity is the energy change that occurs when an atom gains an electron (also measured in k. J). (how well atoms attract electrons) Electron affinity is usually exothermic, but not always. Electron affinity is exothermic if there is an empty or partially empty orbital for an electron to occupy. If there are no empty spaces, a new orbital or energy level must be created, making the process endothermic.

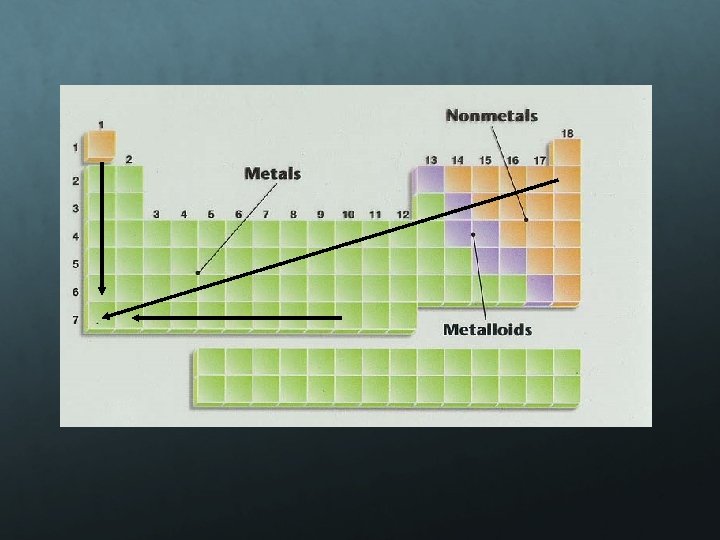
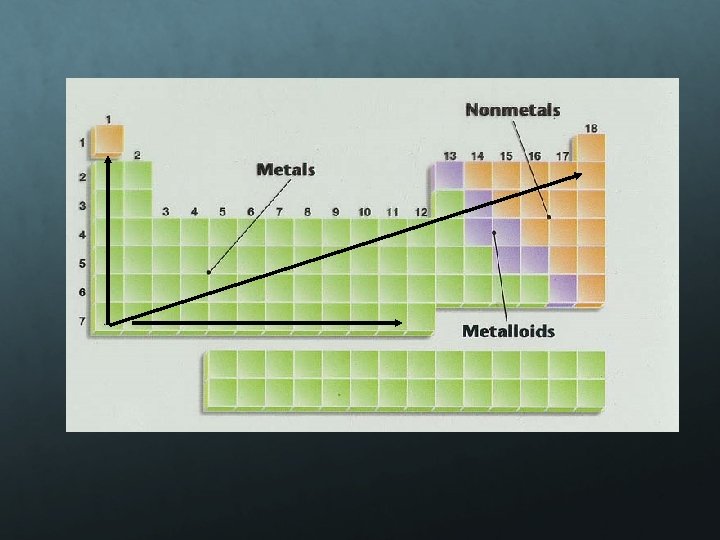
Metals: Metals like to lose valence electrons to form cations to have a fully stable octet. They absorb energy to lose electrons. The electron affinity of metals is lower than that of nonmetals. Nonmetals: Nonmetals like to gain electrons to form anions to have a fully stable octet. They release energy to gain electrons to form an anion; thus, electron affinity of nonmetals is higher than that of metals.
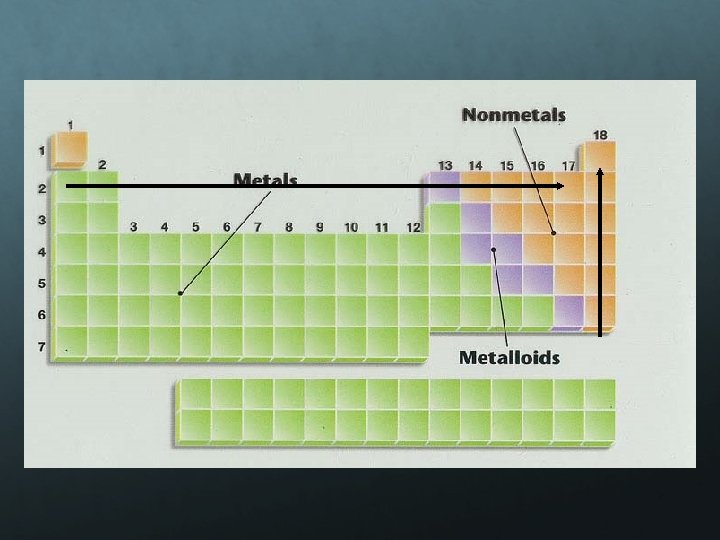

