Unit 3 Electron Configurations Part B Bohrs Model

Unit 3 – Electron Configurations Part B: Bohr’s Model of Hydrogen "Every word I utter is to be understood not as an affirmation but as a question. "

What have we learned so far? Discharging Tubes The Flame Test The light coming out of the excited atomic entities is very specific to particular element! Results are quite reproducible.

Conclusions: �Electromagnetic radiations (lights) coming out or being absorbed by atoms indicate that there are very specific energy changes within the atomic entities (atoms and ions). �No nuclear changes have ever been observed indicate that the energy changes have to come from electrons surrounding the nucleus.

How come? �Classic Model: �Problems with the classic model:

Niels Bohr (1885 – 1962)
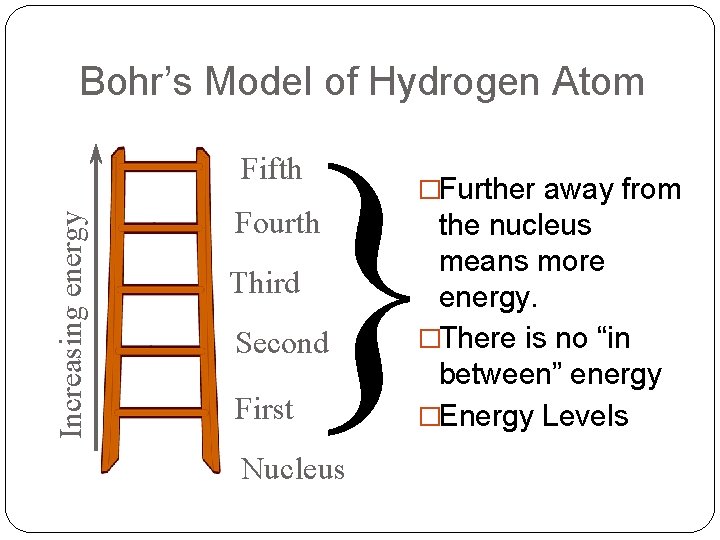
Bohr’s Model of Hydrogen Atom } Increasing energy Fifth Fourth Third Second First Nucleus �Further away from the nucleus means more energy. �There is no “in between” energy �Energy Levels

Bohr’s Model of Hydrogen Atom quantum – quantity of energy gained or lost by an atom when electrons are excited photon – a quantum of light ground state – lowest energy level of an atom excited state – a heightened state of energy in an atom Electrons of hydrogen circle the nucleus in orbits 1. orbits have a fixed amount of energy in the ground state. 2. orbits are a fixed distance from the nucleus. 3. orbits furthest from the nucleus have the greatest energy.

Bohr’s Model of Hydrogen Atom

SUPPLIMENTARY WEBSITES: http: //venables. asu. edu/quant/Dinesh /Bohratom 2. html http: //dl. clackamas. edu/ch 104/lesson 5 bohrmodel. html

Bohr’s Model of Hydrogen Atom
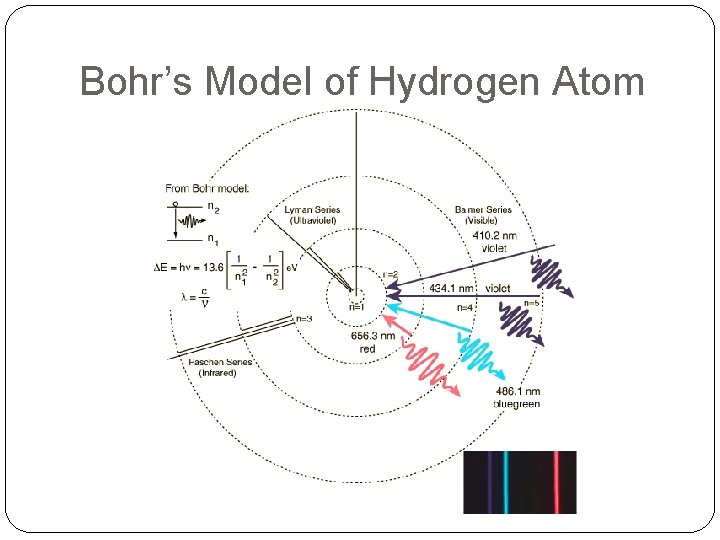
Bohr’s Model of Hydrogen Atom
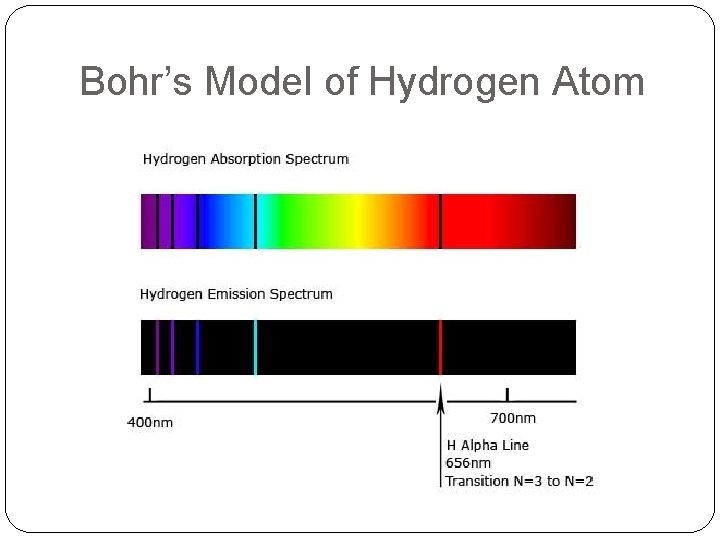
Bohr’s Model of Hydrogen Atom

Bohr’s Model of Hydrogen Atom
- Slides: 13