Unit 2 The Periodic Table The Periodic Table

Unit 2 The Periodic Table

The Periodic Table The most important tool in chemistry Used to understand predict the properties of elements

Dmitri Mendeleev Russian chemist Proposed the first Periodic Table 1871 http: //members. aol. com/profchm/periodic. html

The First Periodic Table Arranged according to increasing atomic mass "The properties of the elements are a periodic function of their atomic masses" – Dmitri Mendeleev http: //members. aol. com/profchm/periodic. html

The First Periodic Table Columns were organized so that elements with similar properties were in the same column.
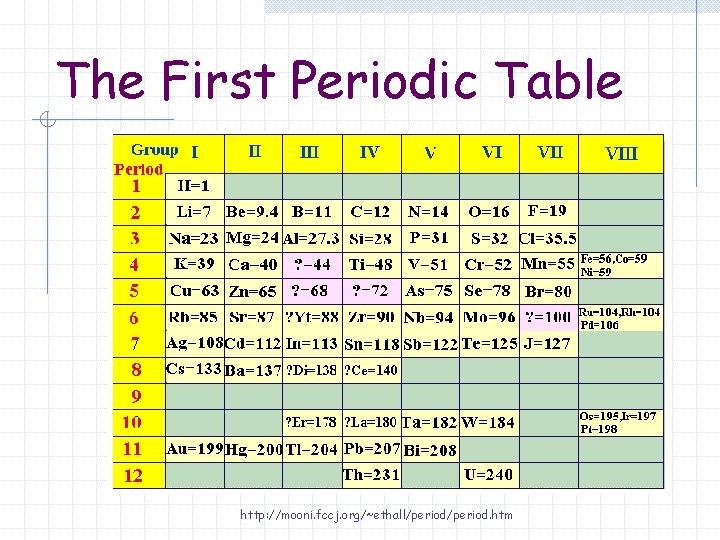
The First Periodic Table http: //mooni. fccj. org/~ethall/period. htm

Henry Moseley British physicist 1914 Similar arrangement as that of the Modern Periodic Table http: //members. aol. com/profchm/periodic. html

Moseley Arranged elements according to increasing atomic number Rearrangement cleared up inconsistencies and contradictions of Mendeleev’s Periodic Table http: //members. aol. com/profchm/periodic. html
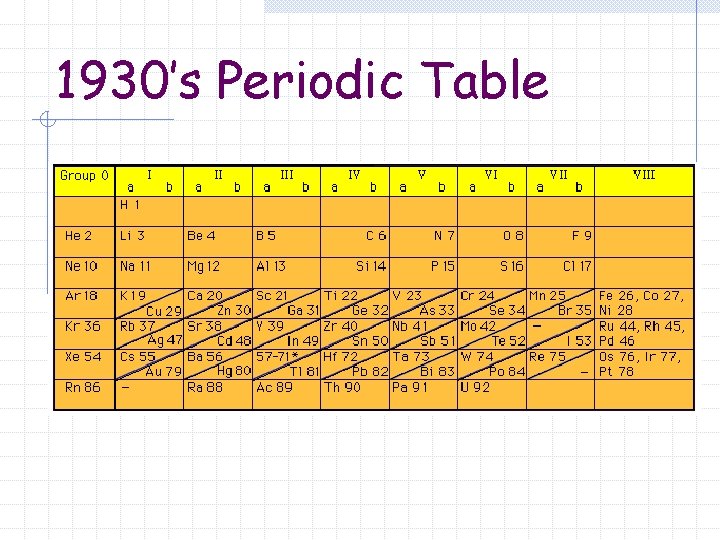
1930’s Periodic Table
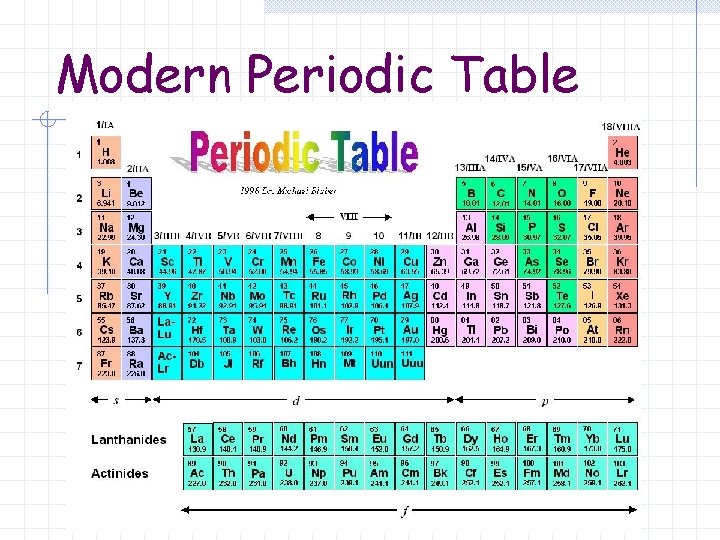
Modern Periodic Table

Group or family A vertical column of elements in the periodic table 18 groups Elements in the same group have similar chemical and physical properties
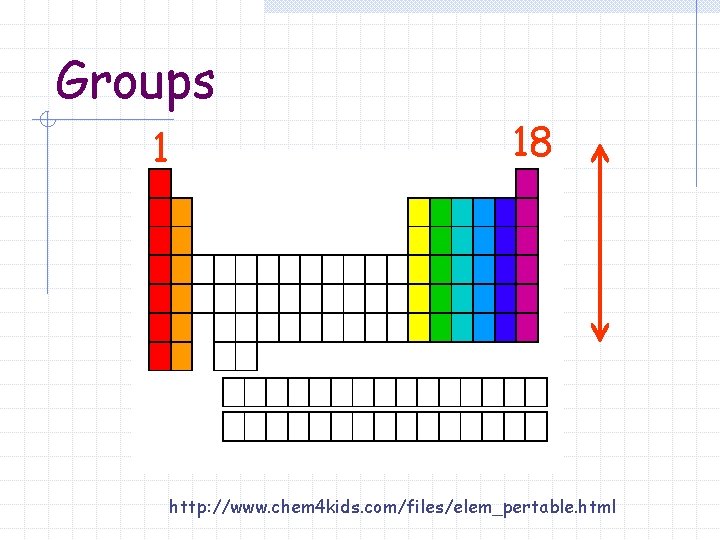
Groups 1 18 http: //www. chem 4 kids. com/files/elem_pertable. html

