UNIT 3 NOTES EDGENUITY CHEMICAL BONDING CHEMICAL BOND

UNIT 3 NOTES EDGENUITY – CHEMICAL BONDING

CHEMICAL BOND – STRONG CONNECTION BETWEEN ATOMS • Ionic – brittle, high melting point, conducts electricity when dissolved in water • Covalent – brittle, low melting point, doesn’t conduct electricity • Metallic – malleable and ductile, high melting point (not as high as ionic), can conduct electricity in solid and molten forms

WHY DO BONDS FORM? • Lower energy = more stability. • Elements want to be stable and have 8 valence electrons • Remember – opposite charges attract or pull towards each other, like charges repel or push away from each other

ELECTRONEGATIVITY • How much an atom wants another electron • Higher values = more reactive • The difference in electronegativity between two atoms affects the type of bond that forms

IONIZATION ENERGY • Energy needed to remove an electron from an atom • Lower values = more reactive • Decreases moving down the groups of the periodic table, increases across a period

IONIC BONDS • Metals tend to lose electrons, non metals tend to gain electrons • Metals and non-metals form IONIC bonds • Ionic bonds – bond resulting from the attraction between oppositely charged ions • Formula unit – the smallest unit of an ionic compound

IONIC BONDS • Sodium has one valence electron. If it loses this electron, it will have 8 valence electrons. • Chlorine has 7 valence electrons. If it gains an electron it will have 8 valence electrons • Sodium basically gives an electron to chlorine. Sodium is + charged ion and Chlorine is a – charged ion. • The + ion and – ion are attracted to one another and an ionic bond is formed

IONIC BONDS • Form crystal lattice structures

COVALENT BOND • Bond resulting from a sharing of electrons between two atoms • Forms when atoms have similar electronegativities • Electronegativity difference is less than 1. 5 for covalent bonds • Examples: water (H 2 O), methane (CH 4) • Typically form between nonmetals

COVALENT BOND Molecule – smallest unit of a covalent compound

METALLIC BONDS • Bond resulting in the sharing of valence electrons among many atoms • Extreme electron sharing – shared among ALL atoms • ‘sea’ of negative electrons around the positive nucleus of many atoms

METALLIC BONDS • The purple electrons are moving easily between the atoms • Causes metals to be malleable and ductile

IONIC BONDS • Cations are positively charged ions. Usually metals. Atoms that lose electrons • Anions are negatively charged ions. Usually nonmetals. Atoms that gain electrons • Ionic compounds from crystals – solids in which the particles are arranged in a regular, repeating pattern

FORMULA UNITS • Smallest repeating unit of an ionic compound. + +2 • Example: Na. Cl (1 Na , 1 Cl )Ca. Cl 2 (1 Ca , 2 Cl -1), Al O (2 Al+3, 3 O-2) 2 3 • Simplest ratio of ions in the crystal lattice • Ionic compounds DO NOT form molecules

POLYATOMIC ION • Is a single unit • Made of atoms that are bonded together covalently • Example: Carbonate – CO 32 - The entire ion has a -2 charge. These act like one thing • Forms ionic bonds with other ions
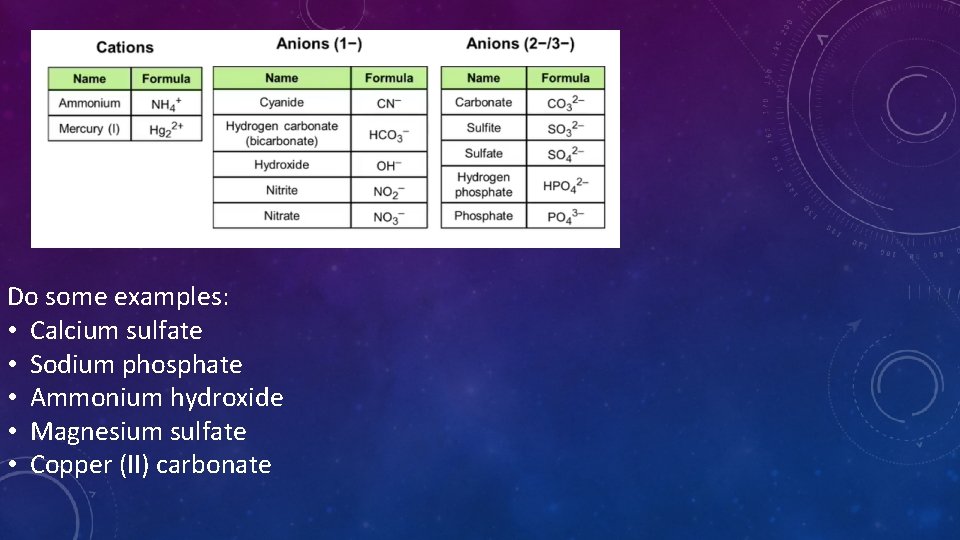
Do some examples: • Calcium sulfate • Sodium phosphate • Ammonium hydroxide • Magnesium sulfate • Copper (II) carbonate

