Unit 2 Metabolic Processes Metabolism and Energy SBI

Unit 2: Metabolic Processes Metabolism and Energy SBI 4 U

Thermodynamics • All cells need a constant supply of energy in order to perform cellular processes • We obtain energy from food and drinks, and convert that into useful energy through a series of chemical reactions

Metabolism - Metabolism is the sum of all anabolic (constructive) and catabolic (deconstructive) reactions that occur

Bond Energy • When chemical bonds are formed between two atoms, energy is released. • The amount of energy needed to break a bond is the same as the energy released when bonds are formed, also known as bond energy.
Metabolism and Energy • Energy is the ability to do work • Energy can exist in the form of light, sound, heat, mechanical, etc. • All forms of energy can exist as potential (stored) or kinetic (motion) energy
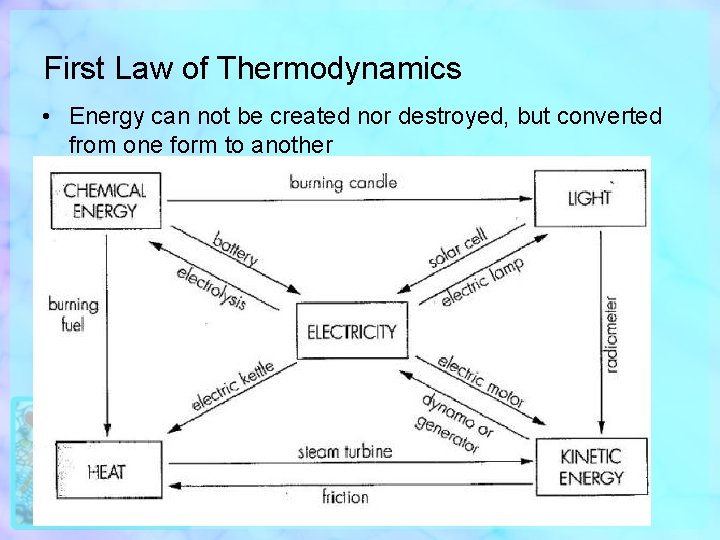
First Law of Thermodynamics • Energy can not be created nor destroyed, but converted from one form to another

Enthalpy - Enthalpy (H) is the amount of energy in a substance - ΔH = the amount of energy that is absorbed or released when a reaction occurs under constant pressure - ΔH = Hproduct - Hreactant
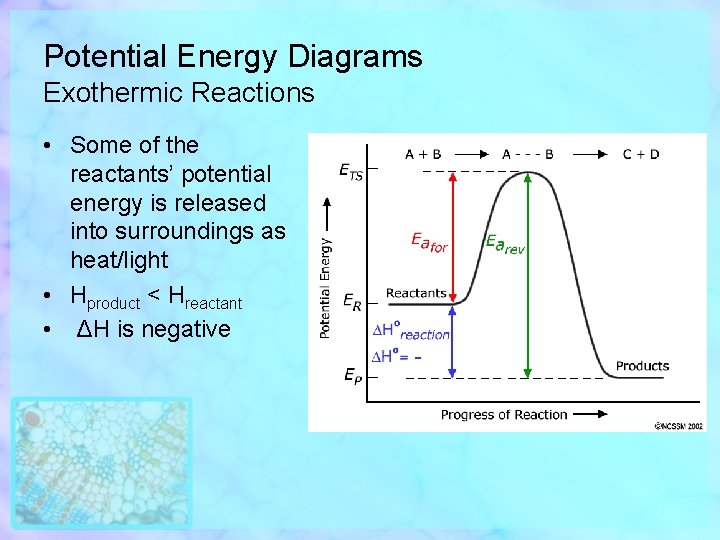
Potential Energy Diagrams Exothermic Reactions • Some of the reactants’ potential energy is released into surroundings as heat/light • Hproduct < Hreactant • ΔH is negative
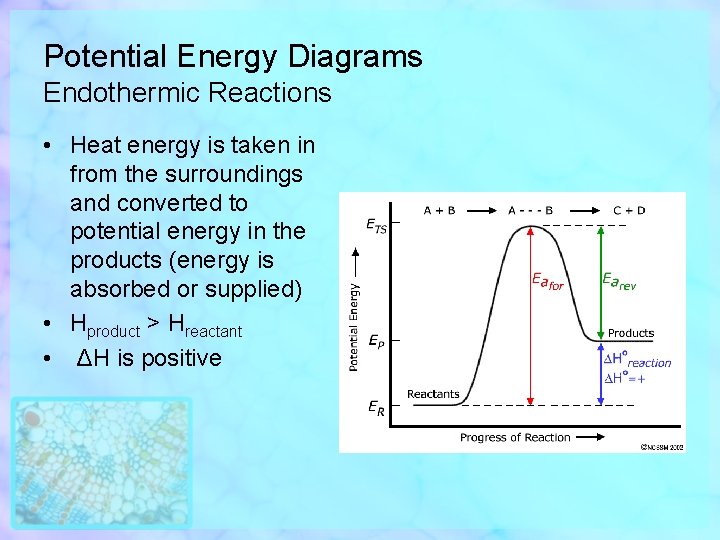
Potential Energy Diagrams Endothermic Reactions • Heat energy is taken in from the surroundings and converted to potential energy in the products (energy is absorbed or supplied) • Hproduct > Hreactant • ΔH is positive