Period A horizontal row of elements in the periodic table 7 periods
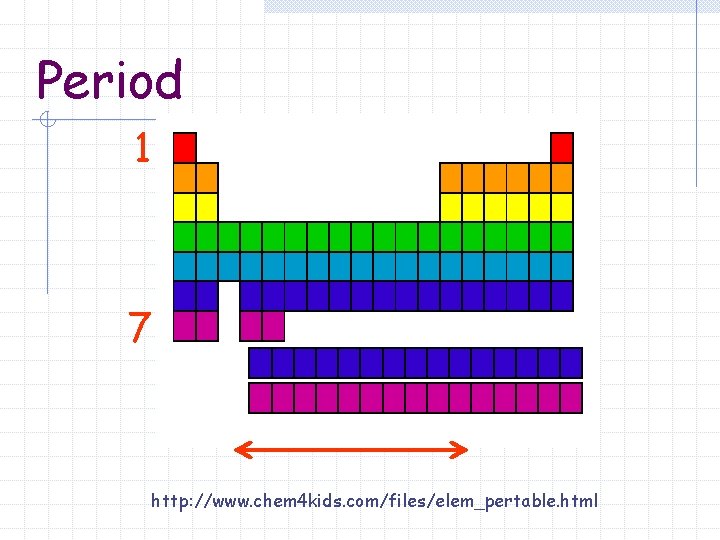
Period 1 7 http: //www. chem 4 kids. com/files/elem_pertable. html

Classes of Elements Metals Nonmetals Metalloids

Properties of Metals Lustrous (shiny) Malleable (pound into sheets) Ductile (drawn into wires)

Properties of Metals Good Conductors (heat / electricity) Solids at room temperature n Exception: Mercury (liquid)

Properties of Nonmetals Not lustrous (not shiny) Poor conductors (heat and electricity) At room temperature, most are brittle solids or gases. One element, bromine, is a liquid.

Properties of Metalloids Properties of both metals and nonmetals Metalloids are semiconductors.

Element Classification Elements classified into 4 categories based on their electron configurations Noble Gases n Representative Elements (A) n Transition Metals (B) n Rare Earth Elements (La & Ac rows) n
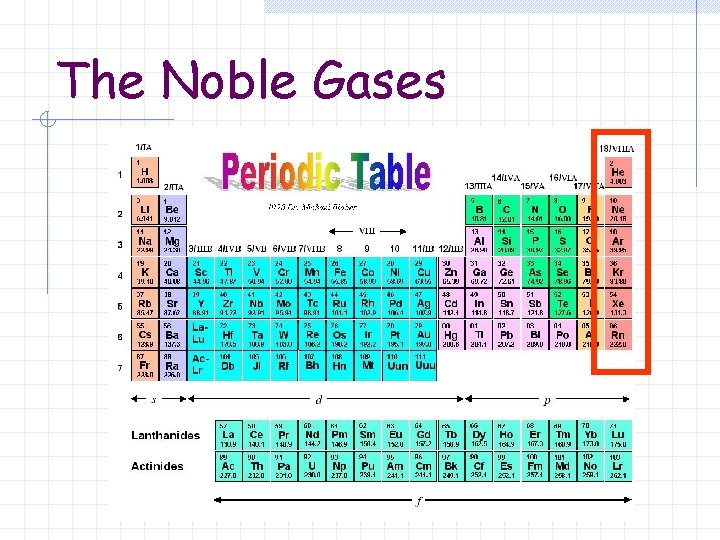
The Noble Gases

The Noble Gases Elements in which outermost electron levels are filled Sometimes referred to as Group 0, 8 A or 18 Sometimes called inert gases
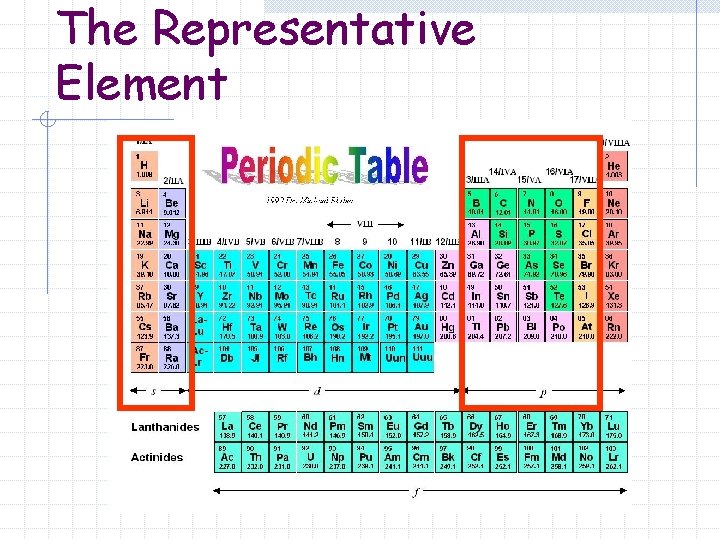
The Representative Element

The Representative Elements in which the outermost electron levels are only partially filled Sometimes called Group A elements

The Representative Elements Some groups have special names Group 1 A (1): Alkali Metals n Group 2 A (2): Alkaline Earth Metals n Group 6 A (16): Chalcogens n Group 7 A (17): Halogens n
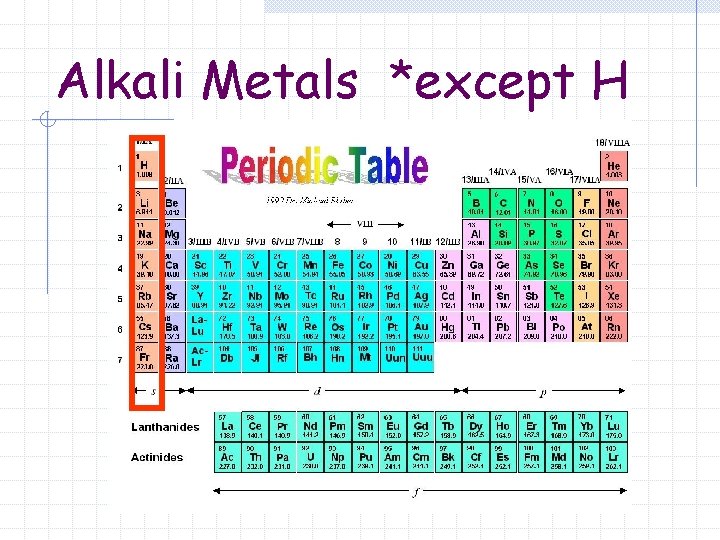
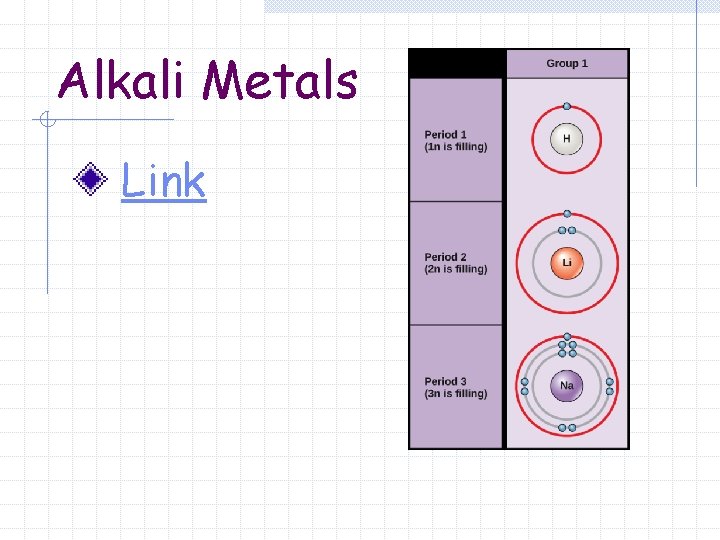
Alkali Metals *except H

