UNIT 3 ATOMS THE BUILDING BLOCKS OF MATTER

UNIT 3 – ATOMS: THE BUILDING BLOCKS OF MATTER - NUCLEAR CHEMISTRY Learning Objectives and Learning Target Packet -Text support Ch. 3 and 21

Atomic Theory Continues… Six Easy Pieces Read Aloud ■ We have come so far in our understanding of atoms. Centuries of researching and countless scientists devoting their lives to create the understanding of the atom today (textbook concepts). ■ However it is NOT over. The more we understand about atoms (how they work, their make up, etc…) the greater our ability to advance science and technology in all aspects of our lives (i. e. medicine).

Check for Understanding ■ What is the charge of a proton? ■ Where would you find a proton in an atom? ■ What is the charge of an electron? ■ Where would you find an electron in an atom? ■ What is the charge of a neutron? ■ Where would you find a neutron in an atom? ■ How big is an electron compared to a proton? ■ How big is a neutron compared to a proton?

The Atom ■ The smallest particle of an element that retains the chemical properties of that element. ■ Consists of two regions: – Nucleus ■ ■ ■ 1. Small region located at the center of an atom 2. Made up of at least one positively charged particle (proton) 3. Made up of usually one or more neutral particles (neutrons) – Region surrounding the nucleus – Electron Cloud ■ ■ 1. Very large compared to size of nucleus 2. Contains the negatively charged particles (electrons)

Refresh: What are Atoms? ■ Atoms are tiny particles that determine properties of all matter. ■ Atoms are the building blocks for molecules. ■ Atoms form elements. ■ Element: A substance that cannot be broken down into simpler substances by chemical means.

Parts of an Atom ■ Proton: A subatomic particle that has a positive charge and is found in the nucleus of the atom. ■ Neutron: A subatomic particle that has NO charge and is found in the nucleus of the atom. ■ Electron: A subatomic particle that has a negative charge and moves around the outside of the nucleus.
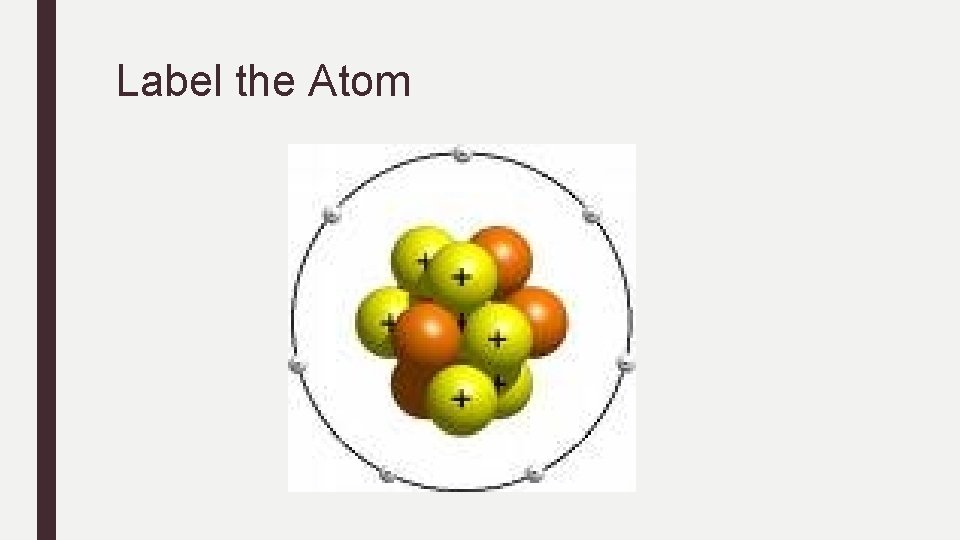
Label the Atom
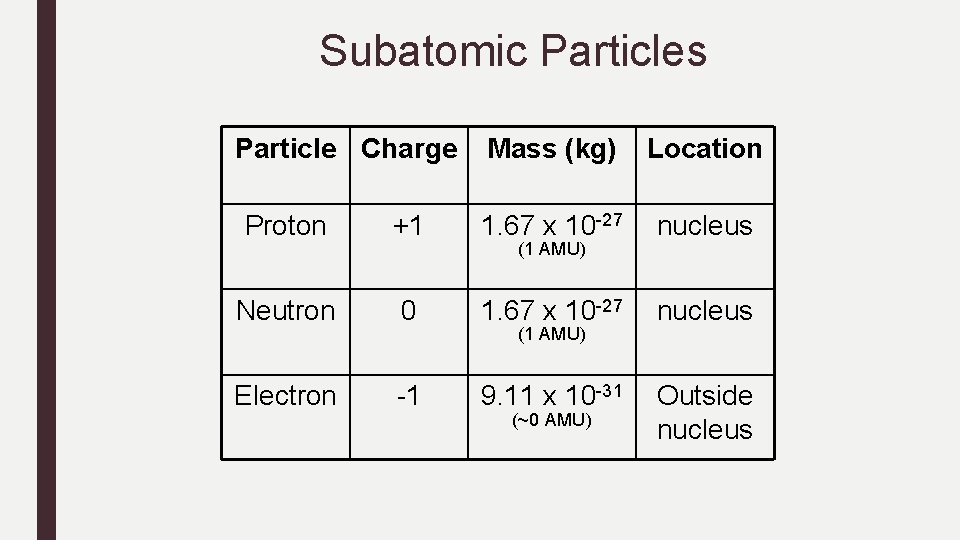
Subatomic Particles Particle Charge Mass (kg) Location Proton +1 1. 67 x 10 -27 nucleus Neutron 0 1. 67 x 10 -27 nucleus Electron -1 9. 11 x 10 -31 Outside nucleus (1 AMU) (~0 AMU)
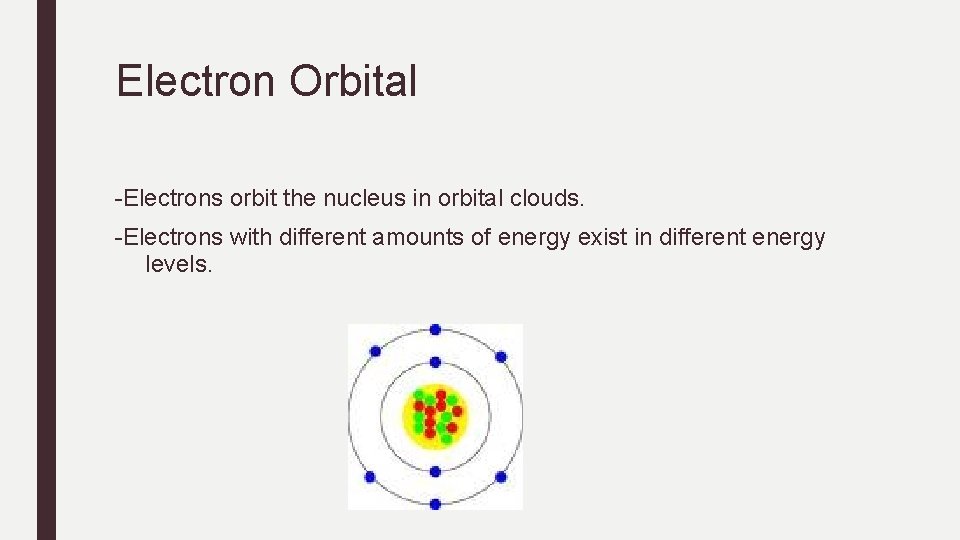
Electron Orbital -Electrons orbit the nucleus in orbital clouds. -Electrons with different amounts of energy exist in different energy levels.
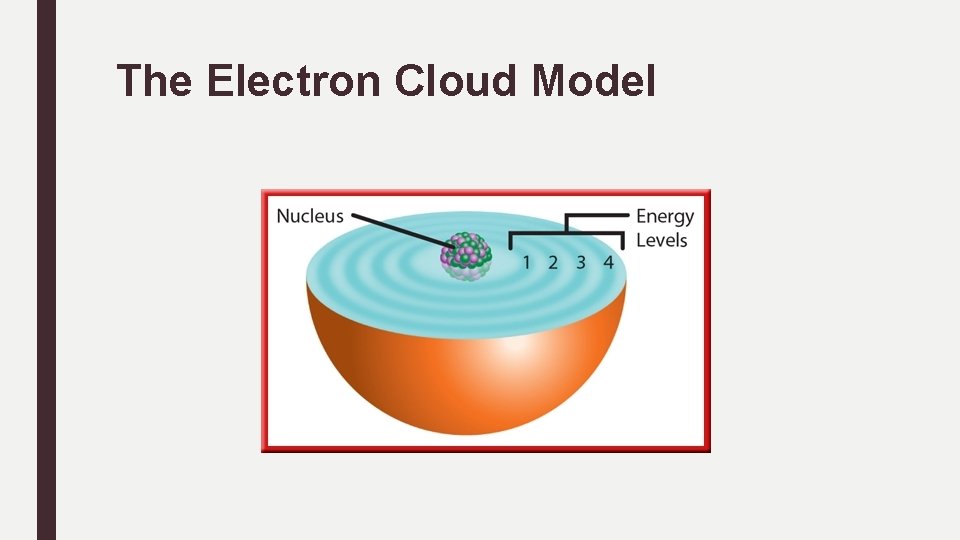
The Electron Cloud Model