Electronegativity is a measure of an atom’s attraction for another atom’s electrons. It is an arbitrary scale that ranges from 0 to 4. The units of electronegativity are Paulings. Generally, metals are electron givers and have low electronegativities. Nonmetals are electron takers and have high electronegativities. What about the noble gases?
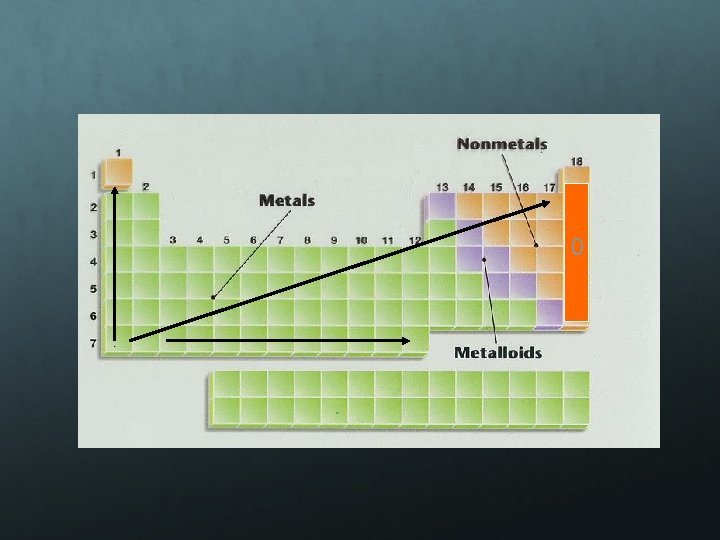
0

Periodic Trends Rap

Electron Configuration Quantum Mechanical Model (electron cloud) Energy is quantized. It comes in chunks. A quanta is the amount of energy needed to move from one energy level to another. Since the energy of an atom is never “in between” there must be a quantum leap in energy. Schrödinger derived an equation that described the energy and position of the electrons in an atom