Alkali Metals Group 1 A (1) of the Periodic Table Elements contain 1 valence electron in the outermost s sublevel.
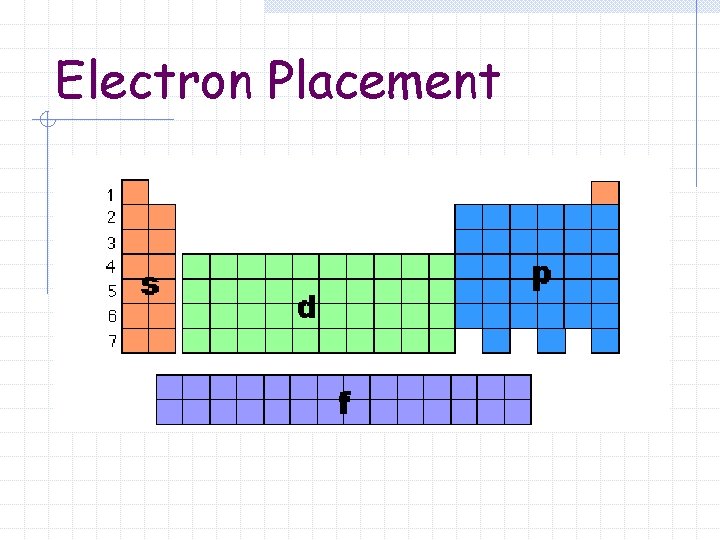
Electron Placement
Alkali Metals Link
Alkaline Earth Metals

Alkaline Earth Metals Group 2 A (2) of the Periodic Table Elements contain 2 valence electrons in the outermost s sublevel.
Halogens

Halogens Group 7 A (17) on the Periodic Table Elements contain 5 valence electrons in the p sublevel and 2 in the s sublevel. Total of 7 valence electrons.
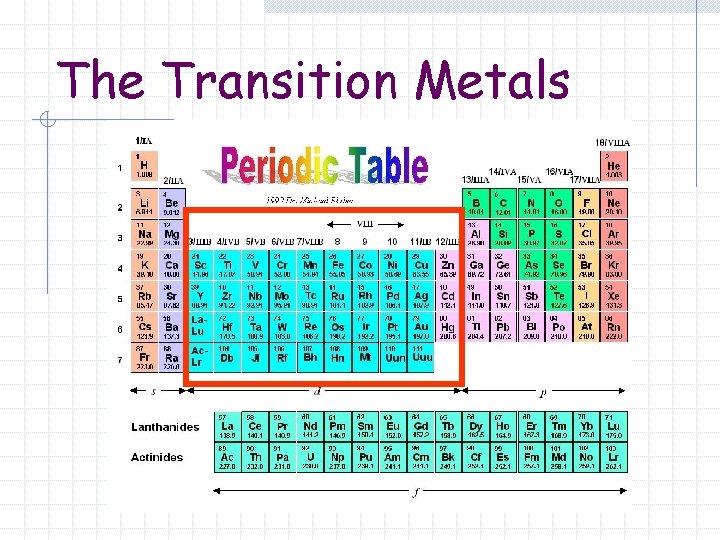
The Transition Metals

The Transition Metals Metallic elements in which the outer most s sublevel is filled and nearby d sublevel contains electrons. Part of the Group B elements
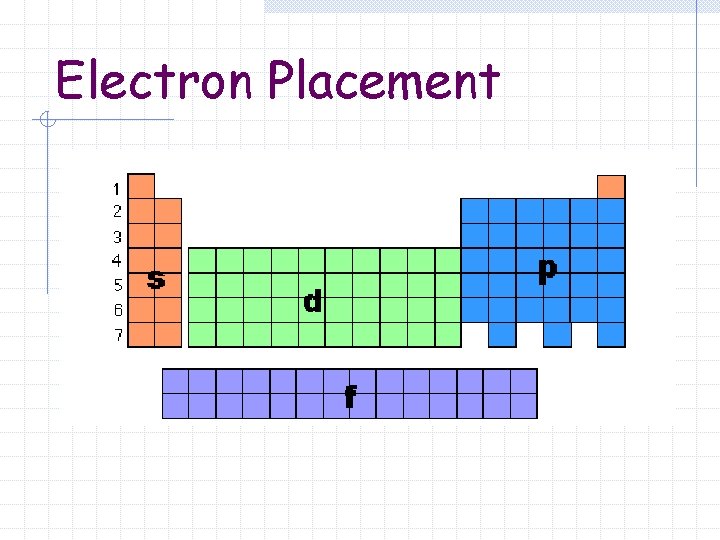
Electron Placement
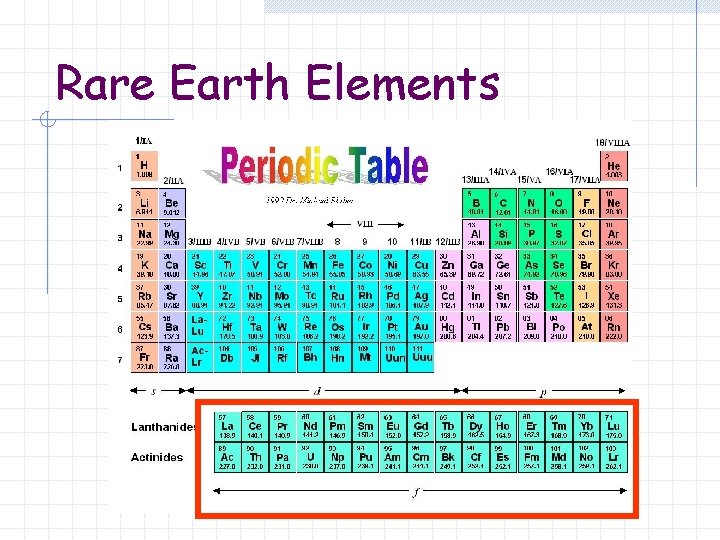
Rare Earth Elements

Rare Earth Elements Sometimes called Inner Transition Metals or Lanthanides and Actinides Metallic elements in which the outermost s sublevel is filled and nearby f sublevel contains electrons

The Inner Transition Metals Part of the Group B elements Moved to the bottom of the Periodic Table to save space



Objective Review the atomic structure of the atom.