Electrons in each energy level • Each energy level can hold a limited number of electrons. • The lowest energy level is the smallest and the closest to the nucleus.
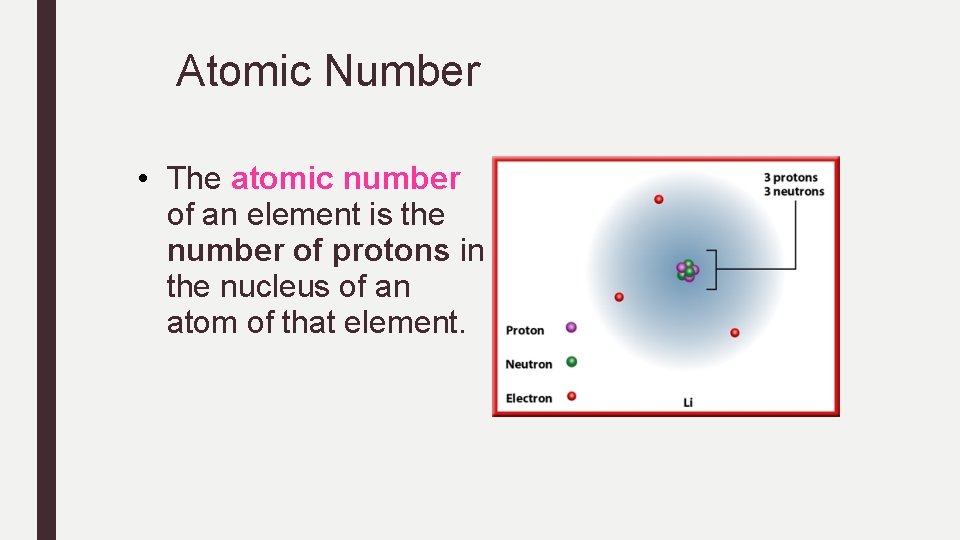
Atomic Number • The atomic number of an element is the number of protons in the nucleus of an atom of that element.

