Unit 2 Metabolic Processes Electron Transport and Chemiosmosis

Unit 2: Metabolic Processes Electron Transport and Chemiosmosis SBI 4 U –

Electron Transport Chain (ETC) • Location: • Intermembrane proteins in the mitochondria • Purpose: • To release the stored energy in NADH and FADH 2 and use it to make ATP
ETC - Main Reactants: 1. NADH 2. FADH 2 3. O 2

ETC - Main Products: 1. ATP 2. H 2 O
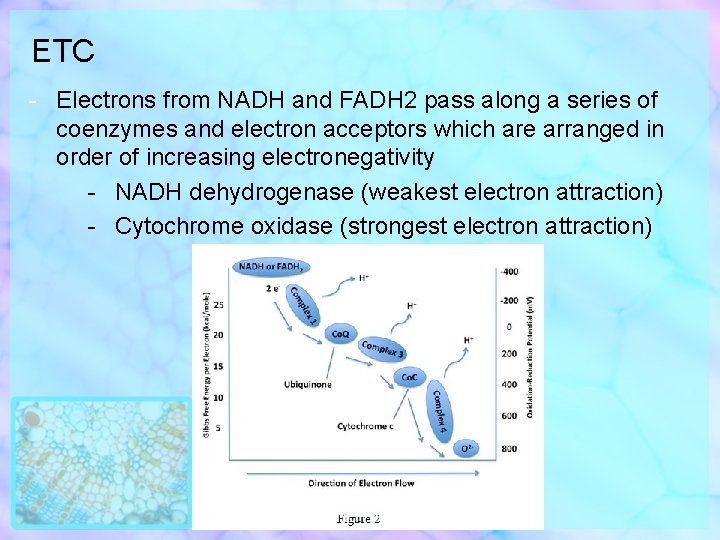
ETC - Electrons from NADH and FADH 2 pass along a series of coenzymes and electron acceptors which are arranged in order of increasing electronegativity - NADH dehydrogenase (weakest electron attraction) - Cytochrome oxidase (strongest electron attraction)

ETC - Electron carriers alternate between reduced and oxidized forms while electrons pass through each complex - Electrons from NADH pass through 3 major complexes - Energy from the transfer of electrons is used to pump 3 H+ out of the matrix - Electrons from FADH 2 pass through 2 major complexes - Energy used to pump 2 H+ out of matrix - An electrochemical (proton) gradient forms across the membrane, with high [H+] in intermembrane space
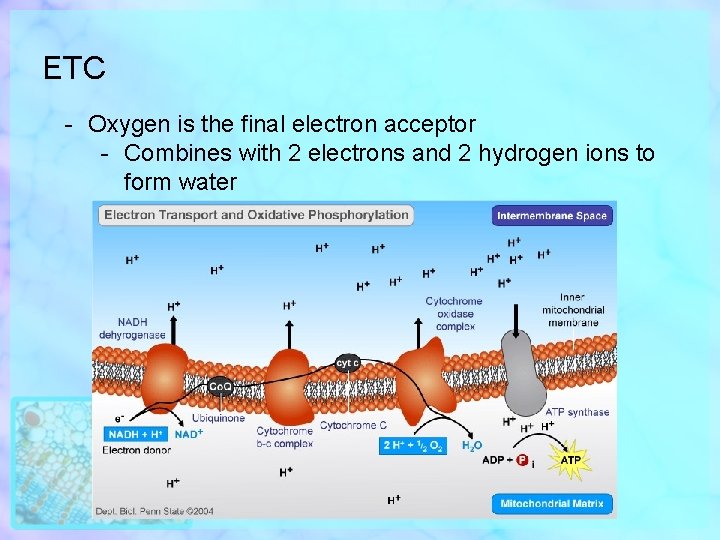
ETC - Oxygen is the final electron acceptor - Combines with 2 electrons and 2 hydrogen ions to form water
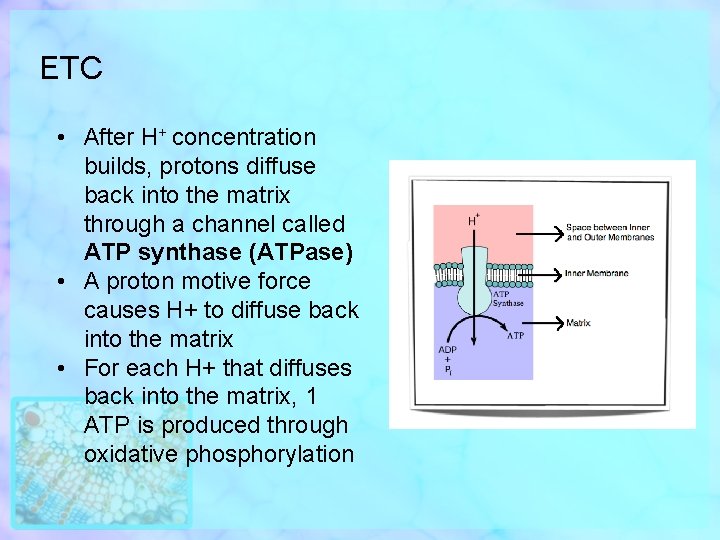
ETC • After H+ concentration builds, protons diffuse back into the matrix through a channel called ATP synthase (ATPase) • A proton motive force causes H+ to diffuse back into the matrix • For each H+ that diffuses back into the matrix, 1 ATP is produced through oxidative phosphorylation

Chemiosmosis - Process that uses the energy from proton gradients to drive the phosphorylation of ADP to form ATP

ATP Yield • Theoretical Yield: • Prokaryotes: 38 ATP • Eukaryotes: 36 ATP • Actual yield often much lower • Some protons are lost through the membrane without passing through ATPase • Some energy from proton gradient used to transport pyruvate • Some energy used to transport ATP out of mitochondria
- Slides: 10