Quantum Mechanical Model Erwin Schrodinger – 1926 Mathematical solution to Schrodinger’s Equation
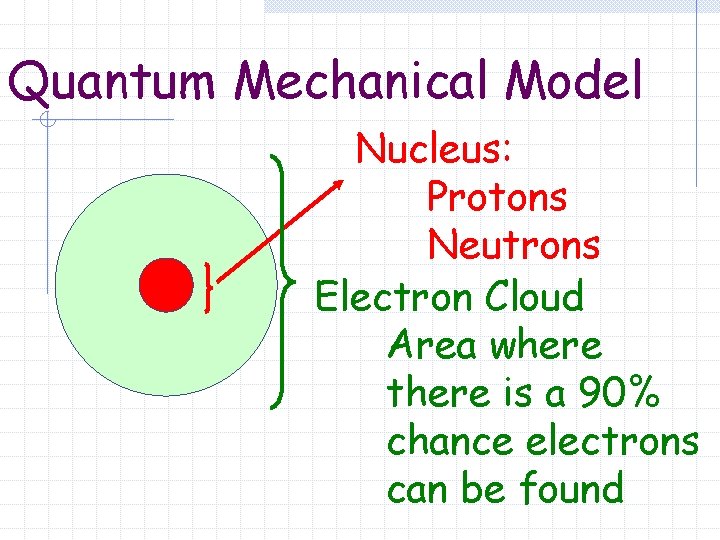
Quantum Mechanical Model Nucleus: Protons Neutrons Electron Cloud Area where there is a 90% chance electrons can be found

Subatomic Particles Subatomic Relative Particle Charge Mass Proton neutron electron 1+ 1 amu 0 1 amu 1 - 0 amu Location nucleus electron cloud

Objective Define atomic radius and account for the trend in atomic radii for elements within a group and period in the periodic table.

Atomic Radius One-half of the distance between the nuclei in a molecule consisting of identical atoms.
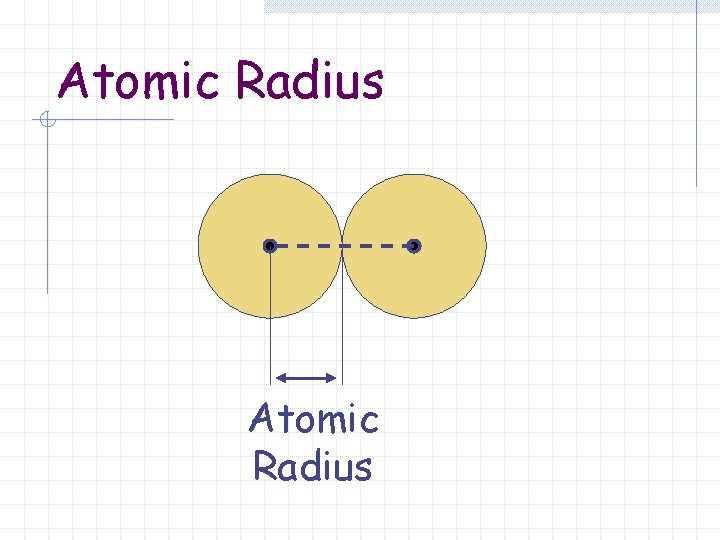
Atomic Radius
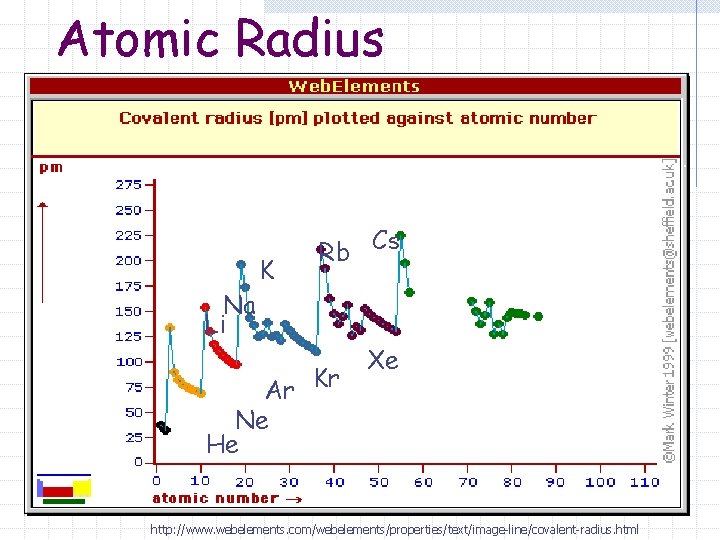
Atomic Radius Na Li K Rb Cs Ar Kr Ne He Xe http: //www. webelements. com/webelements/properties/text/image-line/covalent-radius. html
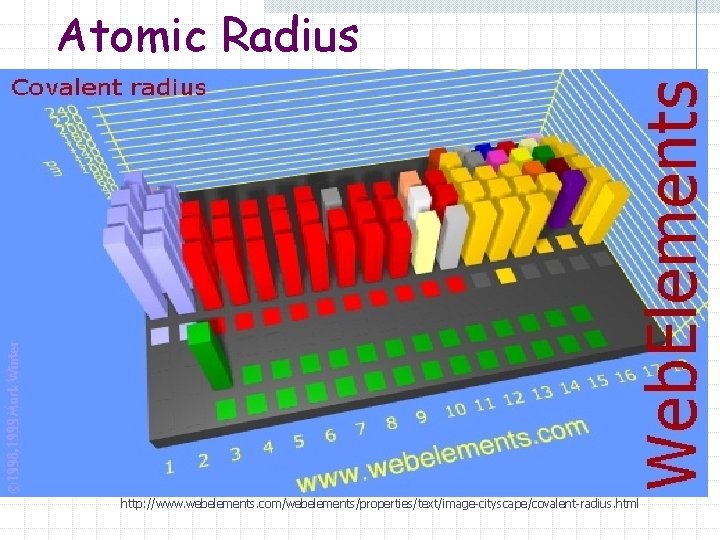
Atomic Radius http: //www. webelements. com/webelements/properties/text/image-cityscape/covalent-radius. html
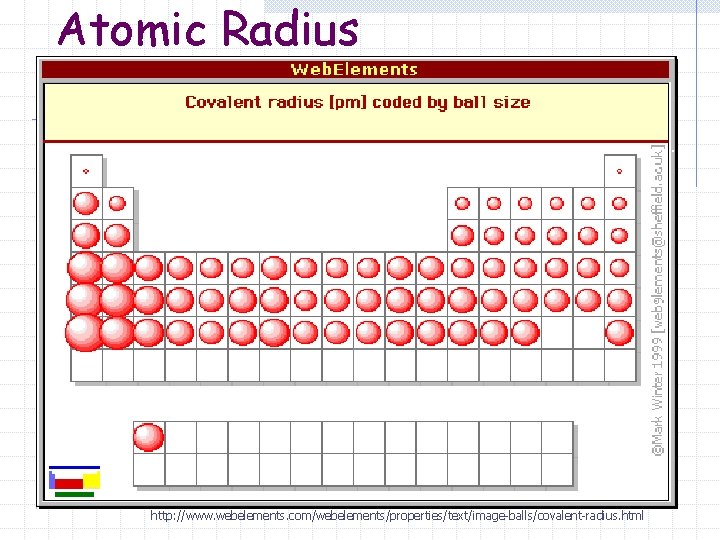
Atomic Radius http: //www. webelements. com/webelements/properties/text/image-balls/covalent-radius. html


