Unit 3 Periodic Table Day 1 Properties of

Unit 3: Periodic Table Day 1: Properties of Elements Lab

Warm Up: Set Up Unit 3 PAGE ? : Write “LT 3 Scale” (you will glue in LT Scale next week) TOP OF PAGE ? and ? WRITE: • “UNIT 3 WARM UPS” • Then, move “sticky note” from unit 2 to this page TOP OF PAGE ? and ? WRITE: • “UNIT 3 TOOLBOX” TIME: 4 minutes WHEN DONE: Make sure everyone at your table has the titles and then give Ms. Myriah a “thumbs up”

Agenda Ø Lab: Properties of Elements Ø Lab Analysis Ø If time: Periodic Table Notes

Long Term Learning Target I can use the periodic table to predict the relative reactivity of metals in groups 1, 2 and 3.
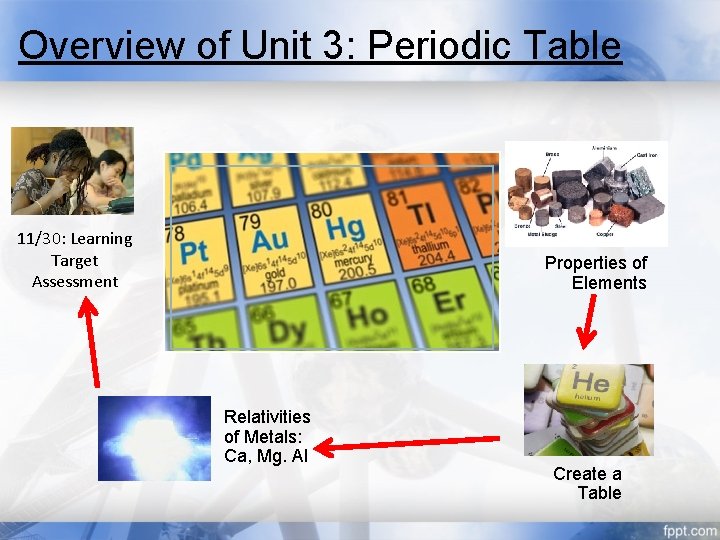
Overview of Unit 3: Periodic Table 11/30: Learning Target Assessment Properties of Elements Relativities of Metals: Ca, Mg. Al Create a Table

Properties of Elements Lab READ and ANNOTATE: Purpose, Introduction, and Safety Pre-Caution, HIGHLIGHT and ANNOTATE: AT LEAST Three things that you need to know to do the lab TIME: 8 MINUTES WHEN DONE: Make Prediction about Pre-Lab Preparation

Properties of Elements Lab READ: Procedure YOU MUST WEAR: Goggles and Lab Coat WRITE: All observation on data table (back page) TIME: 32 MINUTES WHEN DONE: Begin Post-Lab Questions

Properties of Elements Lab Post-Lab ANSWER: Post-lab questions using your data TIME: 24 MINUTES WHEN DONE: TURN in POSTLAB QUESTIONS and GLUE DATA TABLE into next available left-hand page

Periodic Table Notes TURN TO: Periodic Table Notes • Add to Notes from Video TIME: 1 MINUTE WHEN DONE: Think about what you remember about the last video
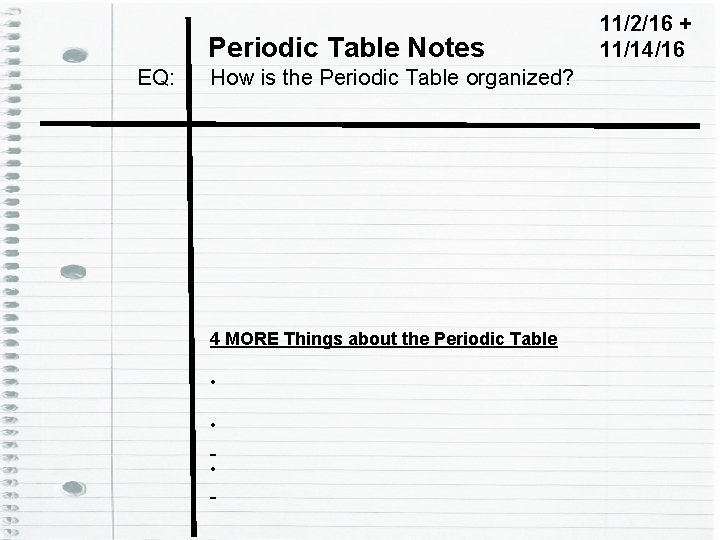
Periodic Table Notes EQ: How is the Periodic Table organized? 4 MORE Things about the Periodic Table • • • 11/2/16 + 11/14/16

Color Code Periodic Table FOLLOW ALONG: On Document Camera COLOR CODE: Groups on Periodic Table THEN: Fold in half and GLUE Periodic Table into Toolbox