Charge of Atoms ■ Atoms are not charged even though they have particles that contain charges. ■ Atoms are neutral because they have EQUAL numbers of protons and electrons. Ex: Helium Atom Charge of 2 protons: Charge of 2 neutrons: Charge of 2 electrons: Total charge of He atom: +2 0 -2 0
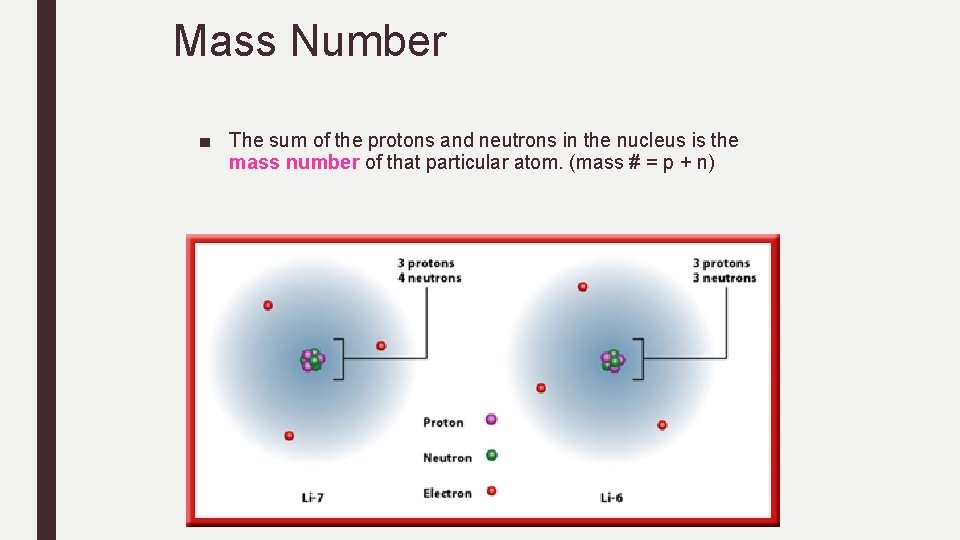
Mass Number ■ The sum of the protons and neutrons in the nucleus is the mass number of that particular atom. (mass # = p + n)

■ Atomic Number vs. Mass Number Atomic Number: Equal to the number of protons in the nucleus of the atom. (number of electrons = the number of protons) ■ Mass Number: Equal to the number of protons AND neutrons in an atom’s nucleus. ■ Average Atomic Weight (below symbol on PT): – Weighted average of the atomic masses of the naturally occurring isotopes of an element – We will round this number to the nearest hundredth (TWO decimal places) ■ Example Oxygen’s average atomic mass is 15. 9994 = 16. 00

Application- TOGETHER Iodine (I) Iron (Fe) Atomic number ____ Atomic Number ____ Mass Number 126 Mass Number ____ Number of Protons ____ Protons____ Number of Neutrons ____ 33 Number of Neutrons Number of electrons ____ Electrons ____ Number of

Application- ON YOUR OWN Nickel (Ni) Atomic number ____ Mass Number ____ Number of Protons ____ Number of Neutrons 31 Number of electrons ____ Radon (Rn) Atomic Number Mass Number 222 Number of Protons____ Number of Neutrons____ Number of Electrons ____

Isotopes • Isotopes of an element have different mass numbers because they have different numbers of neutrons, but they all have the same atomic number.

Isotope Examples ■ Carbon – 12 – 6 protons – 6 electrons – 6 neutrons ■ Carbon – 13 – 6 protons – 6 electrons – 7 neutrons ■ Carbon 14 – 6 protons – 6 electrons – 8 neutrons
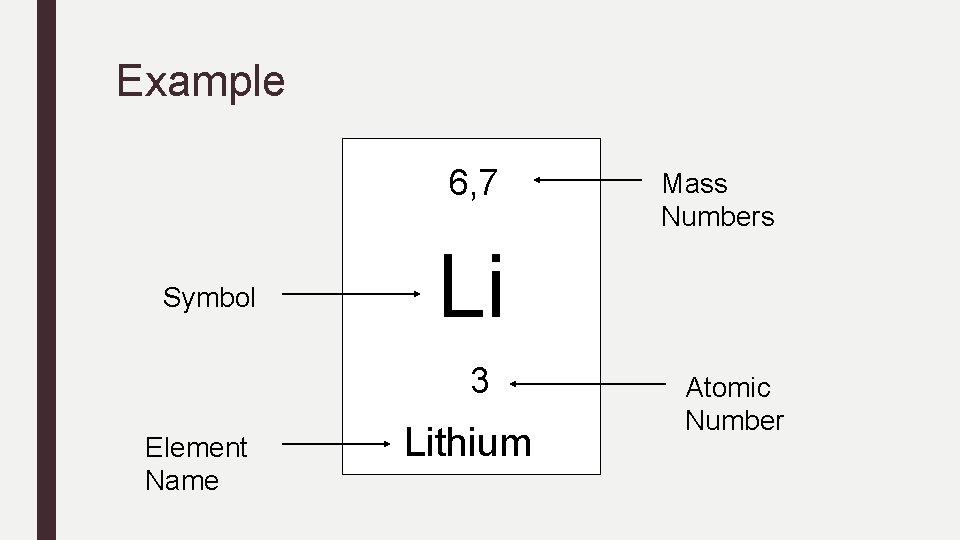
Example 6, 7 Symbol Li 3 Element Name Mass Numbers Lithium Atomic Number

One More Example 107 Ag 47 Silver

Isotope Tables ■ Breaking it down… – To find the symbol – determine the atomic number of the element. This is the number of protons – To find the protons- determine the atomic number of the element. – To find the electrons – equal to the number of protons of a neutral atom – To find neutrons: Mass Number – Atomic Number = Number of Neutrons – To find Mass Number: Atomic Number + Number of Neutrons = Mass Number

