Unit 3 Lecture 4 Periodic Table Trends PERIODIC

Unit 3 Lecture 4: Periodic Table Trends

PERIODIC TABLE AEROBICS!

Periodic Table Trends n Atomic Size/Radius n Ionization Energy n Electronegativity (electron affinity)

Graphing Periodic Trends Activity Use the information in the tables to complete the graph. n Using colored pencil or pen, list under the symbol (in this order!) the Atomic Radius, First Ionization Energy, and Electronegativity. n ¨ Use a different color for each property. Example: Write all of the Atomic radius values in red, all of the First ionization energies in green, and all of the Electronegativities in blue. ¨ Of course, you can pick any colors that are available, as long as you are consistent.

Graphing Periodic Trends Activity Observe the trends in each property as you go down the Alkali metal group, and as you go across Period 3. n Write out each of the statements written on the opposite side, completing each statement with the observed trend (increase or decrease) n

Effective Nuclear Charge (Zeff) (-) Electrons attracted to (+) nucleus n 3 factors that effect this n ¨ The more protons in the nucleus, the greater the Zeff ¨ The more distance between the nucleus and electrons the smaller the Zeff ¨ The more repulsion between electrons the smaller the Zeff

Zeff = Z - S Zeff = effective nuclear charge n Z = # of protons (nuclear charge) n S= screening electrons (core electrons) n Ex. Oxygen: 1 s 22 p 4 n ¨ Zeff n Ex. Fluorine: 1 s 22 p 5 ¨ Zeff n = 8– 2=+6 = 9– 2=+7 Fluorine has a greater effective nuclear change

The Analogy with Atomic Radius n Trey Songz = Nucleus n His Charm = Effective Nuclear Charge n Ladies = Electrons
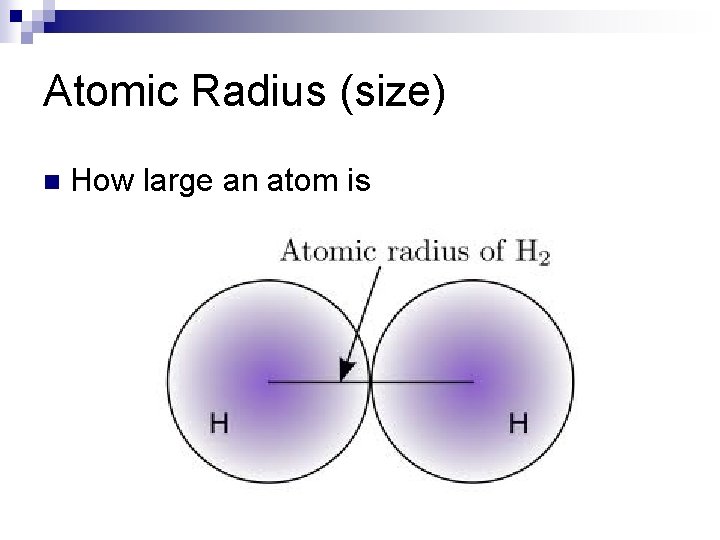
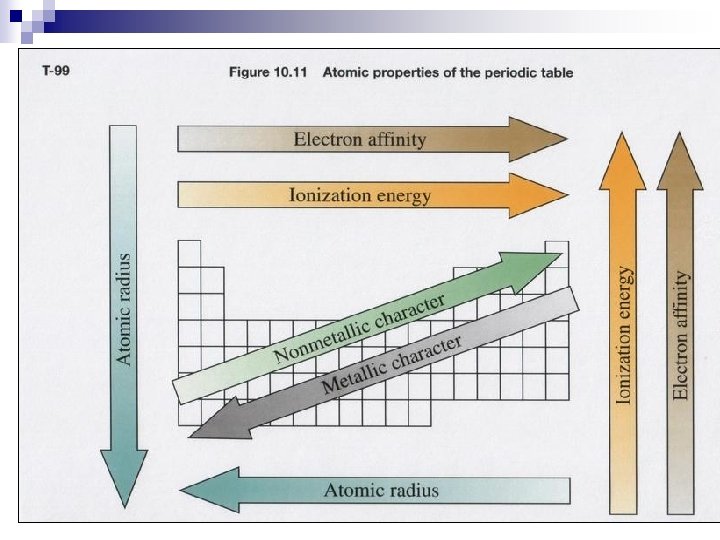
Atomic Radius (size) n How large an atom is

Atomic Radius (notice any patterns? )

Atomic Size/ Atomic radius n down groups Increases ¨Electrons n across ¨There in increased energy levels (shells) periods Decreases. are more valence electrons in the same shell (energy level) so they pull towards the nucleus which has an increased effective nuclear charge with more protons.


