Unit 3 Day 4 Pure Substances Mixtures and

Unit 3 Day 4 -Pure Substances, Mixtures and Solutions

Objectives Students will be able to analyze substances in order to determine if they are pure substances or mixtures. Students will be able to classify pure substances as compounds or elements Students will be able to classify mixtures as heterogeneous or homogenous

Agenda Bellringer “I do” Activity “We do” Activity “You do” Activity Exit Ticket
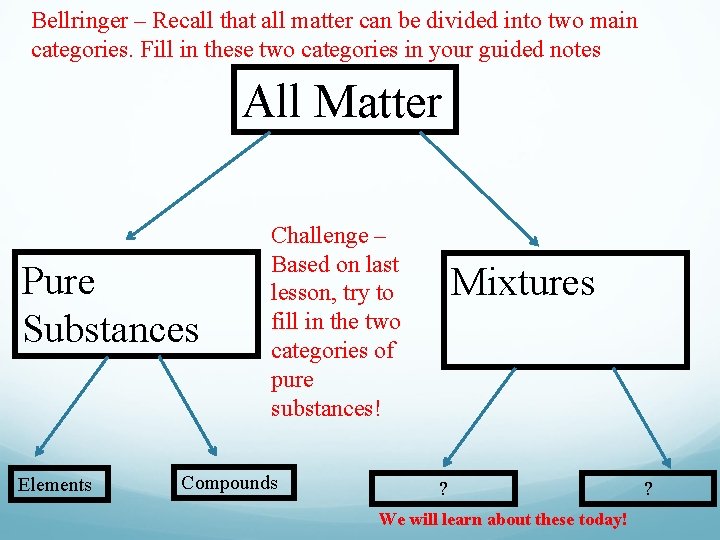
Bellringer – Recall that all matter can be divided into two main categories. Fill in these two categories in your guided notes All Matter Pure Substances Elements Challenge – Based on last lesson, try to fill in the two categories of pure substances! Compounds Mixtures ? We will learn about these today! ?

Review- Elements Element – composed of identical atoms and it is in its purest form. – Elements cannot be split into simpler substances by physical or chemical means. – EX: copper, aluminum, oxygen

Review- Compounds 2. ) Compounds are made up of 2 or more different elements that are chemically combined. • Compounds can be broken into simpler substances by chemical but not physical means. • Example: water, carbon dioxide, nitrogen oxide , cells

Brian Pop • Compounds and Mixtures • http: //www. brainpop. com/science/matteran dchemistry/compoundsandmixtures/ Brain teaser question: What is the difference between a compound a mixture?
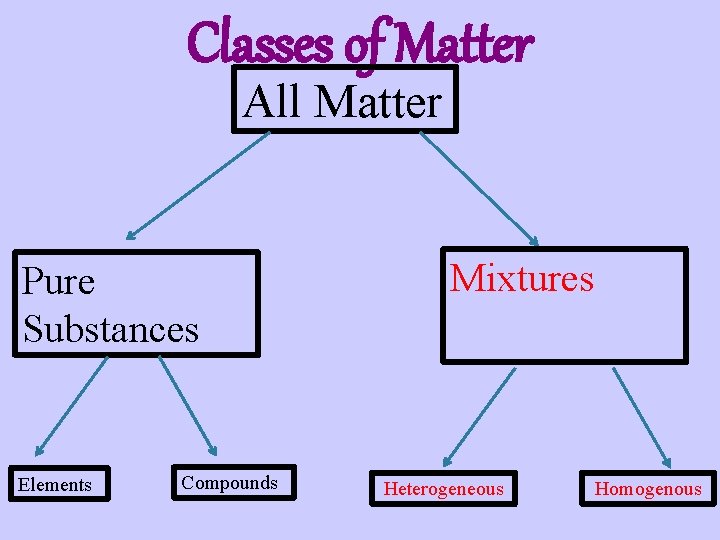
Classes of Matter All Matter Pure Substances Elements Compounds Mixtures Heterogeneous Homogenous

Mixtures Matter that consists of ________ two or more substances mixed ________ together ________but not chemically combined.

Mixtures Can be separated by ______means physical

Properties of Mixtures Substances in a mixture keep most properties of their own _____. Ex. Sugar-water Iron and salt ->

In a mixture: Substances do not change in chemical composition (no new substance is formed)

Classify these examples as a compound or a mixture: 1. Salt: Salt is a compound because it is made of two elements chemically combined (sodium and chlorine…. Remember? )

Compound or a mixture? Salt. Water Salt water is a mixture because the salt and the water are mixed together but do NOT combine chemically
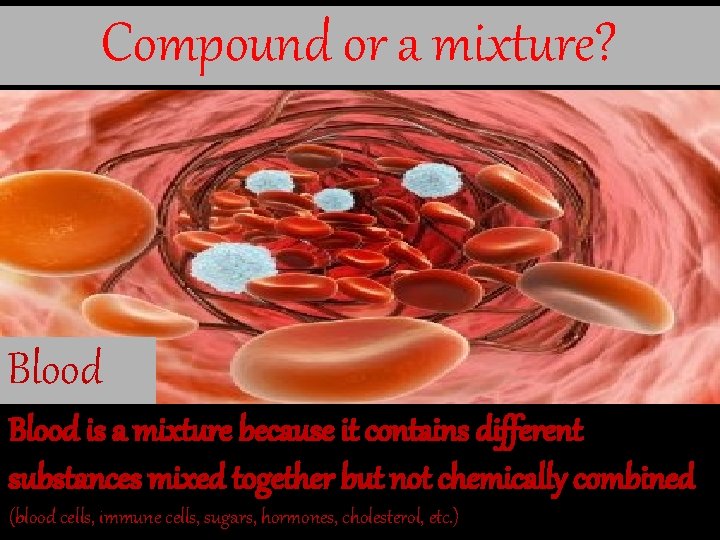
Compound or a mixture? Blood is a mixture because it contains different substances mixed together but not chemically combined (blood cells, immune cells, sugars, hormones, cholesterol, etc. )