Atomic Radius (Period) As you go across a period you add more electrons to the SAME energy level As you go across the period you are also adding more protons to the nucleus

Atomic Radius (Period) Additional protons in the nucleus create a higher “effective nuclear charge. ”

Atomic Radius (Period) The higher “effective nuclear charge” means that there is a stronger force pulling the electrons toward the nucleus (like a magnet).

Atomic Radius Trend (Period) Atomic radius decreases (it gets smaller) as you go from left to right on the periodic table.
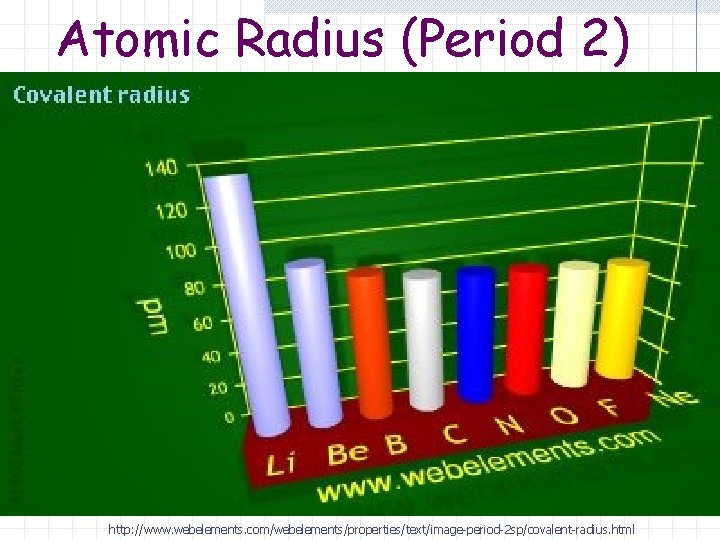
Atomic Radius (Period 2) http: //www. webelements. com/webelements/properties/text/image-period-2 sp/covalent-radius. html
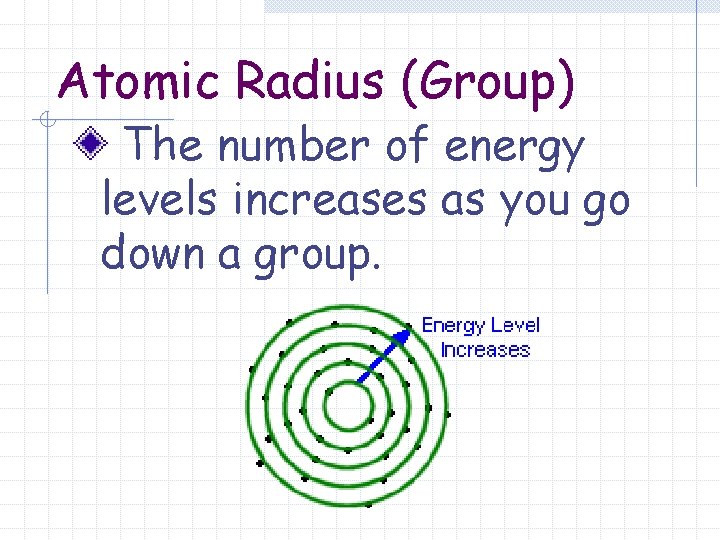
Atomic Radius (Group) The number of energy levels increases as you go down a group.

Atomic Radius (Group) Each additional energy level is further from the nucleus.

Atomic Radius (Group) As you go down a group, the number of protons in the nucleus also increases and so does the “effective nuclear charge. ”

Atomic Radius (Group) Electrons in the inner energy levels “shield” those electrons on the outer energy levels. (shielding effect)

Atomic Radius Trend (Group) Atomic radius increases (it gets larger) as you go down a group.
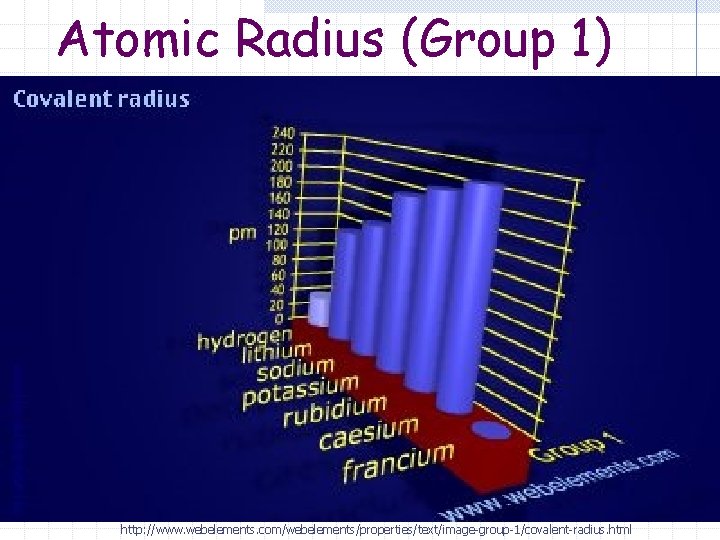
Atomic Radius (Group 1) http: //www. webelements. com/webelements/properties/text/image-group-1/covalent-radius. html

Circle the Element with the Larger Atomic Radius P or S? n P B or Al? n Al N, O, P or S? n P