First Ionization Energy required to remove one electron from an atom in the gas phase.
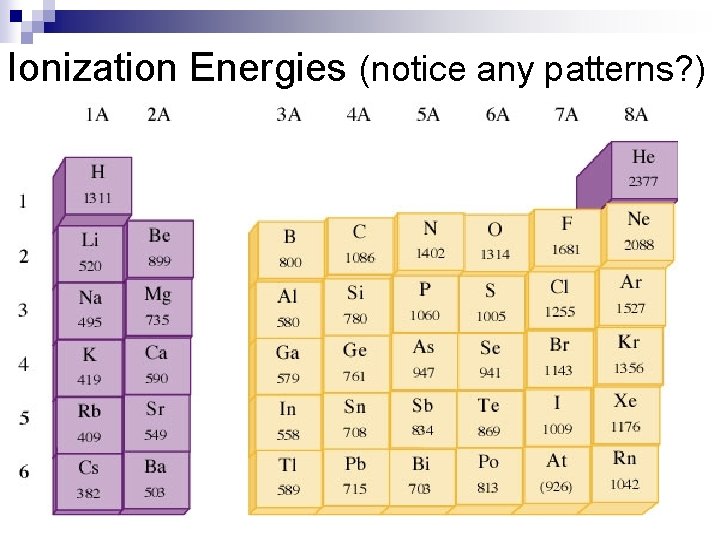
Ionization Energies (notice any patterns? )
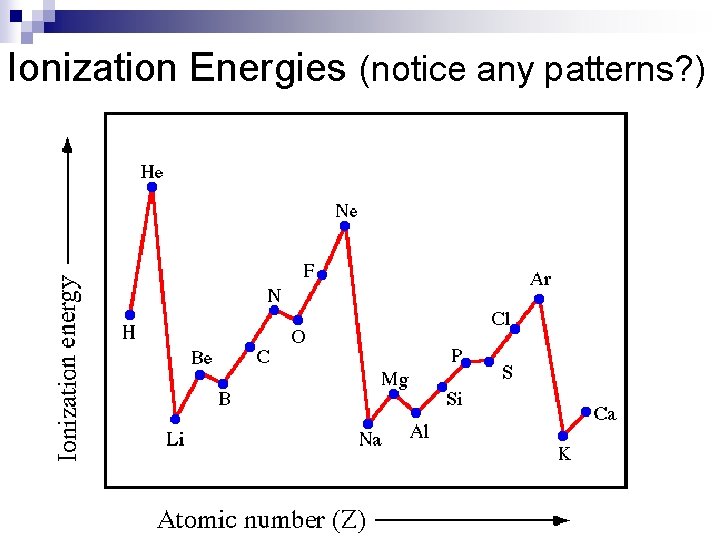
Ionization Energies (notice any patterns? )

Ionization Energy n Down groups Decreases ¨as electrons are further from the nucleus there is more screening of core electrons so less effective nuclear charge. n Across periods increases ¨Increased effective nuclear charge holds electrons more tightly.

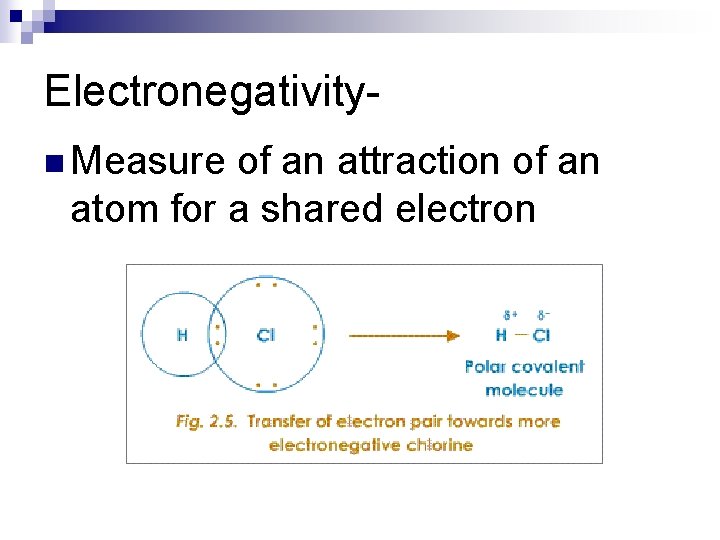
Electronegativityn Measure of an attraction of an atom for a shared electron
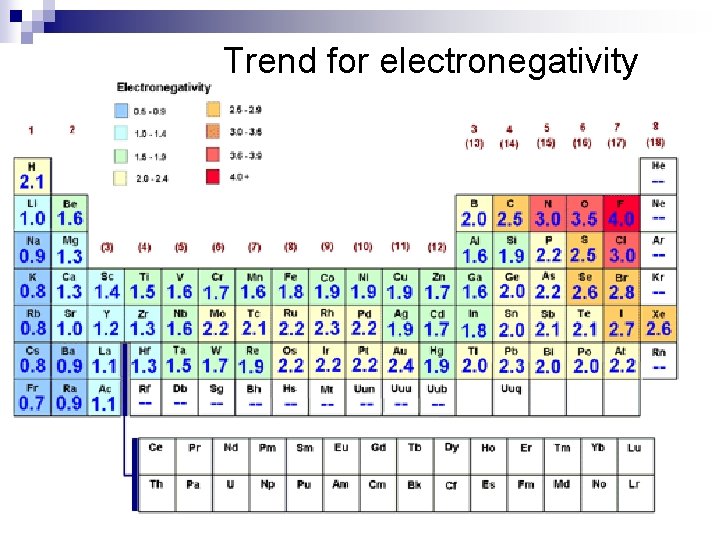
Trend for electronegativity

Trends in Electronegativity: n down groups decreases ¨The electrons are further from the nucleus, so less effective nuclear charge n across ¨as periods increases atoms have more valence electrons in the same energy level and more protons in nucleus they have a higher effective nuclear charge.

- Slides: 22