Unit 2 Notes 2 Atomic Symbols protons mass
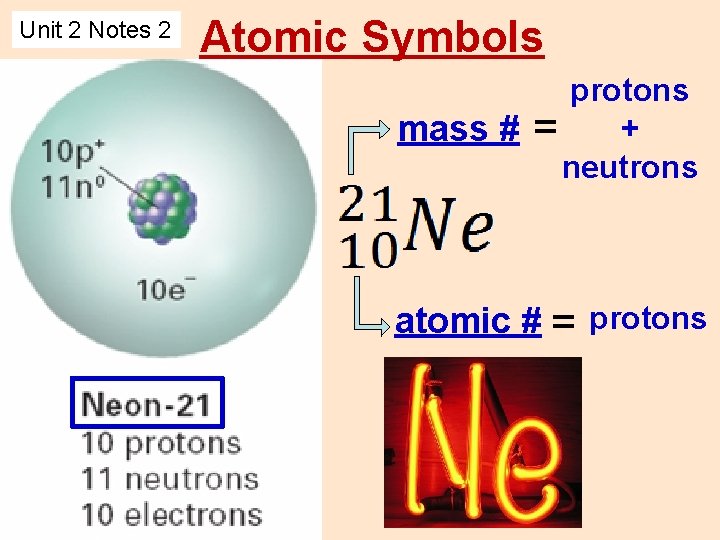
Unit 2 Notes 2 Atomic Symbols protons + mass # = neutrons atomic # = protons
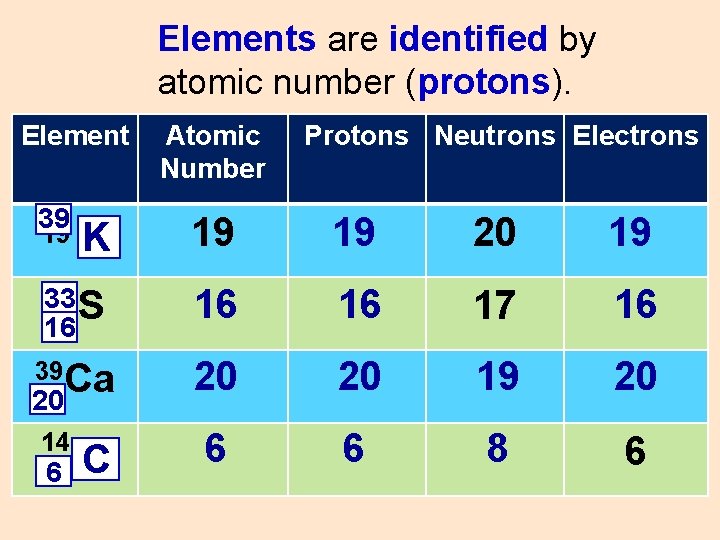
Elements are identified by atomic number (protons). Element 39 Atomic Number Protons Neutrons Electrons K 19 19 20 19 33 S 16 16 16 17 16 39 Ca 20 20 19 20 6 6 8 6 19 20 14 6 C
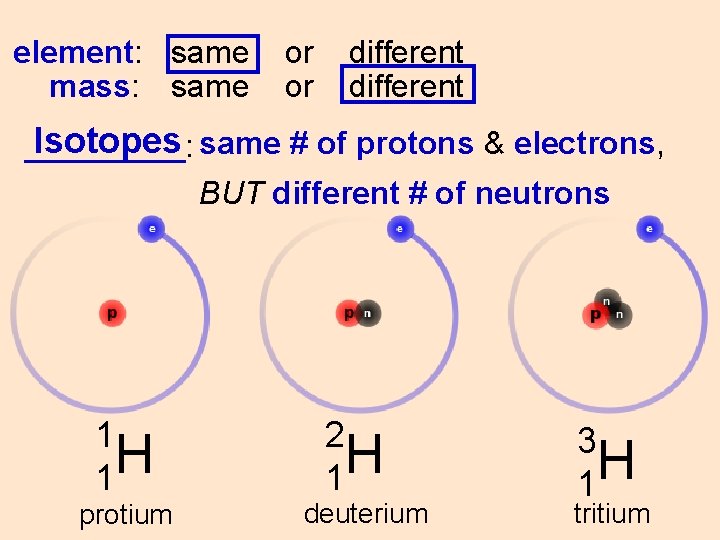
element: same mass: same or or different Isotopes same # of protons & electrons, _____: BUT different # of neutrons 1 H 1 protium 2 H 1 deuterium 3 H 1 tritium

Quick (but challenging) Quiz! 1. The number of protons in the nucleus is… A. the mass number. B. the atomic number. C. the same as the number of neutrons. D. the total of protons and neutrons.

Quick (but challenging) Quiz. 2. The mass number is… A. the number of protons. B. the number of neutrons. C. the total of protons and electrons. D. the total of protons and neutrons.

Quick (but challenging) Quiz. 3. Isotopes of an element have… A. the same mass number. B. different atomic numbers. C. the same number of protons but different mass numbers. D. the same number of protons but different numbers of neutrons.

Quick (but challenging) Quiz. 4. How many neutrons are in an atom of potassium– 40 ? (40 K) (Atomic Number = 19) A. 19 neutrons B. 21 neutrons C. 40 neutrons D. 59 neutrons

Average Atomic Mass If there are 2 naturally occurring isotopes of Neon including Ne-20 and Ne-22. then the average mass of Neon atoms should be 21 amu. ____ Why does the PT lists it as 20. 18 amu? Calculated as a weighted average of atoms in a sample of naturally occurring isotopes.

Average Atomic Mass A weighted average atomic mass is calculated from both the a. masses of each isotope and the b. relative abundance (%) of each isotope. 63 Cu is 69% abundant 65 Cu is 31% abundant To which will the average mass be closer, 63 or 65?

copper-63 has a mass of 62. 93 amu and natural abundance of 69. 15% copper-65 has a mass of 64. 93 amu and has a natural abundance of 30. 85% Calculating Average Atomic Mass Avg. Mass = (Mass 1)(%) + (Mass 2)(%) … = (62. 93)(0. 6915) + (64. 93)(0. 3085) = 63. 55

a. neon-20 neon-22 b. Which isotope is in greater relative abundance in natural samples of neon? because… the weighted average atomic mass of neon from the PT is 20. 18 amu.

Quick Quiz! 1. If an unusually selected sample of sulfur contained 90. 0% sulfur-32 and 10. 0% sulfur-34, what would be its average atomic mass? A. 32. 2 amu B. 32. 9 amu C. 33. 5 amu D. 34. 2 amu Avg. = (M 1)(%) + (M 2)(%) = (32)(0. 900) + (34)(0. 100) = 32. 2

- Slides: 13