Unit 3 Chemistry Ions and Ionic Bonding What

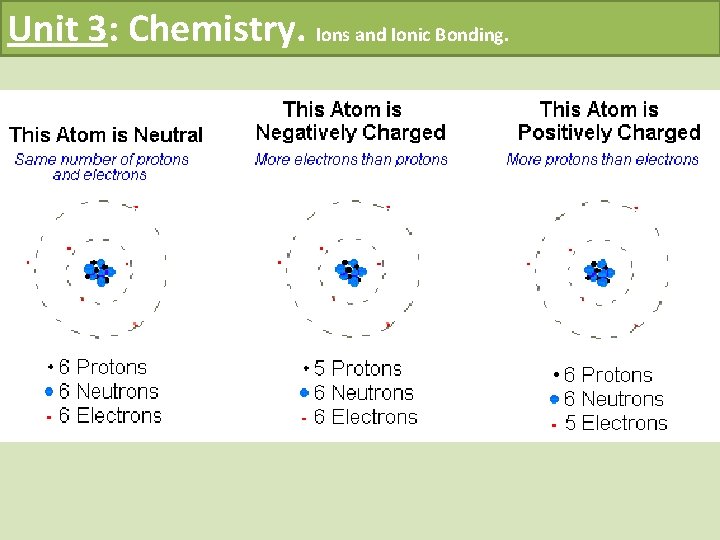
Unit 3: Chemistry. Ions and Ionic Bonding. What are ions? • When atoms gain or lose electrons they become ions • This means they are no longer neutral

Unit 3: Chemistry. Ions and Ionic Bonding. Becoming Happy Atoms • Gaining or losing electrons help elements to become stable (non-reactive) • A stable element has 8 electrons in its valance shell (exception: Helium - He)

Unit 3: Chemistry. Ions and Ionic Bonding. Becoming Stable Atoms – Continued • An atom is likely to lose electrons if it has few valence electrons in its shell Example: metals • An atom is likely to gain electrons if it has many electrons in its shell (non-metals) Example: non-metals

Unit 3: Chemistry. Ions and Ionic Bonding. How to Write an Ion • When an atom loses an electron it becomes positive • When an atom gains an electron it becomes negative • E. g 1) Sodium (Na) loses an electron to become _______ 2) Fluorine (F) gains an electron to become ____
Unit 3: Chemistry. Ions and Ionic Bonding.
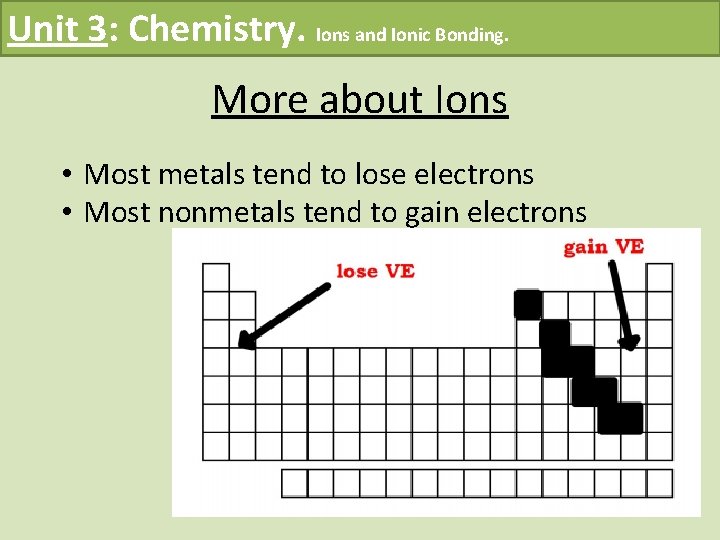
Unit 3: Chemistry. Ions and Ionic Bonding. More about Ions • Most metals tend to lose electrons • Most nonmetals tend to gain electrons

Unit 3: Chemistry. Ions and Ionic Bonding. Examples 1. A) Will chlorine gain or lose electrons? B) How many? C) Does it keep the same outermost level? 2. A) Will aluminum gain or lose electrons? B) How many? C) Does it keep the same outermost level?