Toolbox Entry GLUE or TAPE: Toolbox Entry Table into Toolbox USING: Your knowledge, table partners, and lab analysis FILL IN THE REST of the table TIME: 8 minutes WHEN DONE: Say “hello” in as many languages as possible to your table partners
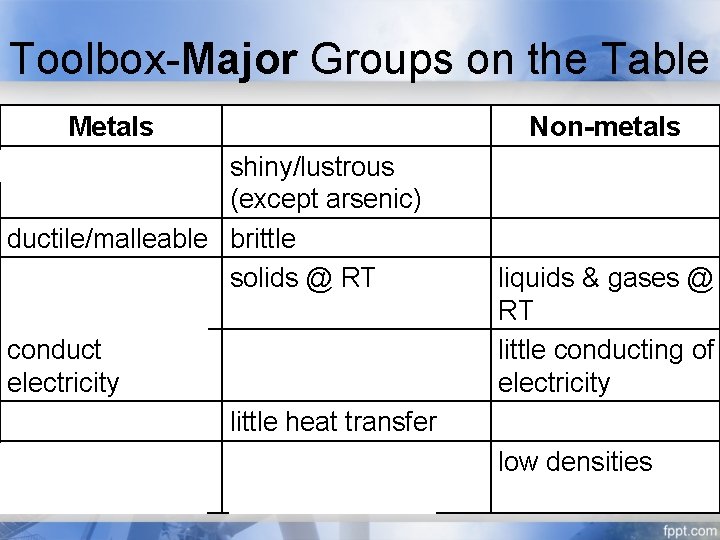
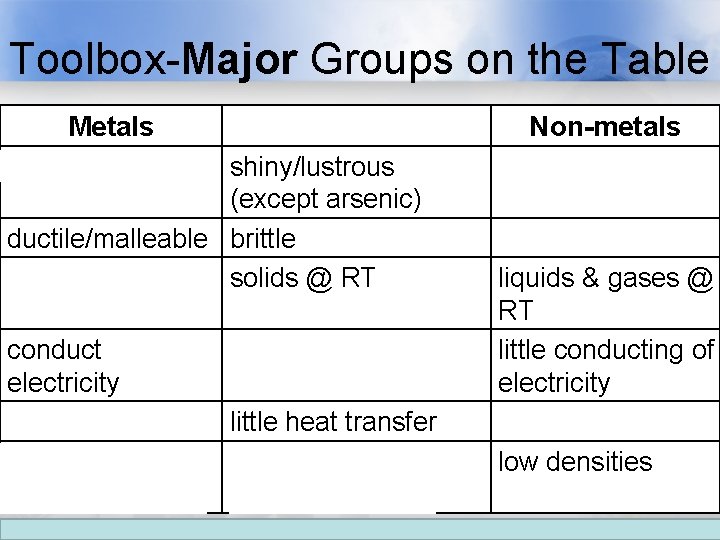
Toolbox-Major Groups on the Table Metals shiny/lustrous Metalloids shiny/lustrous (except arsenic) ductile/malleable brittle solids @ RT (except mercury) conduct electricity conduct heat little heat transfer high densities intermediate densities Non-metals flat/matte brittle if solid liquids & gases @ RT little conducting of electricity little heat transfer low densities
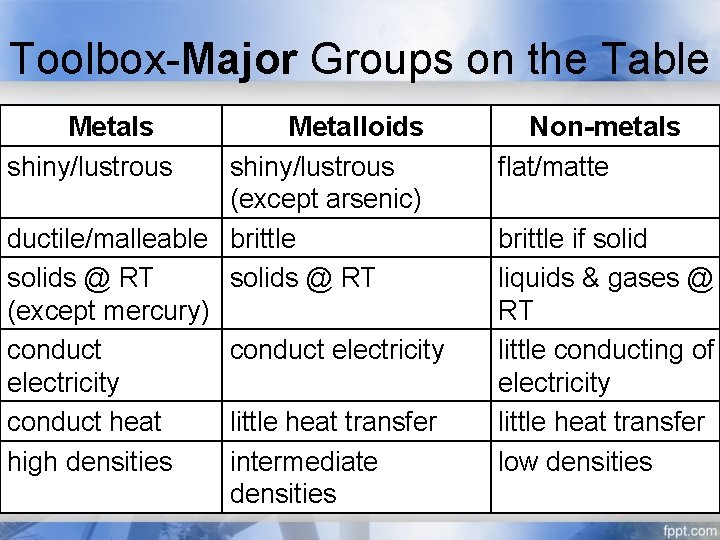
Toolbox-Major Groups on the Table Metals shiny/lustrous Metalloids shiny/lustrous (except arsenic) ductile/malleable brittle solids @ RT (except mercury) conduct electricity conduct heat little heat transfer high densities intermediate densities Non-metals flat/matte brittle if solid liquids & gases @ RT little conducting of electricity little heat transfer low densities
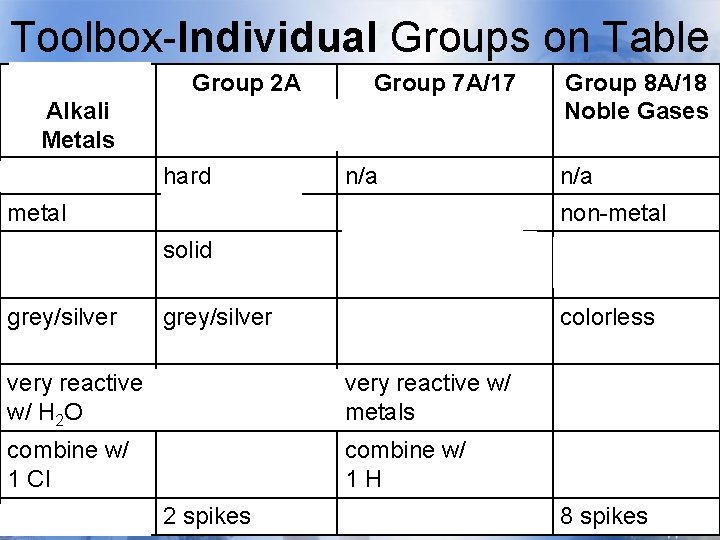