Heterogeneous Mixture A mixture that does not appear to be the ___________ same throughout. _______ • Sand Water • Cereal • Oil and Water

Homogeneous Mixture A mixture that appears to be the same throughout. • Remains constantly and uniformly mixed. • Salt Water • Sugar Water (Kool-Aid)

Solutions • Liquid Mixtures • Solutions are the best mixed of all mixtures • Not easily separated by physical means • Koolaid, Lemonade, Salt Water

Two parts of solutions: Solute the substance that is dissolved Solvent the substance that does the dissolving

If I mix sugar into coffee. . What is the Solute? What is the Solvent?
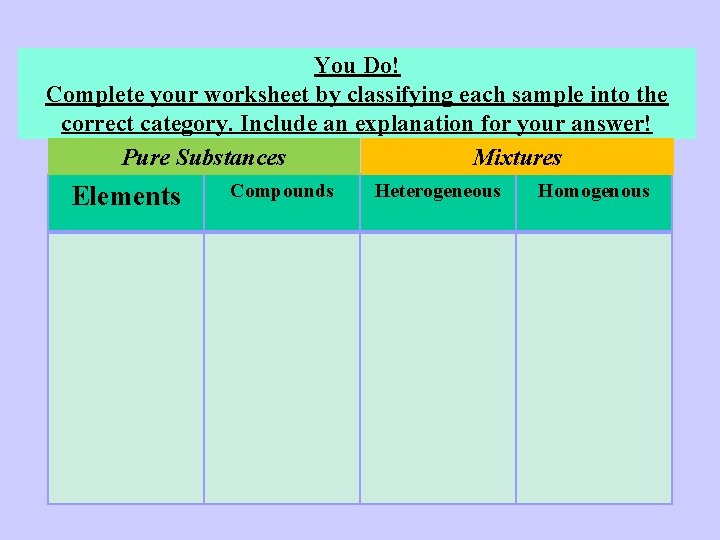
You Do! Complete your worksheet by classifying each sample into the correct category. Include an explanation for your answer! Pure Substances Mixtures Elements Compounds Heterogeneous Homogenous

1) Orange Juice

2) Water

3) Trail Mix

4) Carbon Dioxide

5) Cake Batter

6) Oxygen

7) M & Ms

ANSWERS

1) Orange Juice Orange juice is a mixture as it contains more than one substance in the same place NOT chemically combined. The sugar and other substances can be separated from the water by physical means. However, it is well mixed and looks the same throughout so it is homogenous.

2) Water is a compound as it contains more than one element chemically combined

3) Trail Mix Trail mix is a heterogeneous mixture as it contains more than one substance mixed together but NOT chemically combined and it can be separated through chemical means. You can see and easily separate the different parts.

4) Carbon Dioxide is a compound as it contains more than one element chemically combined

5) Cake Batter Cake batter is a mixture as it contains more than one substance mixed together in the same place but not chemically combined. If it is well mixed and you cannot see the different parts then it is homogenous.

6) Oxygen is an element. Even though it often exists as more than one atom chemically combined into molecules, there is only one type of atom and so it is an element.

7) M & Ms M&Ms are a heterogeneous mixture as you can easily see the different parts and separate them.
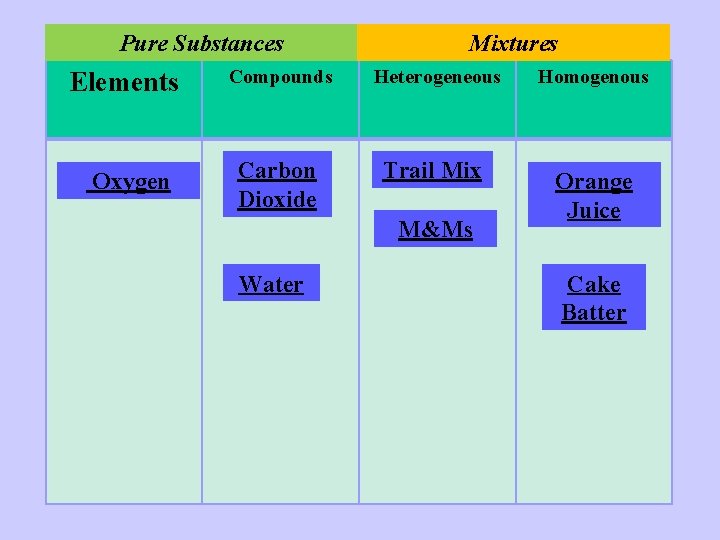
Pure Substances Elements Oxygen Mixtures Compounds Heterogeneous Carbon Dioxide Trail Mix M&Ms Water Homogenous Orange Juice Cake Batter
COMPLETE VENN DIAGRAM INDEPENDENTLY
- Slides: 38