LATTICE ENERGY • Energy released when the gas-phase ions combine to form crystals • Ionic compounds form crystal lattices where each ion is equally attracted to all other oppositely charged ions around it. • Lattice energy is greater for smaller ions and greater for ions with larger charges • It’s a measure of bond strength

PROPERTIES OF IONIC COMPOUNDS • • • High melting and boiling points Crystals or solids at room temperature Ionic compounds are hard Varying solubility Low conductivity as solids, high conductivity as liquids or when dissolved in water (in solution)

NAMING IONIC COMPOUNDS (NOMENCLATURE) • The atom that loses electron(s) becomes positively charged (cation) • The atom that gains electron(s) becomes negatively charged (anion) • These charges must be balanced in an ionic compound

OXIDATION STATE • The charge an element is assigned when bonding, either positive or negative. • For now, the number of electrons gained/lost will correspond to the oxidation state. • Example: Sodium loses 1 electron and has a charge, or oxidation state, of +1. • Fluorine gains 1 electron and has a charge, or oxidation state, of -1.

USING PERIODIC TABLE TO PREDICT CHARGES ON IONS • Metals (form cations) • Group 1(alkali metals): +1 • Nonmetals (form anions) • Group 2 (alkaline earth metals): +2 • Group 16 (oxygen group): -2 • Group 3 (Boron group): +3 • Group 17 (halogens): -1

BALANCING CHARGES • Ionic compounds must have an overall charge of zero • How many -1 ions would bond with a +2 ion? • (+2) + N(-1) = 0. +2 ion. N = 2. So you would need 2 -1 ions to balance one • What is the ratio of +3 ions to -2 ions in a neutral compound? • M(3) + N(-2) = 0. The least common multiple is 6 so you’d need 2 of M = 2 and N = 3.

FIND THE RATIOS FOR THE FOLLOWING • Na+: Br • Al 3+ : Cl • Mg 2+ : O 2 • Al 3+ : O 2 -

NAMING IONIC COMPOUNDS • Name the cation first, then the anion • Metal cations: name of metal • Nonmetal anions: replace ending with –ide • Example: Li. F. Lithium fluoride • Try: Na. Br, Ca. Cl 2, Al 2 O 3, Na 2 O

WRITING IONIC FORMULAS • • Identify cations and charges Identify anions and charges Balance the charges Write a formula using chemical symbols and subscripts Magnesium oxide: Lithium chloride: Aluminum sulfide:
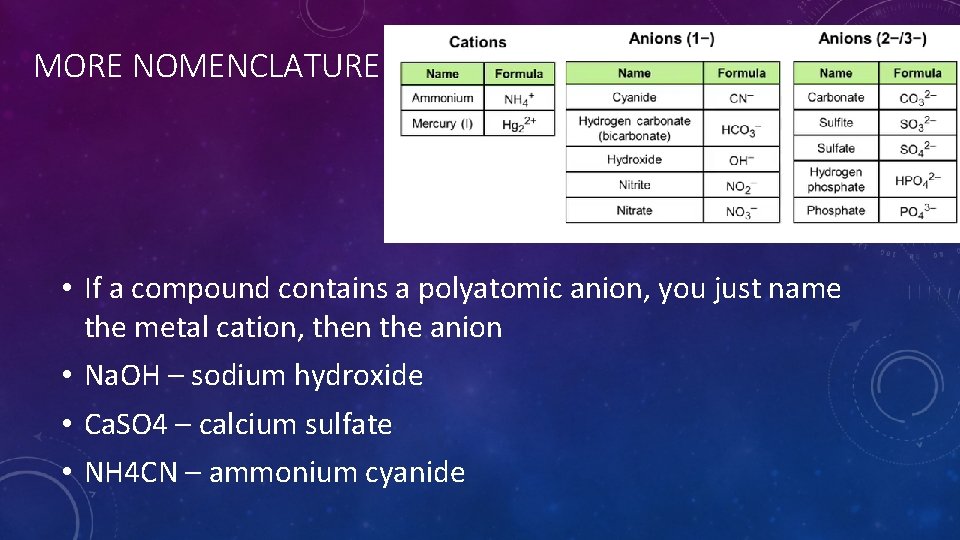
MORE NOMENCLATURE • If a compound contains a polyatomic anion, you just name the metal cation, then the anion • Na. OH – sodium hydroxide • Ca. SO 4 – calcium sulfate • NH 4 CN – ammonium cyanide