Toolbox-Individual Groups on Table Group 1 A Alkali Metals soft Group 2 A Group 7 A/17 Alkaline Earth Halogens Metals hard n/a Group 8 A/18 Noble Gases n/a metal non-metal solid gas, liquid, & solid gases grey/silver yellow/brown/blue colorless very reactive react with H 2 O w/ H 2 O very reactive w/ metals unreactive combine w/ 1 Cl combine w/ 2 Cl combine w/ 1 H n/a 1 spike 2 spikes 7 spikes 8 spikes
Toolbox-Individual Groups on Group 1 A Group 2 A Table Group 7 A/17 Group 8 A/18 Alkali Metals soft Alkaline Earth Halogens Metals hard n/a Noble Gases n/a metal non-metal solid gas, liquid, & solid gases grey/silver yellow/brown/blue colorless very reactive react with H 2 O w/ H 2 O very reactive w/ metals unreactive combine w/ 1 Cl combine w/ 2 Cl combine w/ 1 H n/a 1 spike 2 spikes 7 spikes 8 spikes
Toolbox-Major Groups on the Table Metals shiny/lustrous Metalloids shiny/lustrous (except arsenic) ductile/malleable brittle solids @ RT (except mercury) conduct electricity conduct heat little heat transfer high densities intermediate densities Non-metals flat/matte brittle if solid liquids & gases @ RT little conducting of electricity little heat transfer low densities
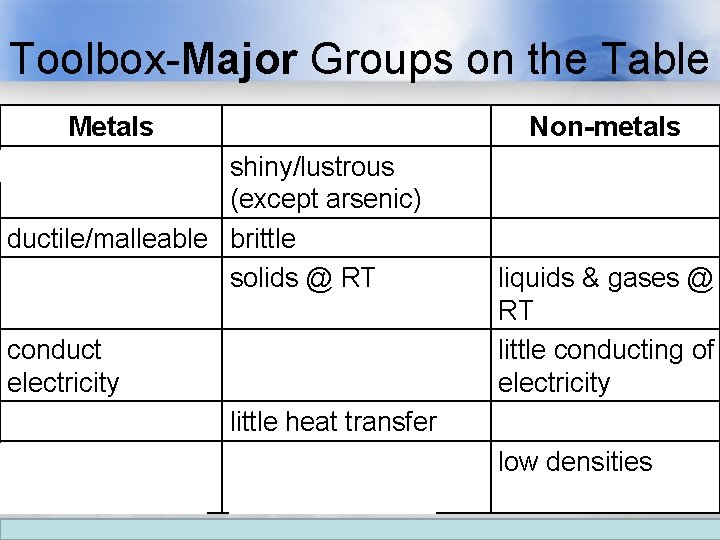