Isotopes
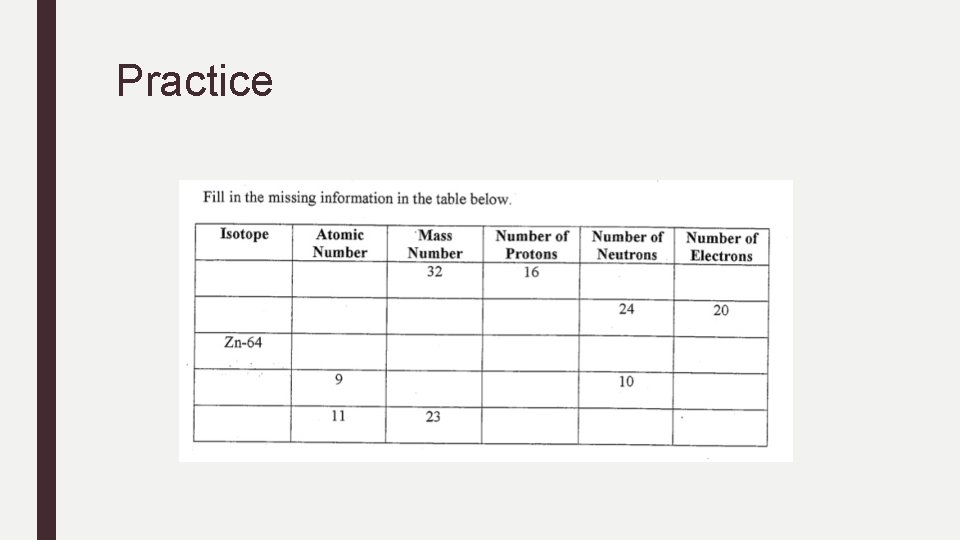
Practice

Molar Mass ■ Molar Mass: mass in grams of one mole of a pure substance. – Units = g/mol ■ To calculate molar mass you must find the atomic mass (Units = g). Ex: Find the atomic mass of Al. Ex: Find the atomic mass of O 2. Ex: Find the atomic mass of CH 4. * To change atomic mass to molar mass just put the answer (g) over moles.

Calculate the molar mass of the following … show ALL your work! ■ 1) Na. Br ■ 2) Pb. SO 4 ■ 3) Ca(OH)2 ■ 4) Na 3 PO 4 ■ 5) (NH 4)2 CO 3 ■ 6) C 6 H 12 O 6 ■ 7) Fe 3(PO 4)2 ■ 8) (NH 4)2 S ■ 9) Zn(C 2 H 3 O 2)2 ■ 10) Ag. F

Warm-up ■ – – – ■ Identify the element: 65 neutrons 48 protons 48 electrons How many protons, neutrons, electrons in the following isotopes – Mn-56 S-31 ■ Determine the molar mass of the following – H 2 O – Na 2 CO 3

AVERAGE ATOMIC MASS Why is the mass on the periodic table not a whole number? Can we have parts of a proton or neutron?

Average Atomic Mass - POGIL ■ Group Roles and Members

What is the average atomic mass of Copper? ■ There are two naturally occurring isotopes of copper. Isotope a. m. u. (atomic mass unit) % Abundance Copper-63 62. 929601 69. 17 Copper-65 64. 927794 30. 83 ■ a. m. u. means atomic mass unit and is the mass of one atom of that particular isotope. ■ – Average atomic mass = Σ (a. m. u. x % abundance in decimal form) Σ means the “sum of” ■ =(62. 929601 x 0. 6917) + (64. 927794 x 0. 3083) ■ =63. 55 amu and also grams/mol…. check the periodic table…. . the mass number is the average atomic mass for copper on your periodic table!
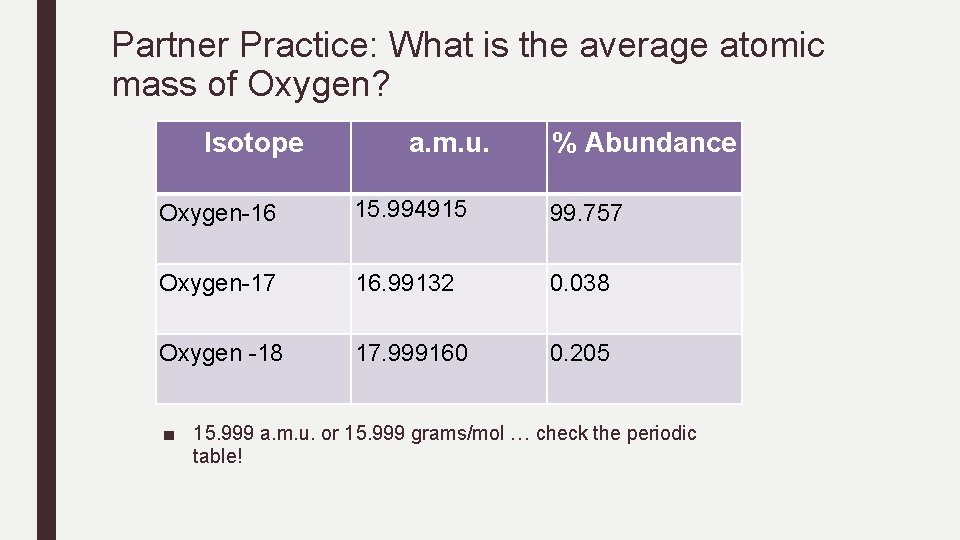
Partner Practice: What is the average atomic mass of Oxygen? Isotope a. m. u. % Abundance Oxygen-16 15. 994915 99. 757 Oxygen-17 16. 99132 0. 038 Oxygen -18 17. 999160 0. 205 ■ 15. 999 a. m. u. or 15. 999 grams/mol … check the periodic table!

Write it Out!! ■ Describe in your OWN words how you can calculate the average atomic mass of an element. – Be sure to describe what information you need to know and how you use it. – **Be prepared to share out your response!**

PERCENT ABUNDANCE What isotopes are most common?