WRITING THE FORMULAS FOR IONIC COMPOUNDS CONTAINING POLYATOMIC IONS • The polyatomic ion acts as a single unit. Balance the charges just like before. Use parenthesis if you have more than one polyatomic ion • Calcium bicarbonate: • Ca has a +2 charge. HCO 3 is -1 charge. You need two HCO 3 -1 to balance. • Ca(CO 3)2

IONIC COMPOUND WITH TRANSITION METALS • Transition metals can have more than one oxidation state or charge. When naming compounds, the charge is indicated by roman numerals. • Copper (II) sulfate. Cu 2+ SO 42 -. The formula would be Cu. SO 4. • Mn. O. Mn is a transition metal so we can’t predict the charge. O is -2 charge. Since there is only one Mn in the formula, the charge must be +2 to balance the charge. The name is Manganese (II) oxide

METALLIC BONDING • Properties of metals • Large atoms • Relatively low electronegativities • Low ionization energy • Electrons of metals are mobile and shared among all the metal nuclei – these are delocalized electrons
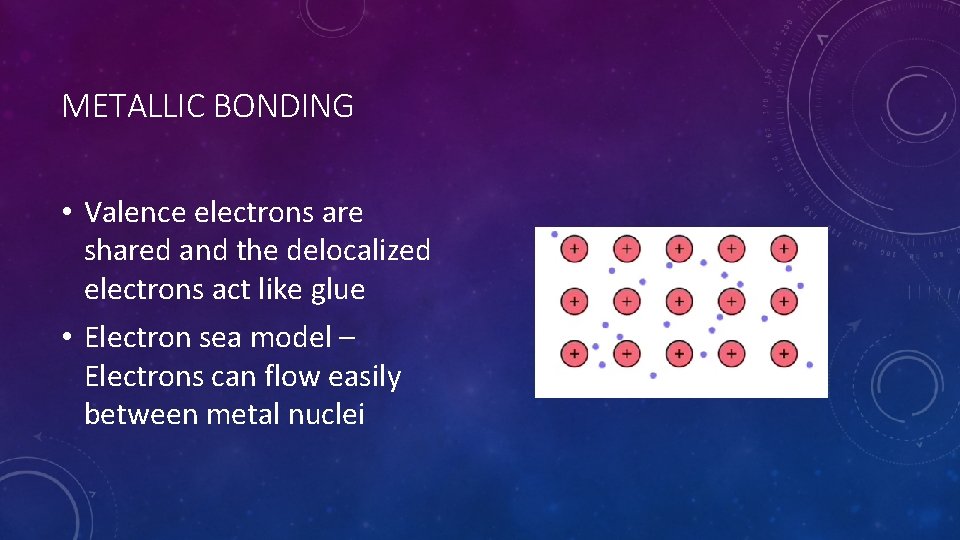
METALLIC BONDING • Valence electrons are shared and the delocalized electrons act like glue • Electron sea model – Electrons can flow easily between metal nuclei

MOLECULAR ORBITALS • Atomic Orbitals that overlap or combine to form shared orbitals • Metals have many atoms and many molecular orbitals • In metals, there could be thousands of atoms and orbitals. These metal molecular orbitals combine to form bands • Electrons can move between bands. • This theory explains the properties of metals more than the electron sea model