Circle the Element with the Larger Atomic Radius Rb or Cs? n Cs Na or K? n K Li, Be, Na, or Mg? n Na
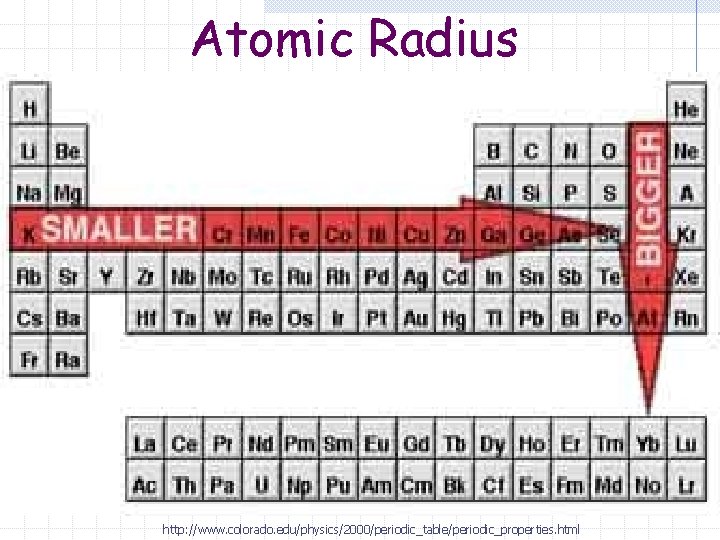
Atomic Radius http: //www. colorado. edu/physics/2000/periodic_table/periodic_properties. html

Objective Define ionization energy and account for the trend in ionization energy within a group and period in the periodic table.

Objective Distinguish and account for the differences between first, second, and third ionization energies.

Ion and Ionization: Any process that results in the formation of an ion. Ion: An atom or group of atoms that has a positive or negative charge.

Ionization Energy The energy required to remove an electron from an atom.

Ionization Energy Removing an electron results in the formation of a cation (positively charged ion). “cat-eye-on”
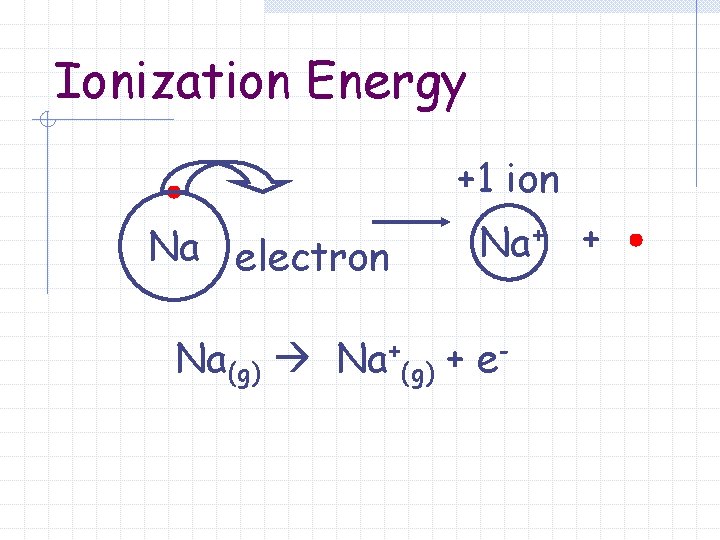
Ionization Energy +1 ion Na electron Na+ + Na(g) Na+(g) + e-

1 st Ionization Energy required to remove the first outermost electron.
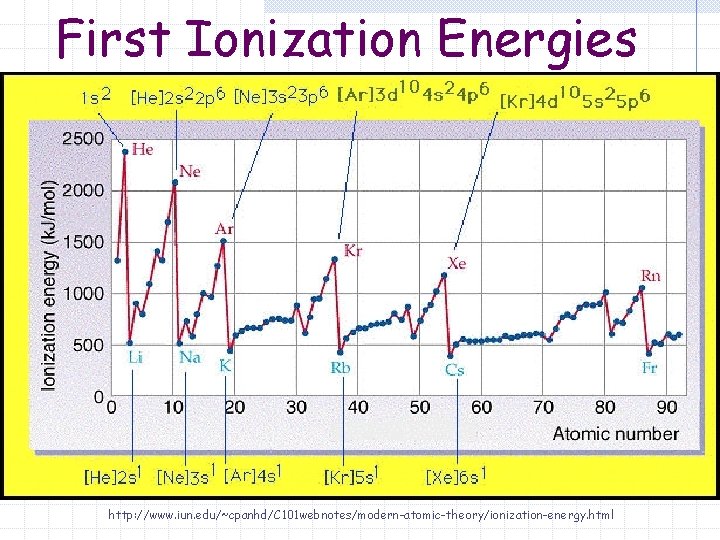
First Ionization Energies http: //www. iun. edu/~cpanhd/C 101 webnotes/modern-atomic-theory/ionization-energy. html

First Ionization Energies (Group) As you go down a group electrons are added to energy levels that are further from the nucleus

First Ionization Energies (Group) Outer energy level electrons are also “shielded” from the full affect of the effective nuclear charge by the inner energy level electrons.

First Ionization Energies (Group) As you go down a group the first ionization energy decreases (gets smaller). Less energy is required to remove electrons from an element as you go down a group.

First Ionization Energies (Period) The effective nuclear charge increases as you go across the period (left to right). Electrons are held tighter by the nucleus and require more energy to be removed.

First Ionization Energies (Period) First Ionization Energy increases (gets bigger) as you go left to right across a period.
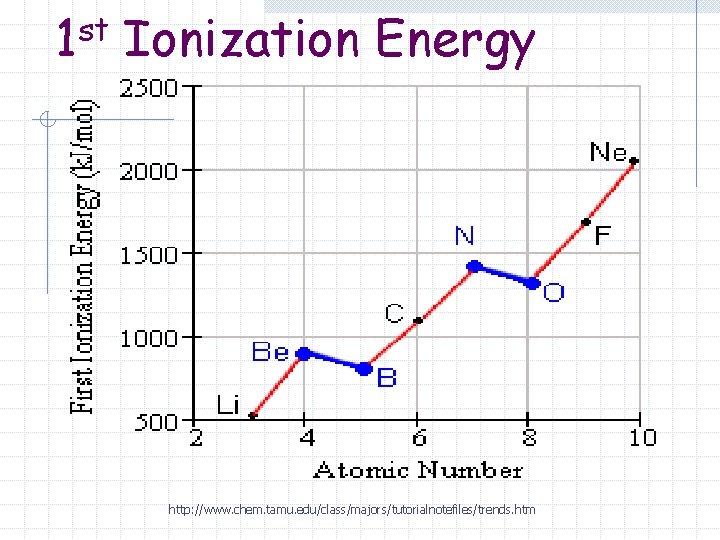
1 st Ionization Energy http: //www. chem. tamu. edu/class/majors/tutorialnotefiles/trends. htm

nd 2 Ionization Energy required to remove a second electron from the outermost electrons.