Lithium has two isotopes… Li-6 and Li-7. What is the % abundance of each? Isotope a. m. u. % Abundance Lithium-6 6. 015122 ? Lithium-7 7. 016004 ? ■ Look up Lithium’s average mass on the periodic table: 6. 94 ■ Now it is time for some ALGEBRA! – 6. 94 = 6. 015122(x) + 7. 016004 (1 -x). Solve for x … – 6. 94 = 6. 015122 x + 7. 016004 -7. 016004 x (apply distributive property here!) – Combine like terms ■ ■ 6. 94 – 7. 016004 = -1. 000882 x -0. 076004 = -1. 000882 x and therefore x=0. 0759 and 1 -x = 0. 9241
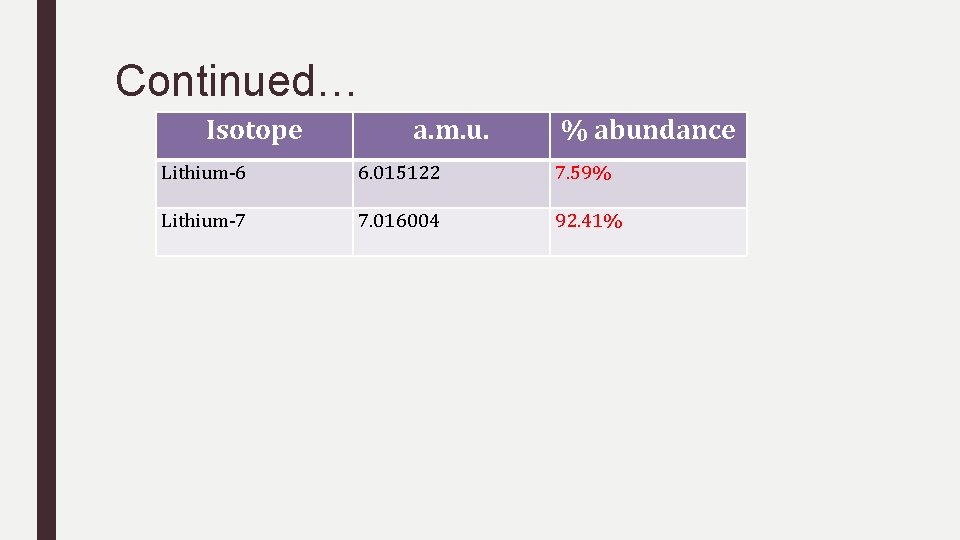
Continued… Isotope a. m. u. % abundance Lithium-6 6. 015122 7. 59% Lithium-7 7. 016004 92. 41%
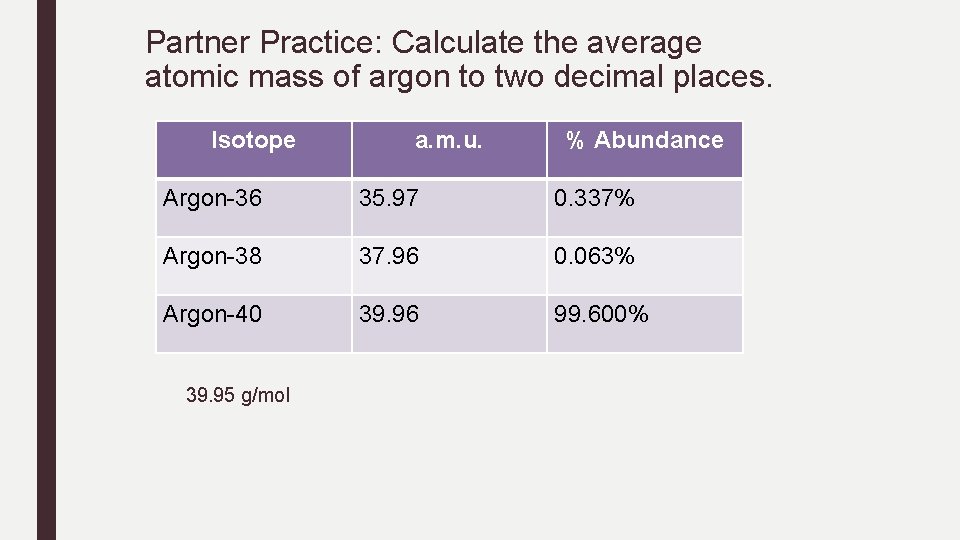
Partner Practice: Calculate the average atomic mass of argon to two decimal places. Isotope a. m. u. % Abundance Argon-36 35. 97 0. 337% Argon-38 37. 96 0. 063% Argon-40 39. 96 99. 600% 39. 95 g/mol

Application… Skittlium! ■ Lab Reflection ■ Question #5: – Determine the average atomic mass for the following sample of skittlium: (Please show your work for full credit!) Type of Skittlium-58 Skittlium-59 Skittlium-60 Number of Atoms 3 9 1 Skittlium-62 6 3

THE MOLE
Mole Music Video ■ http: //www. youtube. com/watch? v=o. Ik. C 7 SRq. XP 0

THE MOLE & AVOGADRO’S NUMBER NA 23 = 6. 02 x 10 http: //ed. ted. com/lessons/daniel-dulek-how-big-is-a-mole-not-the-animal-the-other-one

Counting Particles

Scientists Counting Unit ■ Since atoms are so small, it is hard to count individual atoms. ■ To solve this problem, chemists count by moles. – Moles: SI unit for measuring the amount of a substance. One mole of Carbon

Mole Continued – One mole of anything contains 6. 02 x 1023 “particles” – Particles can be atoms, ions, molecules, electrons, formula units, etc. Avogadro’s Number: 6. 02 x 1023 = 1 mole 602, 200, 000, 000

How Big is Avogadro’s Number? ■ One mole of sheets of paper stacked one on top of the other would reach beyond the solar system. ■ One mole of basketballs could create a new planet the size of the earth. ■ One mole of rice grains would cover the land masses of Earth in a depth of 75 meters. ■ If you had a mole of pennies and gave away 1 million dollars of it (100, 000 pennies) a day to everyone in the world it would take you more than 3000 years to distribute all your money.

Conversions with Avogadro’s Number 1. Start with what you know. 2. Use the following conversion factor: 6. 02 x 1023 particles 1 mole 3. Cancel units. 4. Solve (preform the math).
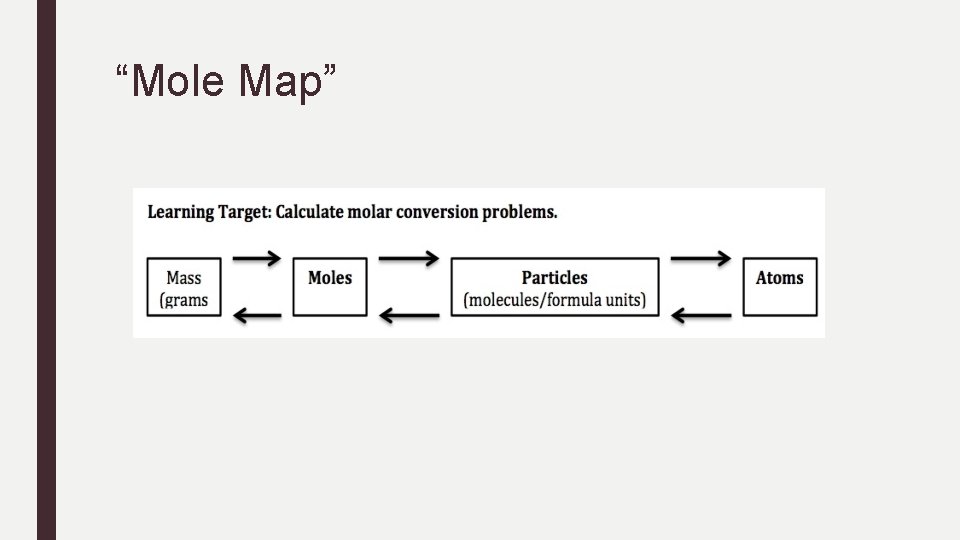
“Mole Map”