MORE PROPERTIES OF METALS • Malleability (can be shaped), Ductility (can be stretched into wires), Luster (shininess) • Due to delocalized electrons. • Luster is due to movement of electrons between bands • Conductivity – delocalized electrons move easily and can carry electrical current and thermal energy • More valence electrons, larger atomic radius corresponds to higher conductivity

ALLOYS • Combination of two or more metals. Considered a homogeneous mixture. • Properties change compared to the pure metal • Composition can vary – different percentages of elements in the alloy

COVALENT BONDING • Formed between two nonmetals. Electrons are SHARED and orbitals overlap • Sigma bond – formed from the overlap of an s, p, or d orbital of one atom with an s, p, or d orbital of another atom
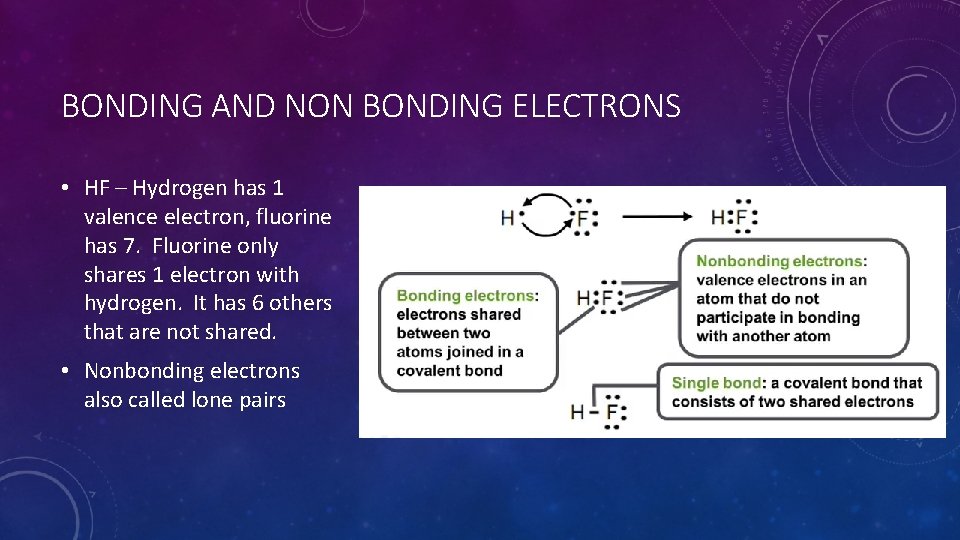
BONDING AND NON BONDING ELECTRONS • HF – Hydrogen has 1 valence electron, fluorine has 7. Fluorine only shares 1 electron with hydrogen. It has 6 others that are not shared. • Nonbonding electrons also called lone pairs

EQUAL AND UNEQUAL SHARING OF ELECTRONS • In H 2 and F 2, electrons are shared equally. The elements involved have the same electronegativity. They both want the electron the same amount. This is a NONPOLAR BOND • POLAR BOND – formed when two atoms have a greater difference in electronegativity. Electrons are not shared equally. The more electronegative element pulls them more. HF is an example. This causes a partial charge on the atom. In HF, the F would have a slight negative charge and H would have a slight positive charge. H 2 O is another example.

LEWIS STRUCTURES • Use chemical symbols and dots for the valence electrons Do on whiteboard

DRAWING LEWIS STRUCTURES OF MOLECULES • Determine the number of valence electrons of each element • Draw the central atom first – often is the first element listed in the formula • Add the other atoms in the formula around the central atom • Form covalent bonds between the central atoms and those around it • Draw the final structure with a line for each bonding pair of electrons, and dots for the nonbonding electrons. • Check that you have all electrons accounted for. Add them up! • Examples: CHCl 3, PH 3, H 2 O, CH 4

OCTET RULE • In most cases, atoms are most stable with a full valence electron shell • Expanded octet – allows for more than 8 valence electrons to be involved in bonding. Some atoms have empty d orbitals that can be used for bonding • There are exceptions • Elements in periods 3 or higher can have more than 8 valence electrons due to the d orbitals. Example SF 6. Draw the lewis structure • Do examples: As. Cl 5, Se. F 4, IF 3,