Third Ionization Energy required to remove a third electron from the outermost electrons.

Ionization Energies Once an electron is removed from an atom, the nucleus holds onto the other electrons tighter. It requires more energy to remove additional electrons.

Ionization Energies st 1 IE < nd 2 IE < rd 3 IE

Circle the Element with the Larger Ionization Energy P or S? n S B or Al? n B N, O, P or S? n O

Circle the Element with the Larger Ionization Energy Rb or Cs? n Rb Na or K? n Na Li, Be, Na, or Mg? n Be

Objective Define electronegativity and state the group and period trend in electronegativity.

Electronegativity The tendency for an atom to attract electrons to itself when it is chemically combined with another element.
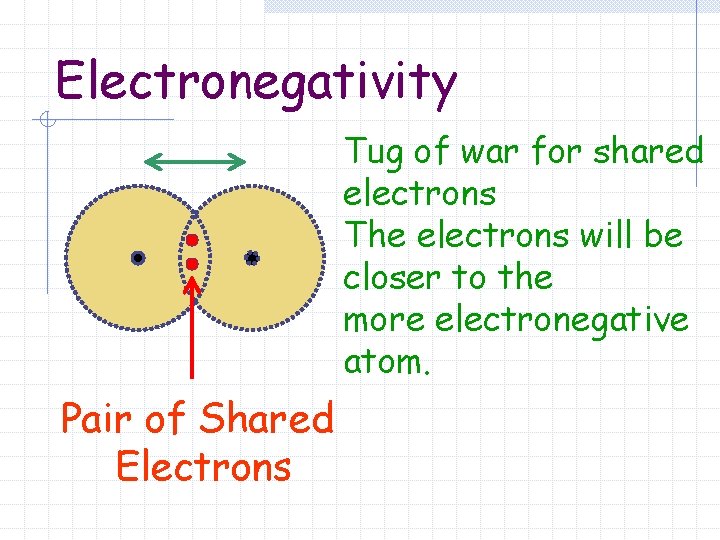
Electronegativity Tug of war for shared electrons The electrons will be closer to the more electronegative atom. Pair of Shared Electrons

Electronegativity Fluorine is the most electronegative element – it is assigned a value of 4. 0 All other elements are compared to fluorine on this relative scale.
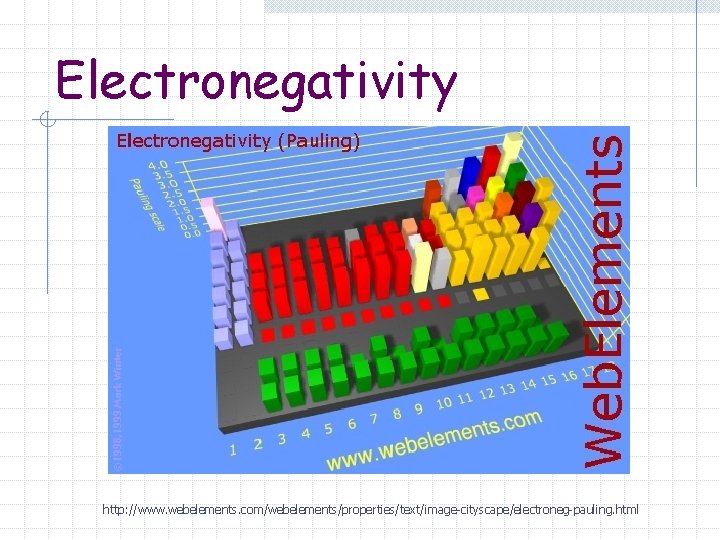
Electronegativity http: //www. webelements. com/webelements/properties/text/image-cityscape/electroneg-pauling. html

Electronegativity Noble gases are often omitted because they tend not to form compounds.
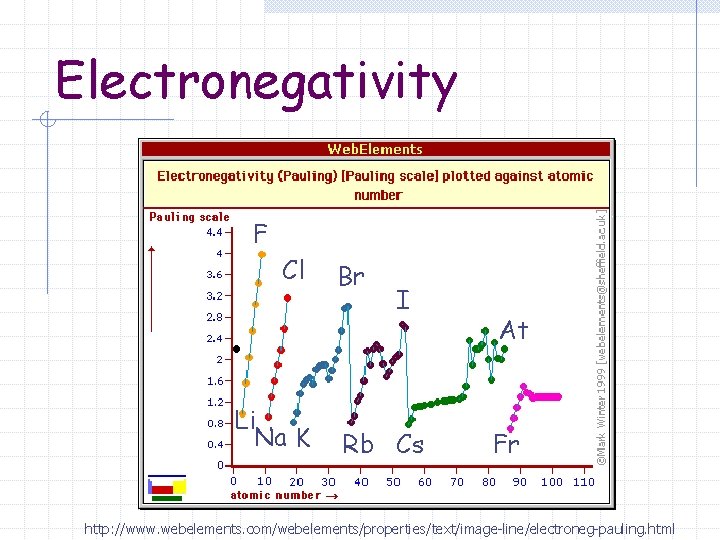
Electronegativity F Cl Li Na K Br I Rb Cs At Fr http: //www. webelements. com/webelements/properties/text/image-line/electroneg-pauling. html

Electronegativity http: //www. webelements. com/webelements/properties/text/image-balls/electroneg-pauling. html

Electronegativity (Group) As you go down a group electrons are added to energy levels that are further from the nucleus

Electronegativity (Group) Outer energy level electrons are also “shielded” from the full affect of the effective nuclear charge by the inner energy level electrons.

Electronegativity (Group) As you go down a group the electronegativity decreases (gets smaller) The nucleus has less of an pull or attraction on the electrons shared in a chemical bond.
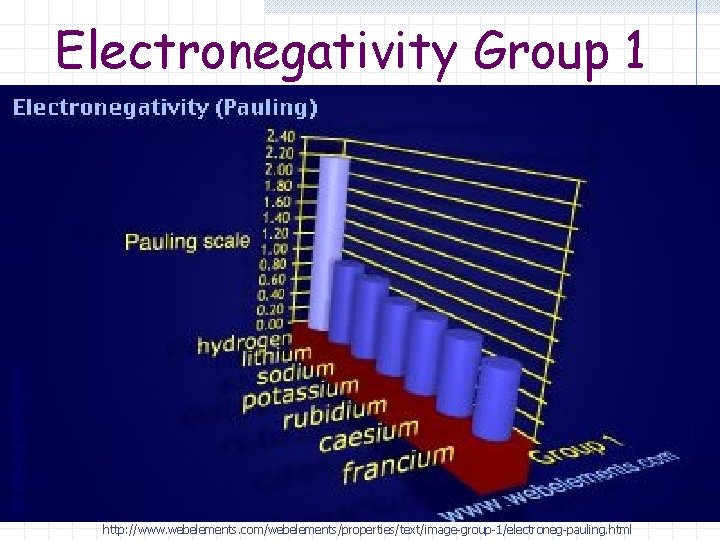
Electronegativity Group 1 http: //www. webelements. com/webelements/properties/text/image-group-1/electroneg-pauling. html

Electronegativity (Period) The effective nuclear charge increases as you go across the period (left to right) Electrons are held tighter and pulled closer to the nucleus.