Atomic Orbitals Within each energy level the complex math of Schrödinger's equation describes several shapes. These are called atomic orbitals Regions where there is a high probability of finding an electron
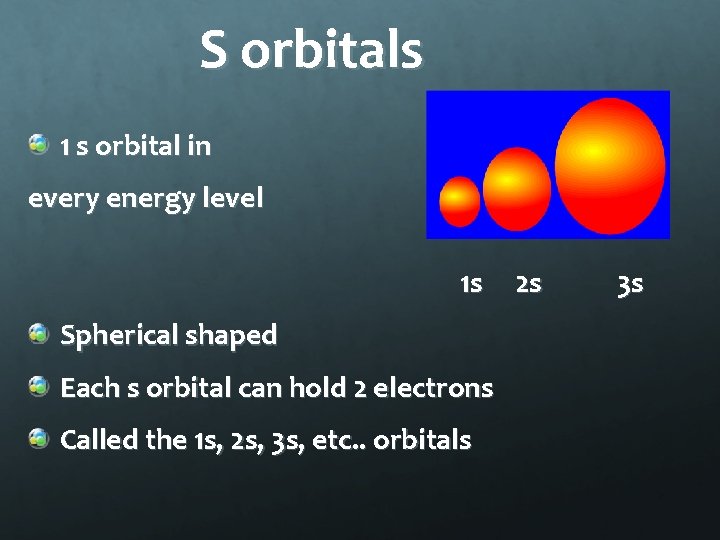
S orbitals 1 s orbital in every energy level 1 s Spherical shaped Each s orbital can hold 2 electrons Called the 1 s, 2 s, 3 s, etc. . orbitals 2 s 3 s
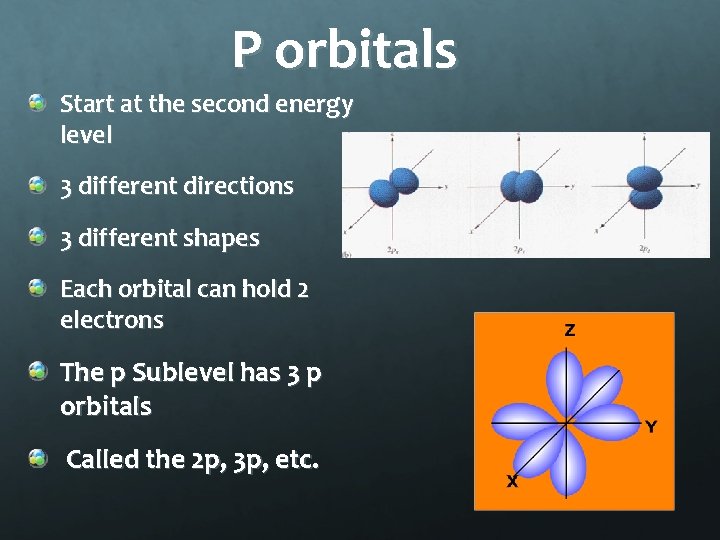
P orbitals Start at the second energy level 3 different directions 3 different shapes Each orbital can hold 2 electrons The p Sublevel has 3 p orbitals Called the 2 p, 3 p, etc.
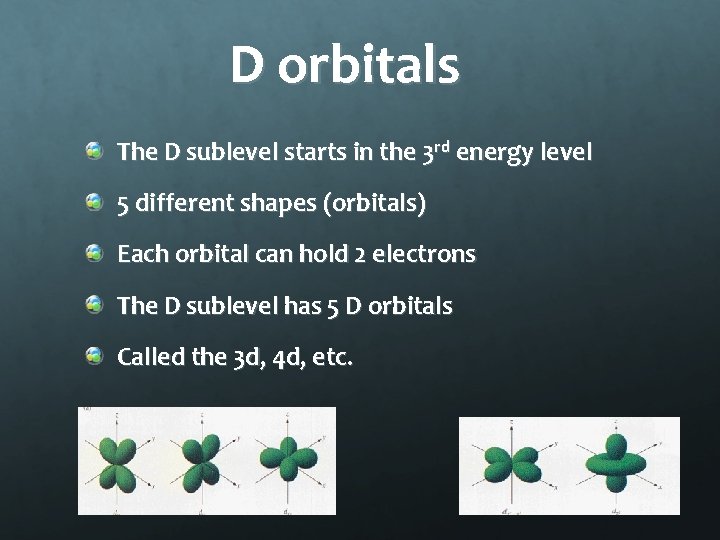
D orbitals The D sublevel starts in the 3 rd energy level 5 different shapes (orbitals) Each orbital can hold 2 electrons The D sublevel has 5 D orbitals Called the 3 d, 4 d, etc.

F orbitals The F sublevel starts in the fourth energy level The F sublevel has seven different shapes (orbitals) 2 electrons per orbital The F sublevel has 7 F orbitals ONLY 4 F and 5 F
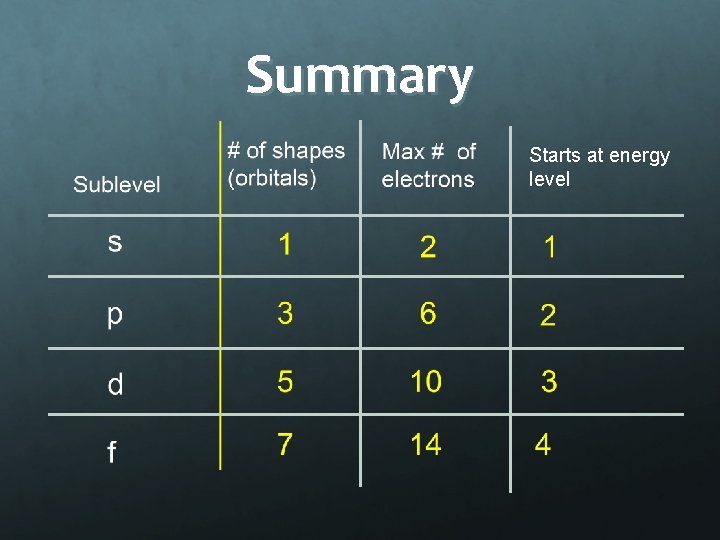
Summary Starts at energy level