DOUBLE BONDS • Involve 4 shared electrons, or two pair • O 2 is an example. Draw the Lewis symbol

TRIPLE BONDS • 3 pairs of electrons are shared – 6 electrons total. • Example: N 2. Draw Lewis structure
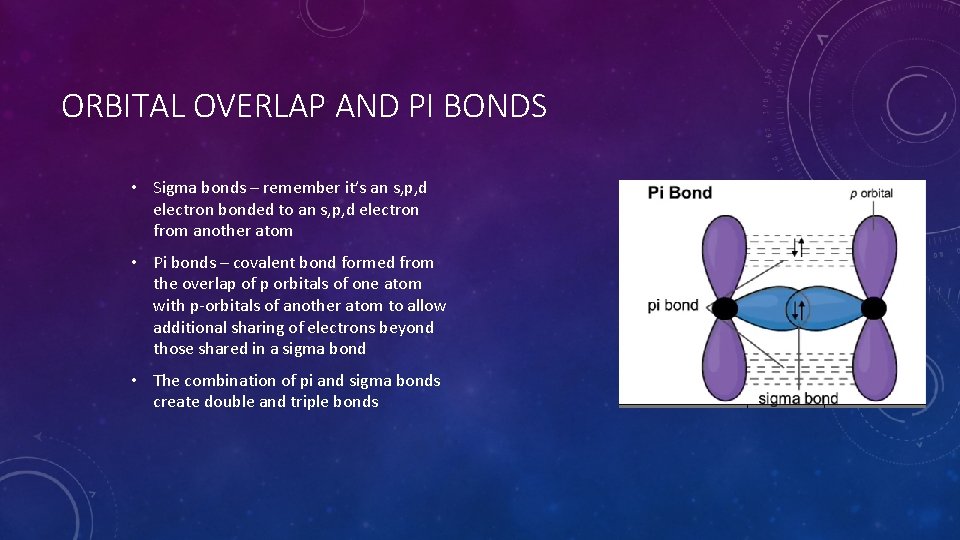
ORBITAL OVERLAP AND PI BONDS • Sigma bonds – remember it’s an s, p, d electron bonded to an s, p, d electron from another atom • Pi bonds – covalent bond formed from the overlap of p orbitals of one atom with p-orbitals of another atom to allow additional sharing of electrons beyond those shared in a sigma bond • The combination of pi and sigma bonds create double and triple bonds

DOUBLE AND TRIPLE COVALENT BONDS • Double bond • One sigma bond (s) • One pi bond (p) • Overlap of two p orbitals • Triple bond • One sigma bond • Two pi bonds

PROPERTIES OF COVALENT COMPOUNDS • Composed of molecules • Do not ionize in solution • Poor conductors • Low melting and boiling points • Gases or liquids at room temperature usually

NAMING IONIC COMPOUNDS - REVIEW • Name the cation and then the anion (ending in –ide) • These names don’t indicate how many of each atom is in the compound – this is determined from the charges on the ions • Charge is indicated by a roman numeral for transition metals that can have several different charges

NAMING COVALENT COMPOUNDS OR MOLECULES • The element farther to the left on the periodic table is named first • The second element is given an –ide ending • Do not include roman numerals • Prefixes are used to indicate how many of each atom are present in the compound • Mono = one. Don’t use if the first element has one. • Di = two • Tri = three • Tetra = four Name the following: NI 3, SO 2, CO, CBr 4, P 4 O 6

SOME COMPOUNDS HAVE COMMON NAMES

COMPOUNDS CONTAINING HYDROGEN • Don’t follow IUPAC naming rules • Some compounds don’t use prefixes to indicate the number of hydrogen atoms • H 2 S – hydrogen sulfide • HF – hydrogen fluoride • HCl – hydrogen chloride (hydrochloric acid) • H 2 SO 4 – Hydrogen sulfate (sulfuric acid)