Toolbox-Major Groups on the Table Metals shiny/lustrous Metalloids shiny/lustrous (except arsenic) ductile/malleable brittle solids @ RT (except mercury) conduct electricity conduct heat little heat transfer high densities intermediate densities Non-metals flat/matte brittle if solid liquids & gases @ RT little conducting of electricity little heat transfer low densities
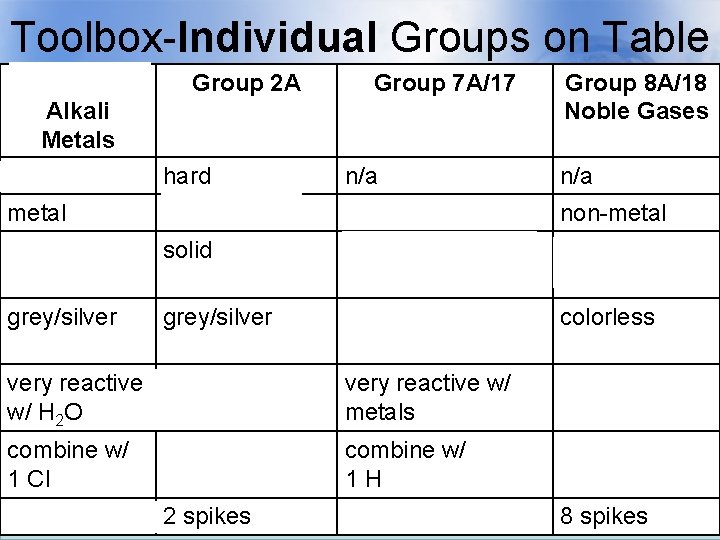
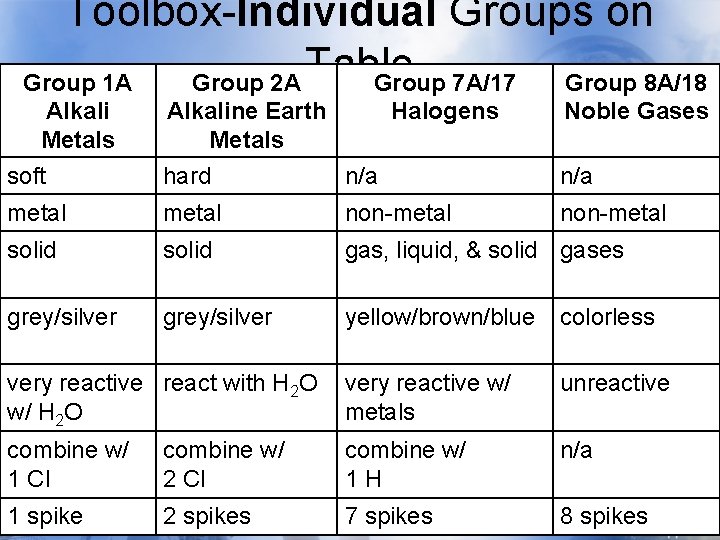
Toolbox-Individual Groups on Table Group 1 A Alkali Metals soft Group 2 A Group 7 A/17 Alkaline Earth Halogens Metals hard n/a Group 8 A/18 Noble Gases n/a metal non-metal solid gas, liquid, & solid gases grey/silver yellow/brown/blue colorless very reactive react with H 2 O w/ H 2 O very reactive w/ metals unreactive combine w/ 1 Cl combine w/ 2 Cl combine w/ 1 H n/a 1 spike 2 spikes 7 spikes 8 spikes
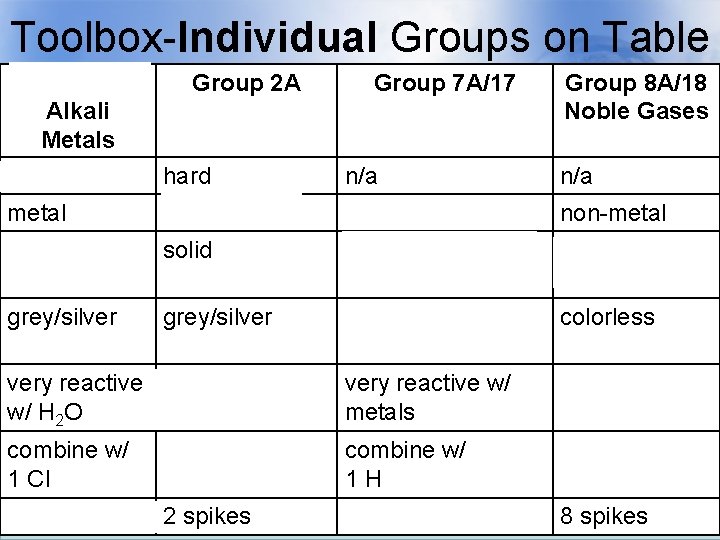
Toolbox-Individual Groups on Table Group 1 A Alkali Metals soft Group 2 A Group 7 A/17 Alkaline Earth Halogens Metals hard n/a Group 8 A/18 Noble Gases n/a metal non-metal solid gas, liquid, & solid gases grey/silver yellow/brown/blue colorless very reactive react with H 2 O w/ H 2 O very reactive w/ metals unreactive combine w/ 1 Cl combine w/ 2 Cl combine w/ 1 H n/a 1 spike 2 spikes 7 spikes 8 spikes
- Slides: 20