Atomic Orbitals
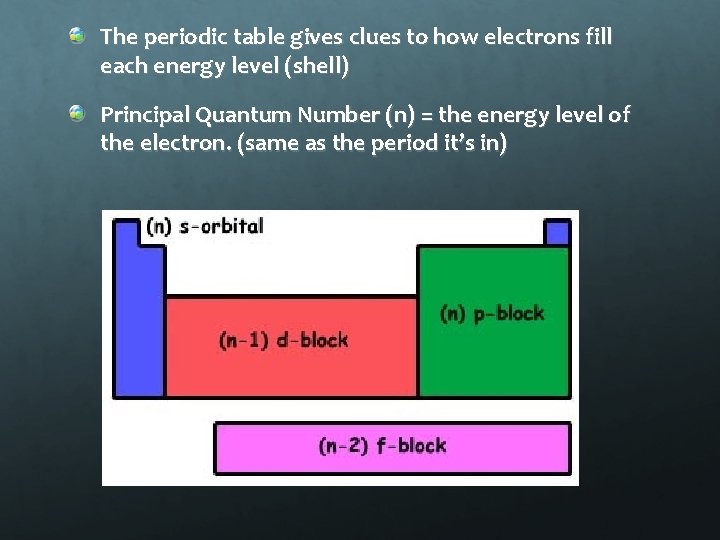
The periodic table gives clues to how electrons fill each energy level (shell) Principal Quantum Number (n) = the energy level of the electron. (same as the period it’s in)
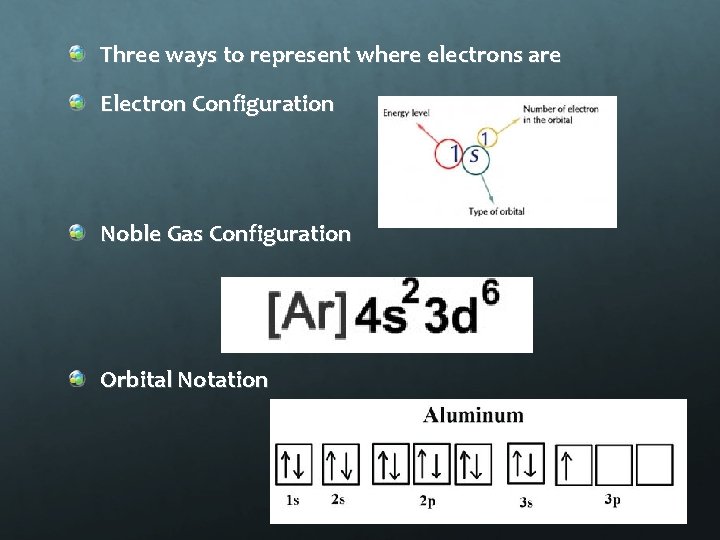
Three ways to represent where electrons are Electron Configuration Noble Gas Configuration Orbital Notation

Orbital Filling Rules!!!! Aufbau principle- electrons enter the lowest energy first. This causes difficulties because of the overlap of orbitals of different energies. Pauli Exclusion Principle- at most 2 electrons per orbital - different spins Hund’s Rule- When electrons occupy orbitals of equal energy they don’t pair up until they have to.



Stable Electron Configurations All elements on the periodic table (except for Noble Gases) have incomplete outer energy levels Valence electrons- electrons in outer energy level of atom Elements will gain, lose, or share electrons to get full outer levels (octet rule) Eight electrons = STABLE!!! Electron dot diagrams help to visualize valence electrons Symbol represents nucleus and inner electrons Dots represent valence electrons Group # = valence electrons