Unit 3: Chemistry. Ions and Ionic Bonding • The force of attraction between a positively and negatively charged ion. • Metals tend to lose electrons in chemical reactions to form positive ions • Nonmetals tend to gain electrons forming negative ions • Ionic bonding usually occurs between metals and nonmetals


Unit 3: Chemistry. Ions and Ionic Bonding Review • An ion forms when an atom gains or loses an electron _______ • Positively and negatively charged ions are ionic attracted by ______ bonds • These bonds usually form between metals and Non-metals _______. acids salts • Examples: ______, bases, & _____.
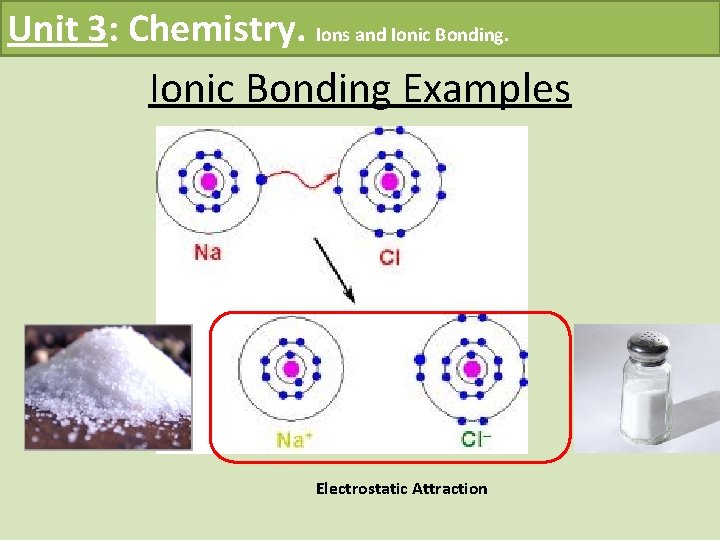
Unit 3: Chemistry. Ions and Ionic Bonding Examples Electrostatic Attraction
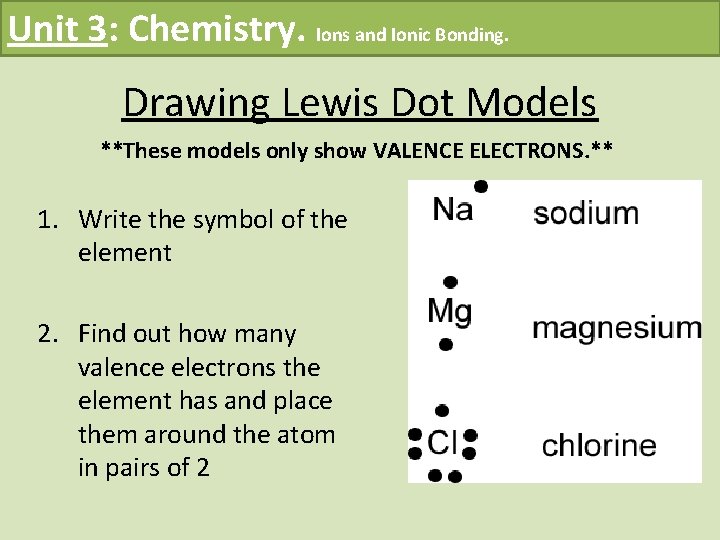