Entropy • Entropy (S) is a measure of the amount of randomness/disorder in a system

Second Law of Thermodynamics - Entropy is constantly increasing. - Energy is spontaneously converted to a less ordered form

Entropy • Entropy can never be a negative value • The change in entropy (ΔS) during a chemical reaction can be positive (increase in entropy) or negative (decrease in entropy) • ΔS = Sfinal - Sinitial

Entropy • Entropy will increase when: • solid changes to liquid or gas • liquid changes to gas • fewer moles of reactant converted to more moles of product • complex molecules broken down to simpler molecules • diffusion from high concentration to low concentration • temperature increases

Examples • • • Steam Water decrease Solid CO 2 sublimes to gas increase N 2 O 4(g) 2 NO 2(g) increase H 2 O heated from 25⁰C to 50⁰C increase C 6 H 6(l) + O 2(g) 6 CO 2(g) + 3 H 2 O(l) increase

Predicting Reactions - Spontaneous reactions occur without outside influence - Two factors allow us to predict if a reaction will occur - Enthalpy (must be exothermic) OR - Entropy (must be increasing)

Gibb’s Free Energy • When reactions are endothermic and increasing entropy, we use Gibb’s free energy • ΔG = ΔH – TΔS • When free energy increases, a reaction is endergonic • When free energy decreases, a reaction is exergonic • When: • ΔG < 0, spontaneous, exergonic reaction • ΔG > 0, not spontaneous, endergonic reaction • ΔG = 0, reaction is at equilibrium
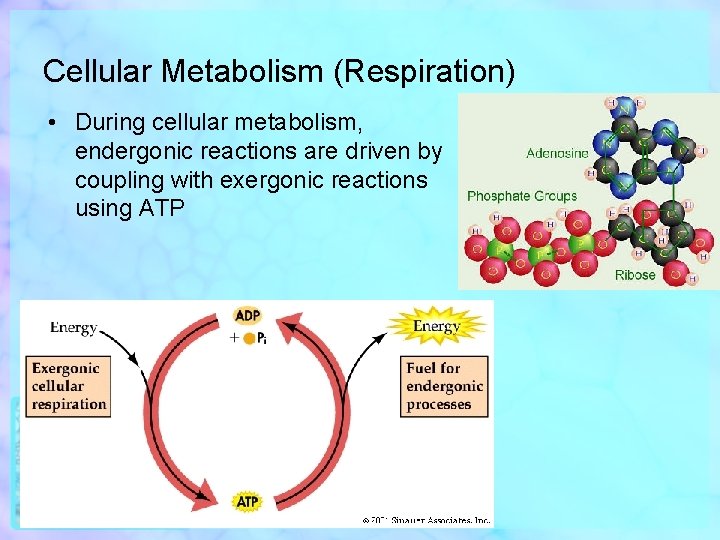
Cellular Metabolism (Respiration) • During cellular metabolism, endergonic reactions are driven by coupling with exergonic reactions using ATP

Anabolic Reaction Catabolic Reaction Photosynthesis - creates order - free energy increases Reactions in the sun - emits light - decreases order ATP Production - decreases entropy - free energy increases Cellular Respiration - disorder increases - free energy decreases Metabolic Reactions

Electron Carriers • Redox reactions are coupled reactions • The substance that accepts electrons is reduced • The substance that donates electrons is oxidized • The reduced form always has more energy, so electrons carry reducing power

Electron Carriers • Electron carriers are compounds that pick up electrons from high energy compounds and delivers them to low energy compounds • NAD+ (nicotinamide adenine dinucleotide) + e- NADH • FAD (flavin adenine dinucleotide) + e- FADH 2
- Slides: 20