Electronegativity (Period) Electronegativity increases (gets bigger) as you go left to right across a period.
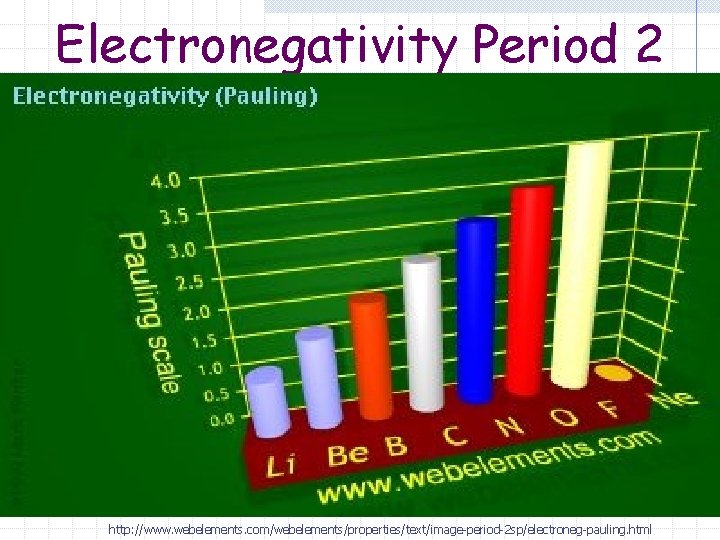
Electronegativity Period 2 http: //www. webelements. com/webelements/properties/text/image-period-2 sp/electroneg-pauling. html

Circle the Element with the Larger Electronegativity P or S? n S B or Al? n B N, O, P or S? n O

Circle the Element with the Larger Electronegativity Rb or Cs? n Rb Na or K? n Na Li, Be, Na, or Mg? n Be

Metallic Properties ØIncrease down a group. ØDecrease across a period (L R). Most metallic element- Fr

Objective Write the electron configurations of an element using the periodic table.

Electron The electron is the most important subatomic particle in determining physical and chemical properties.

Periodic Table The periodic table is arranged in a way that elements with the same number of “valence electrons” are in the same group.

Valence Electrons in the highest occupied energy level of an atom.

Blocks The periodic table can be divided into sections or blocks s-block n p-block n d-block n f-block n
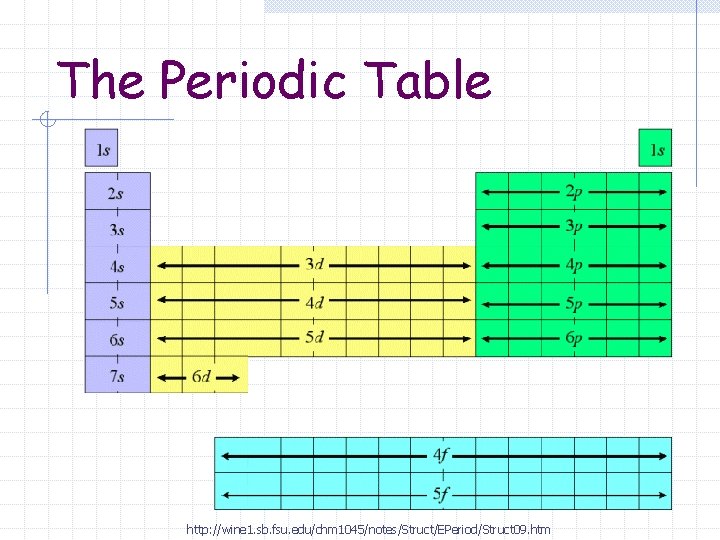
The Periodic Table http: //wine 1. sb. fsu. edu/chm 1045/notes/Struct/EPeriod/Struct 09. htm

Blocks s-block n Groups 1 – 2 p-block n Groups 13 – 18

Blocks d-block Groups 3 – 12 n Transition Metals n f-block Lanthanide and Actinide series n Inner Transition Metals n

Electron Configurations Using the periodic table to write electron configurations for elements.

Electron Configurations Read the periodic table as you would a book Left to Right n Top to Bottom n

Electron Configurations Write down all filled sublevels and stop at the element of interest

Shorthand Notation and Electron Configuration Use shorthand notation by writing the symbol of the previous noble gas and continue writing the electron configuration from there.

Electron Configurations P 1 st and 2 nd energy levels are full 1 s 2 2 p 6 n 3 s is also full 3 s 2 n 3 p is not, 3 squares in 3 p 3 n 1 s 2 2 p 6 3 s 2 3 p 3

Electron Configurations Use the periodic table to write electron configurations for the following elements.
![Electron Configuration P Shorthand Notation – start with previous noble gas [Ne] n Next Electron Configuration P Shorthand Notation – start with previous noble gas [Ne] n Next](http://slidetodoc.com/presentation_image_h2/0cc69d77022ce62c4b893fb3b0dce2c5/image-119.jpg)
Electron Configuration P Shorthand Notation – start with previous noble gas [Ne] n Next period is 3 – 3 s is full and 3 blocks into 3 p 3 s 2 3 p 3 n [Ne] 2 3 s 3 3 p

Electron Configuration Mn Look at the periodic table n all of 1 s, 1 p, 2 s, 2 p, 3 s and 3 p are full takes you up to Ar n 4 s is full and then we are 5 squares into 4 d n
![Electron Configuration Mn [Ar] 2 4 s 5 3 d Electron Configuration Mn [Ar] 2 4 s 5 3 d](http://slidetodoc.com/presentation_image_h2/0cc69d77022ce62c4b893fb3b0dce2c5/image-121.jpg)
Electron Configuration Mn [Ar] 2 4 s 5 3 d

Review Atomic Structure Electron Configuration

Lithium (Li) atomic number: ____ number of electrons: ___ group number: ___ period number: ___ group name (if any): ________

Lithium (Li) electron configuration:

Argon (Ar) atomic number: ____ number of electrons: ___ group number: ___ period number: ___ group name (if any): ________

Argon (Ar) electron configuration:

Titanium atomic number: ____ number of electrons: ___ group number: ___ period number: ___ group name (if any): ________

Titanium electron configuration:
- Slides: 128