Unit 3: Chemistry. Ions and Ionic Bonding. Drawing Lewis Dot Models **These models only show VALENCE ELECTRONS. ** 1. Write the symbol of the element 2. Find out how many valence electrons the element has and place them around the atom in pairs of 2
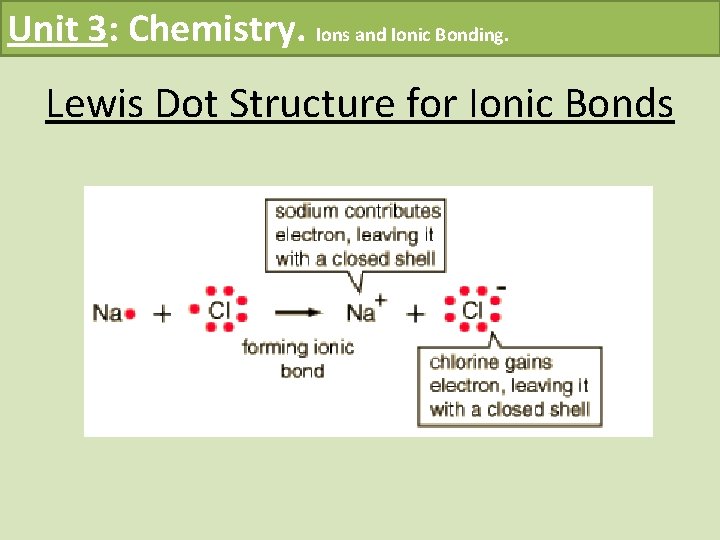
Unit 3: Chemistry. Ions and Ionic Bonding. Lewis Dot Structure for Ionic Bonds
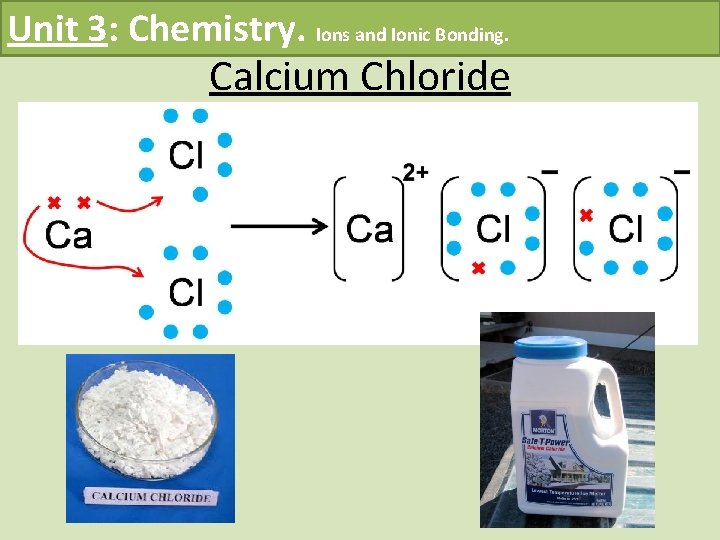
Unit 3: Chemistry. Ions and Ionic Bonding. Calcium Chloride
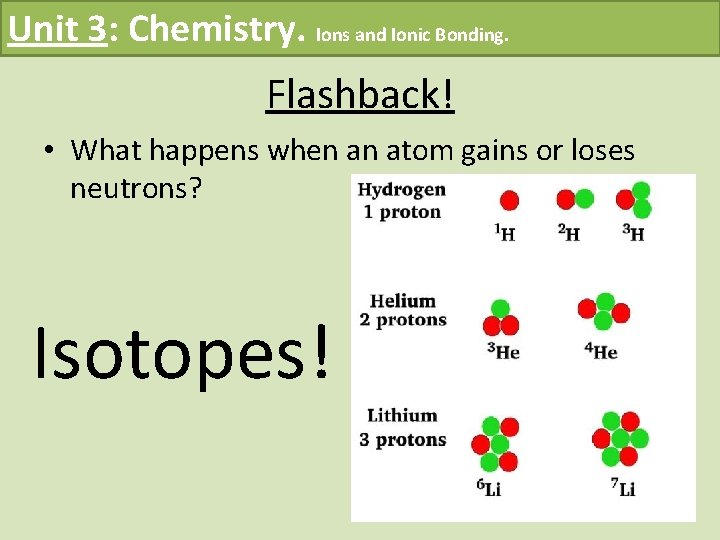
Unit 3: Chemistry. Ions and Ionic Bonding. Flashback! • What happens when an atom gains or loses neutrons? Isotopes!

Unit 3: Chemistry. Ions and Ionic Bonding. Exit Ticket • In your journals, please answer the following questions: 1. What is an ion and how do they form. 2. How does ionic bonding occur?
- Slides: 16