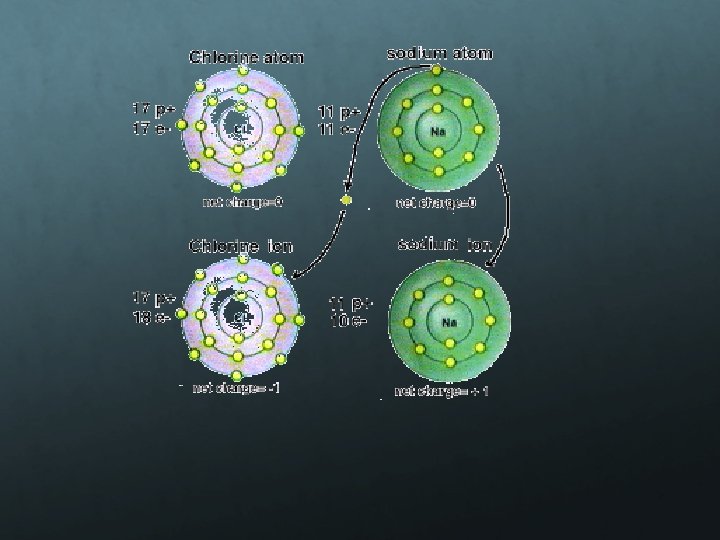
Ions Charged atoms where the number of protons and electrons is not equal Charge indicates how many electrons are added or subtracted Negative charge- ADD electrons Positive charge- SUBTRACT electrons Example Sodium ion Na atomic number 11 = 11 electrons Na+ subtract one electron = 10 electrons


Drawing Electron Dot Diagrams Determine number of valence electrons from periodic table Draw the symbol for the element Place dots around the symbol, one per side, until all valence electrons are accounted for Example- Aluminum with 3 valence electrons Al


A Chemical Bond Forces that hold groups of atoms together and make them function as a unit. A bond will form if the energy of the pairing is lower than that of the separate atoms. Some elements have stronger attractions to e- when bonded- helps to predict ELECTRONEGATIVITY (EN) Relative attraction an atom has for shared electrons in a covalent bond Unit- paulings Arbitrary number used for comparison purposes F is 4. 0, Cs/Fr 0. 7 Copyright © Cengage Learning. All rights reserved 36

Increase from left to right in a period Decrease from top to bottom in a group Nonmetals higher than metals Metals on left side and nonmetals on right side most reactive (alkali metals and halogens) Electrons attracted to the higher EN element Using EN to predict bonds Ionic Bonds Metal + nonmetal Covalent Bonds (nonmetal + nonmetal) Polar EN is different Nonpolar EN is same value

Arrange the following bonds from most to least polar: a) N–F O–F C–F b) C–F N –O Si–F c) Cl–Cl B–Cl S–Cl a) C–F, N–F, O–F b) Si–F, C–F, N–O c) B–Cl, S–Cl, Cl–Cl

Types of Chemical Bonds Ionic Bonds Some elements achieve stable configurations by transferring electrons Example- sodium and chlorine Sodium 1 valence electron Chlorine 7 valence electrons Both want to be stable Sodium will lose the one electron, and chlorine will gain that electron, forming IONS (atoms that have gained or lost electrons) Na+ Copyright © Cengage Learning. All rights reserved Cl 39

Charge on ion represented by + or – sign Positive ion- cation Negative ion- anion (use suffix –ide) Na+Cl- is sodium chloride (Na. Cl) Groups 1, 2, and 3 will lose electrons Groups 5, 6, and 7 will gain electrons Group 4 will go either way

Ionic compounds- compounds that contain ionic bonds Chemical formula- shows ratios of ions contained in the bond Na+Cl- one to one Na. Cl Mg 2+Cl- one to two Mg. Cl 2 Crystal Lattices Each ionic compound makes specific shape based on arrangement Crystal- solid whose particles are arranged in a lattice structure (Na. Cl- cubes, ruby- hexagonal) Properties of ionic compounds High melting point, boiling point Poor conductor when solid, good when molten/dissolved Crystal structure- shatters when hit

Covalent Bonds Nonmetals have high ionization energy Don’t usually form ions-share electrons to get to stable energy level Covalent bond-chemical bond in which two atoms share a pair of valence electrons May share one (single bond), two (double bond), or three pairs (triple bond) 43

Chemical Bonds Form molecules Neutral group of atoms that are joined together by one or more covalent bonds May exist as diatomic molecules Made of 2 atoms of same element