WRITING FORMULAS FOR COVALENT COMPOUNDS 1. Identify the elements involved 2. Write the chemical symbols in the correct order (the order they appear on the periodic table – left elements first) 3. Use prefixes to determine how many of each atom are present 4. Write the chemical formula using subscripts to indicate number of each atom Try the following: • • Nitrogen trifluoride Dihydrogen monoxide Boron tribromide Phosphorus trihydride

NAMING ACIDS AND BASES • Acids - increase concentration of H+ ions in aqueous solution • May be ionic or covalent • Generally contain H atoms bonded to other atoms or to polyatomic ions • Turn blue litmus paper red • Taste sour • Bases – increase concentration of OHions in aqueous solution • May be ionic or covalent • Ionic bases contain OH- in formula • Covalent bases do not contain OH • Turn red litmus paper blue • Taste bitter • Feel slippery or soapy

NAMING BINARY ACIDS (CONTAIN TWO ATOMS) • Start with hydro- to indicate H. • Add the root of the nonmetal • Add –ic and acid. Example: HCl, HF

NAMING OXYACIDS (CONTAIN POLYATOMIC IONS WITH OXYGEN) • -ate anions make –ic acids • Example: HNO 3 -1 is the nitrate ion. Nitric acid is the name • -ite anions make –ous acids. • Example: HNO 2 -1 is the nitrite anion. Nitrous acid is the name.

NAMING BASES • Covalent bases are usually amines (a form of ammonia – NH 3) • A hydrogen ion can attach to a lone pair of electrons • Naming is based on the number of groups attached to N • Ionic bases follow the same rules as other ionic compounds

INTERMOLECULAR FORCES • Intramolecular forces – act within a molecule, hold a molecule together (chemical bonds) • Intermolecular forces – act between molecules, affect interactions between molecules, much weaker than chemical bonds • Hydrogen bonds • London dispersion forces • Van der Waals forces

HYDROGEN BONDS • The attraction between H bonded to an electronegative atom and an electronegative atom of a different molecule. • Can also be in different regions of the same large molecule • Example: Water – H has a partial positive charge and O has a partial negative charge because of the high electronegativity of oxygen (it’s a polar bond). These partial charges are attracted to one another and form hydrogen bonds between the water molecules (the dotted line is a hydrogen bond)

PROPERTIES OF SUBSTANCES WITH H-BONDS • • Relatively high melting and boiling points High surface tension and viscosity (resistance to flow) Dissolves many ionic and polar covalent substances Solid water (ice) is less dense than liquid water. Uncommon as most substances contract when frozen. • H bonds are the strongest intermolecular force

VAN DER WAALS FORCES • Attractive or repulsive intermolecular forces that are the result of random, short-lived redistribution of electrons • Polar molecule – uneven distribution of electrons over the entire molecule • Polar bond – uneven distribution of electrons over a single bond • Dipole-dipole ineraction • Interactions between polar molecules • Positive poles attracting negative poles • Positive poles repelling positive poles • Can exist between different moleculs

DIPOLE-INDUCED DIPOLE INTERACTION • Interactions between polar and nonpolar molecules • Polar molecule induce charge redistribution in nonpolar molecule • Electrons in nonpolar molecule are evenly distributed. • When the nonpolar molecule gets near the polar molecule with partial charges, it causes the electrons in the nonpolar molecule to shift (attract or repel) • These are weaker than dipole-dipole forces

LONDON DISPERSION FORCES • Van der Waals forces that cause molecules to move apart in the absence of any other intermolecular forces • Induced dipole-induced dipole interaction – are a result of random electron motions • The weakest intermolecular forces • Will affect all substances but only important in nonpolar substances, as polar substances have the other forces which are much stronger

VAN DER WAALS FORCES • A gecko can stick to glass because of the structure of its feet. The feet are covered with millions of tiny hairs – Van der Waals forces between hair tips and the surface create and adhesive force • Water striders – can walk on water because of the high surface tension of water that is due to the hydrogen bonding • DNA – hydrogen bonding holds the two sides of the molecule together, but can be broken during DNA replication • Protein structure – hydrogen bonding is important in specific places • Dish soap – grease is nonpolar and don’t dissolve in water. Soap has an ionic or polar end, and a nonpolar end. The polar end sticks to water molecules and the nonpolar end sticks to grease, allowing grease to be dissolved in water


- Slides: 62