Practice Conversions: Moles to Particles 1. Determine how many particles of sucrose are in 3. 50 moles of sucrose. 2. Determine the number of atoms in 2. 50 mol of Zn.

Practice Conversions: Moles to Particles 3. Given 3. 25 mol Ag. NO 3, determine the number of formula units. 4. Calculate the number of molecules in 1. 15 mol of water.

Warm-Up Calculate the number of molecules in 1. 15 mol of water.
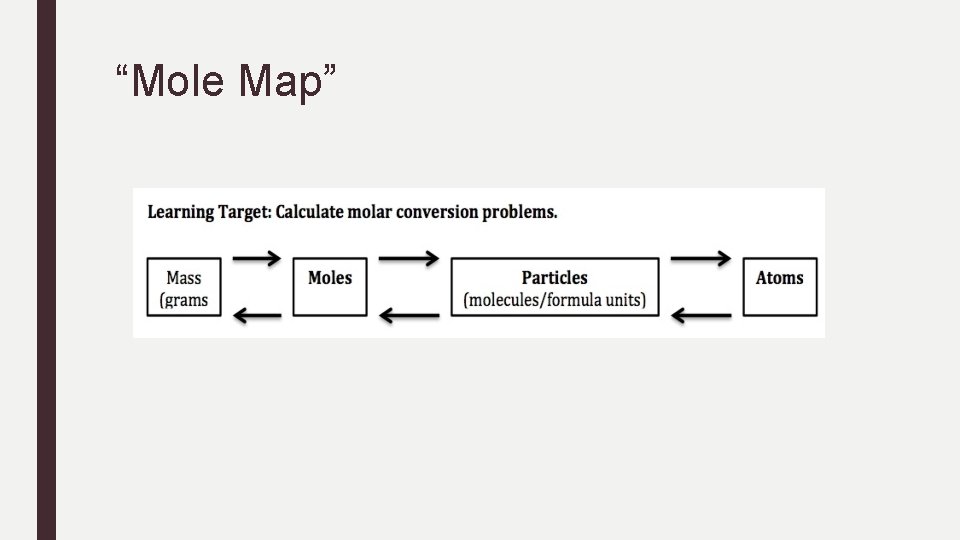
“Mole Map”

Practice Conversions: Particles to Moles 1. How many moles are in 5. 75 x 1024 atoms Al?

Practice Conversions: Particles to Moles 2. How many ATOMS are in 3. 75 x 1024 molecules of CO 2? 3. How many moles are in 2. 50 x 1020 atoms of Fe?

– Mole Conversion Worksheet Practice, Practice

Check for Understanding ■ 1. 5 moles Cu = ____ atoms Cu ■ 8. 51 x 1023 SO 3 = ____ moles SO 3 Solutions: – 9. 0 x 1023 atoms Cu – 28. 3 g SO 3

MOLAR MASS AND CONVERSIONS Adding Mass-Mole and Mole-Mass Finally, put it all together and what do you get? !

Molar Mass ■ Molar Mass: mass in grams of one mole of a pure substance. – Units = g/mol ■ To calculate molar mass you must find the atomic mass (Units = g). Ex: Find the atomic mass of Al. Ex: Find the atomic mass of O 2. Ex: Find the atomic mass of CH 4. * To change atomic mass to molar mass just put the answer (g) over moles.

Mole-Mass Conversion Factor: Mass (g) Moles P. Table 1 Ex: While working in a Chemistry lab, Gary needs 3. 00 moles of Mn for a chemical reaction. How much Mn does Gary need to mass?

Practice with Mole-Mass Conversions ■ Determine the moles in each of the following: ■ Determine the mass in grams of each of the 4. 25. 5 g Ag following: 1. 3. 57 mol Al 5. 125 g Zn 1. 42. 6 mol Si 1. 3. 45 mol Co 6. 1. 45 kg Fe
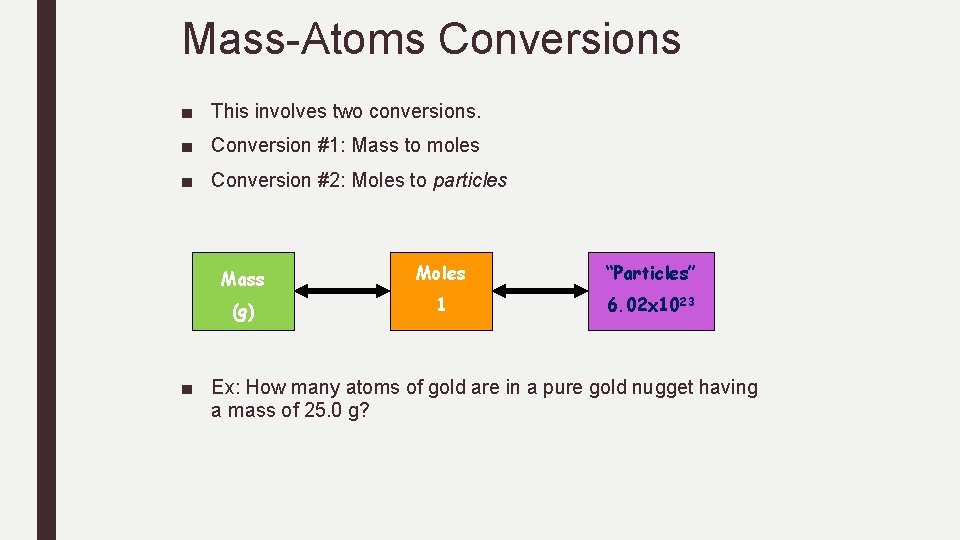
Mass-Atoms Conversions ■ This involves two conversions. ■ Conversion #1: Mass to moles ■ Conversion #2: Moles to particles Mass Moles “Particles” (g) 1 6. 02 x 1023 ■ Ex: How many atoms of gold are in a pure gold nugget having a mass of 25. 0 g?