Chemical Bonds May form single or multiple bonds Subscript tells how many of each element are present

Chemical Bonds When atoms share electrons, they rarely share equally One element will “attract” electrons more than the others Polar Covalent Bond- a covalent bond in which electrons are not shared equally Atom with greater attraction gets a partial negative charge (δ), lesser attraction partial positive charge (δ+)

Naming Compounds Binary Compounds Composed of two elements Ionic and covalent compounds included Binary Ionic Compounds Metal—nonmetal Binary Covalent Compounds Nonmetal—nonmetal Copyright © Cengage Learning. All rights reserved 47

Stereochemistry- VSEPR Theory All molecules have 3 D shape Stereochemistry- study of shapes of molecules VSEPR theory Valence Shell Electron Pair Repulsion Helps to understand predict molecuar geometry (from Lewis Dot diagrams) Developed by Gillespie and Nyholm in 1956 -57 Rules based on the idea that the arrangement in space of the covalent bonds formed by an atom depends on the arrangement of valence ee- try to push each other far away while still bonding to central atom

Restricted VSEPR rules Valence e- pairs (both shared and lone) arrange themselves around the central atom in a molecule in such a way as to minimize repulsion (as far away from each other as possible) When predicting molecular geometry, double and triple bonds act like single bonds Lone pairs of e- occupy more space than bonding e-

Steps to draw VSEPR molecules Draw Lewis Diagram Count the number of bonding and lone pairs surrounding the central atom Multiple bonds count as one pair Shape molecule in order to minimize repulsion

Stereochemistry- VSEPR Theory All molecules have 3 D shape Stereochemistry- study of shapes of molecules VSEPR theory Valence Shell Electron Pair Repulsion Helps to understand predict molecuar geometry (from Lewis Dot diagrams) Developed by Gillespie and Nyholm in 1956 -57 Rules based on the idea that the arrangement in space of the covalent bonds formed by an atom depends on the arrangement of valence ee- try to push each other far away while still bonding to central atom e- pair repulsion

Restricted VSEPR rules Valence e- pairs (both shared and lone) arrange themselves around the central atom in a molecule in such a way as to minimize repulsion (as far away from each other as possible) When predicting molecular geometry, double and triple bonds act like single bonds Lone pairs of e- occupy more space than bonding e-

Steps to draw VSEPR molecules Draw Lewis Diagram Determine the central atom (lowest EN) Count the number of bonding and lone pairs surrounding the central atom Multiple bonds count as one pair Shape molecule in order to minimize repulsion Find on VSEPR chart
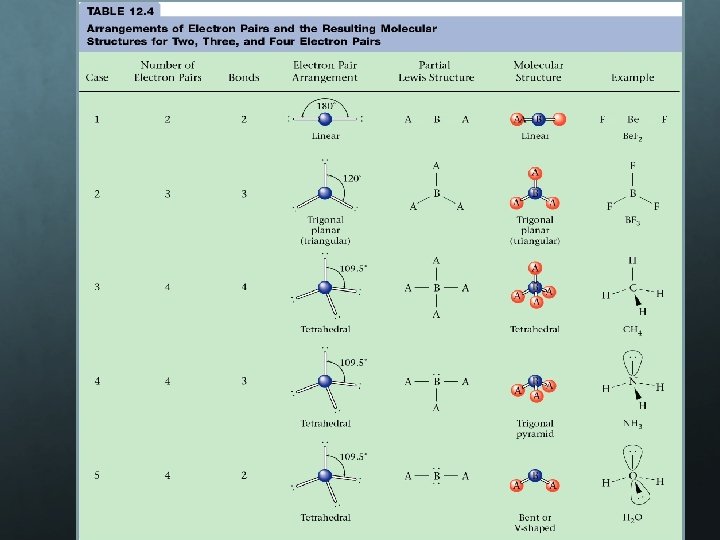

EXAMPLES Water, H 2 O 2 bond pairs 2 lone pairs The molecular geometry is BENT.

As a Lewis Dot diagram: Has two electron pairs - two sets of double bonds – two bonding pairs Creates a LINEAR shape according to VESPR
- Slides: 56