Practice Mass-Atom Conversions How many atoms are in the following samples? 1. 55. 2 g Li 2. 0. 230 g Pb 3. 45. 6 g Si
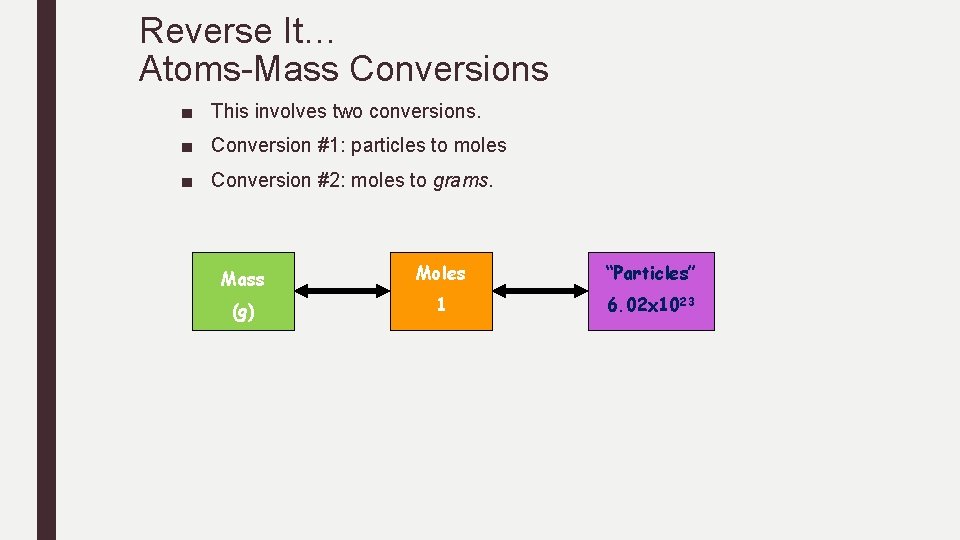
Reverse It… Atoms-Mass Conversions ■ This involves two conversions. ■ Conversion #1: particles to moles ■ Conversion #2: moles to grams. Mass Moles “Particles” (g) 1 6. 02 x 1023

REVERSE IT…. Practice Atom-Mass Conversions How many grams are in the following samples? 4. 6. 02 x 1024 atoms Bi 5. 3. 40 x 1022 atoms He 6. 1. 50 x 1015 atoms U

Tie up your shoes its… PRACTICE TIME!!! Mole Conversion Worksheet
Mole Music Video ■ http: //www. youtube. com/watch? v=o. Ik. C 7 SRq. XP 0

REVIEW Unit 3 – Atoms: The Building Blocks of Matter & Nuclear Chemistry

Review Questions ■ What is the charge of a proton? ■ Where would you find a proton in an atom? ■ What is the charge of an electron? ■ Where would you find an electron in an atom? ■ What is the charge of a neutron? ■ Where would you find a neutron in an atom? ■ How big is an electron compared to a proton? ■ How big is a neutron compared to a proton? ■ What part of the atom takes up the most space? ■ What part of the atom contains the most mass?

Complete the following table Particle Charge Mass (amu) Location +1 Neutron -1 1
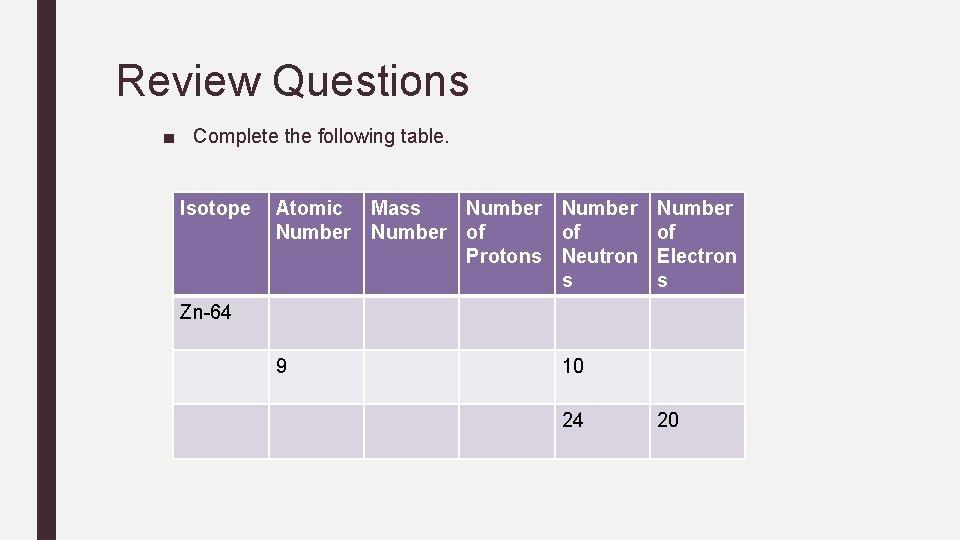
Review Questions ■ Complete the following table. Isotope Atomic Mass Number of of Protons Neutron s Number of Electron s Zn-64 9 10 24 20

Individual Review Time ■ – – – Review Ch. 3 Worksheets, Practice Packet, and Notes! Atomic Structure Isotopes Mole Conversions Molar Mass Average Atomic Mass % Abundance

NUCLEAR CHEMISTRY Explore Pages 649 -671 10 minutes Unit Packet Questions and Practice

Nuclear Chemistry – Pages 649671 – Nuclear Reactions = Changes in the # of protons or neutrons in an atom’s nucleus – Nuclide - The nucleus of an atom – Stability of nuclide depends on the ratio of neutrons to protons ■ Too few or too many neutrons make the nuclide unstable and nuclide decays to form more stable nuclides. ■ The most stable nuclei cluster over a range of neutron-proton ratios called the band of stability (page 683 in book) ■ For lower atomic numbers, typically 1: 1 ratios of protons to neutrons, for higher atomic numbers typically 1: 1. 5 ■ Nuclides are most stable when they reach a magic numbers (natural occurrences that are more stable).

Types of Radiation ■ ■ ■ alpha = rapidly moving helium nuclei (2 protons and 2 neutrons) ■ positron or + = particle with same mass as an electron, but a positive charge ■ ■ Other types of radiation do exist – microwaves, radar, light, etc. beta negative ( -) = extremely fast electrons, little mass gamma – electromagnetic radiation – no mass, no charge, energy only Radiation is classified as ionizing or non-ionizing – Ionizing radiation = enough energy to change atoms and molecules into ions (alpha, beta, X-rays, gamma)/Can cause changes in living cells – Non-ionizing radiation = cannot ionize matter (radio, light)

What is nuclear decay? ■ Nuclear decay is an exothermic process discovered by Henri Becquerel – Each decay results in a nucleus that contains less energy – Stability of nuclide depends on the number of neutrons to protons ■ ■ Too few or too many neutrons makes the nuclide unstable Nuclide decays to form more stable nuclides ■ Describing Nuclear Decay = Equations are used to represent – Initial nucleus = parent nucleus, new nucleus = daughter nucleus – After any decay, the daughter nuclide is less energetic than the parent – The daughter is also more stable – better neutron/proton ratio

Alpha Emission – Occurs with heavy nuclei – decreases neutrons and protons to reach stability. – In alpha emission, the parent nuclei decays into the daughter nuclide by emitting alpha particles (helium nucleus, 42 He) – Example – Uranium-238 – Nuclide loses 2 protons and 2 neutrons

Beta Emission ■ 1. Occurs if too many neutrons to protons ■ 2. Either electron or positron is released from the parent nuclide ■ 3. -, an electron is given off, but is it not one of the original electrons a. The electron is formed from the change of a neutron into 1 proton and 1 electron b. The daughter nuclide thus has a new atomic number because the number of protons increases by 1; mass stays the same c. New atomic number = new element symbol ■ 4. Example: Thorium-234 Pa 91 ■ 5. Positron or + emission occurs when the parent nuclide has too few neutrons for the number of protons positron 234 90 Th 0 -1 + a. Results in conversion of a proton into a neutron – Releases b. Decreases the atomic number by one, mass stays the same c. New atomic number = new element symbol ■ 6. Example: Potassium-38 38 Ar 18 38 0 19 K +1 +

Electron Capture ■ 1. Occurs with too few neutrons to protons, like positron ■ 2. An inner orbital electron is captured by the nucleus of its own atom. ■ 3. This electron combines with a proton and a neutron is formed. ■ 4. Decreases the number of protons and increases neutrons. ■ 5. The atomic number is decreased by one, but the mass stays the same. ■ 6. Electron capture is shown on the left side of the equation. 106 0 106 Pd 47 Ag + -1 e 46

Neutron and Gamma Decay ■ Gamma Decay – Equation results in no change in parent nuclide. ■ Neutrons can also be released from nuclear reactions – 253 Es + 4 He 1 n + 256 99 2 0 101 Md

Types of Radiation Compared Type of Radiation Alpha Symbol Change in Mass # Change in Atomic # Decreases by 4 2 Charge 2+ Beta No Change Increases by 1 1 - Gamma No Change 0

Nuclear Fission ■ Nuclear Fission = a very heavy nucleus splits into more stable nuclei or intermediate mass and releases large amounts of energy – Can occur spontaneously or when nuclei are bombarded with particles. – A chain reaction = a reaction in which the material that starts the reaction is also one of the products and can start another reaction. ■ ■ ■ For example, when fission of uranium=235 occurs, the 2 or 3 neutrons given off cause the fission of other uranium-235 nuclei. Continues until all atoms are split or neutrons fail to hit uranium-235 nuclei. (mass hits a certain level. The minimum amount of nuclide that provides the number of neutrons to sustain a chain reaction = critical mass. Uncontrolled chain reactions = nuclear bombs Controlled chain reactions = nuclear reactors (used to produce energy or radioactive nuclides. ) Nuclear Power Plants use heat from reactors to produce electrical energy.

Nuclear Fusion – Light mass nuclei combine to form heavier, more stable nucleus. ■ Like Nuclear Fission, releases high amounts of energy - Releases even more energy per gram of fuel than nuclear fission. ■ Occurs on the sun and in other stars. – Like Nuclear Fission, uncontrolled fusion reactions are used to make bombs – uncontrolled fusion reactions of hydrogen are the source of energy for the hydrogen bomb. – Fission reactions are used to provide heat and pressure to trigger the fusion. – We are investigating ways to use this power in a controlled way.

Calculating Half-lives ■ A. Half-life (t 1/2) = time required for half of the atoms of a radioactive nuclide to decay. ■ B. Solve half-live problems, first determine the rate constant (k) k = ln(0. 5)/half life years ■ C. Use the k value to determine how long it takes for decay to occur t 1/2 = ln(percent left as decimal)/k

Half-life Examples ■ The radioactive isotope cobalt-60 has a half-life of 5. 27 years. What is the rate constant? K = ln(0. 5)/5. 27 = -0. 132 ■ How long will it take 20% of the cobalt-60 to change to nickel 60? t 1/2 = ln(. 80)/-0. 132 = 1. 69 years ■ How long will it take 70% of the cobalt-60 to decay to nickel 60? t 1/2 = ln(. 30)/-0. 132 = 9. 12 years

BE PREPARED FOR THE SPIRAL ASSESSMENT!
- Slides: 83