Unit 2 Biochemistry p H and Water Water

Unit 2 - Biochemistry
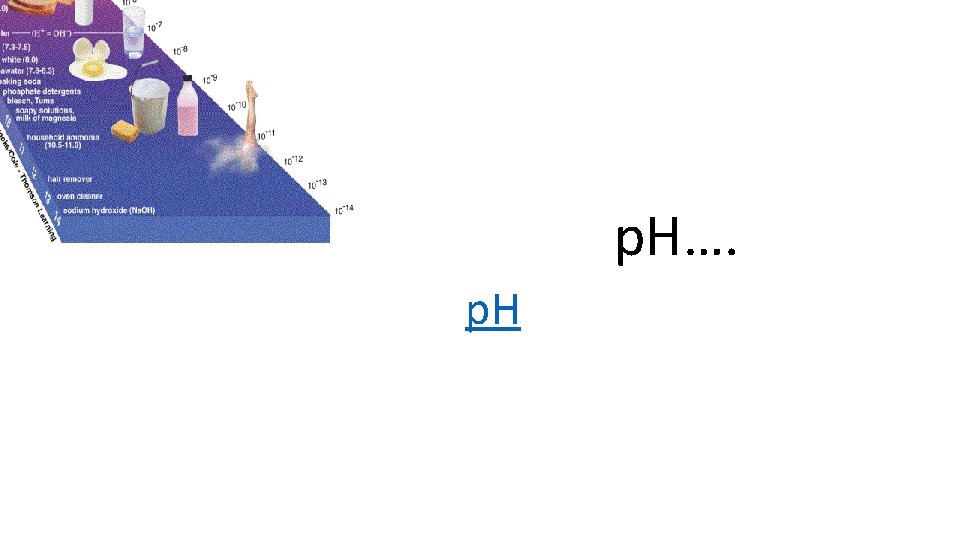
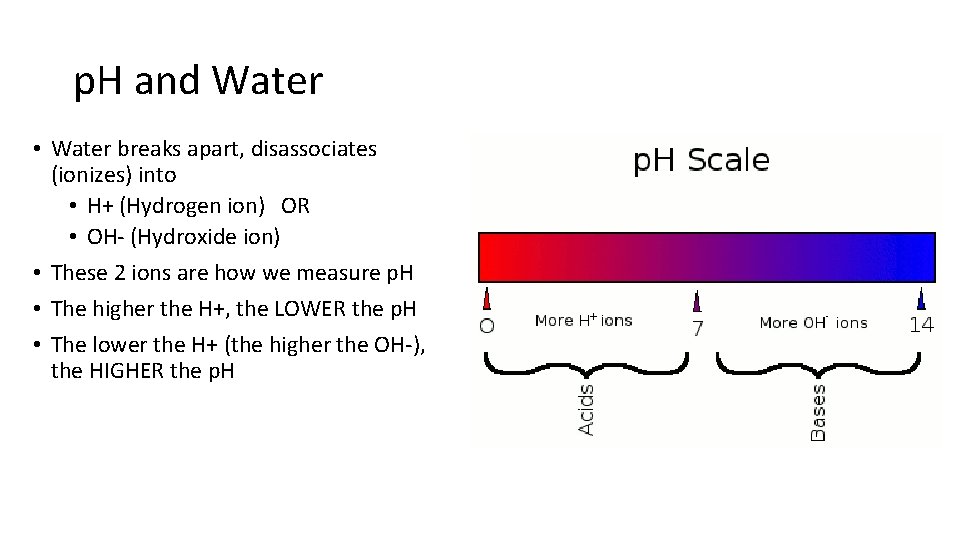
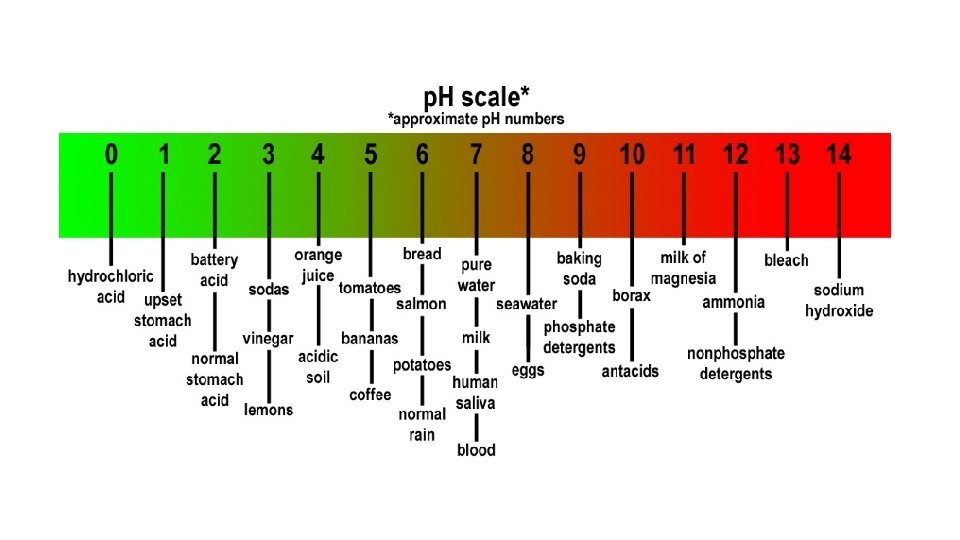
p. H and Water • Water breaks apart, disassociates (ionizes) into • H+ (Hydrogen ion) OR • OH- (Hydroxide ion) • These 2 ions are how we measure p. H • The higher the H+, the LOWER the p. H • The lower the H+ (the higher the OH-), the HIGHER the p. H

WATER!!! • Properties…. Cohesion • Adhesion • Capillary action • Hydrogen bonding… • Properties of Water Video

Organic vs Inorganic Compounds • Organic Compounds Contain • Inorganic Compounds do NOT contain Carbon • Organic Chemistry – the branch of science devoted to the study of Carbon and it’s compounds

Why is Carbon So Important? ● Most all living things are comprised of Hydrogen, Oxygen, Nitrogen, and Carbon. H, O, N, C. (think HONK!!) ● Carbon can form strong covalent bonds, meaning electrons are shared between atoms. ● Carbon can make four bonds with other atoms.

Organic Compounds • Monomers • Single unit • Molecule • Polymers • Many units strung together • Multiple monomers

Types of Organic Compounds found in the body • Carbohydrates • Lipids • Proteins • Nucleic Acids

Carbohydrates • The most important function of carbohydrates is to create energy. • The 4 types of Carbohydrates are sugars, starches, cellulose, and glycogen • Simple sugars are: • Glucose and fructose – these are each monosaccharides – meaning they are made of 1 carbohydrate molecule.
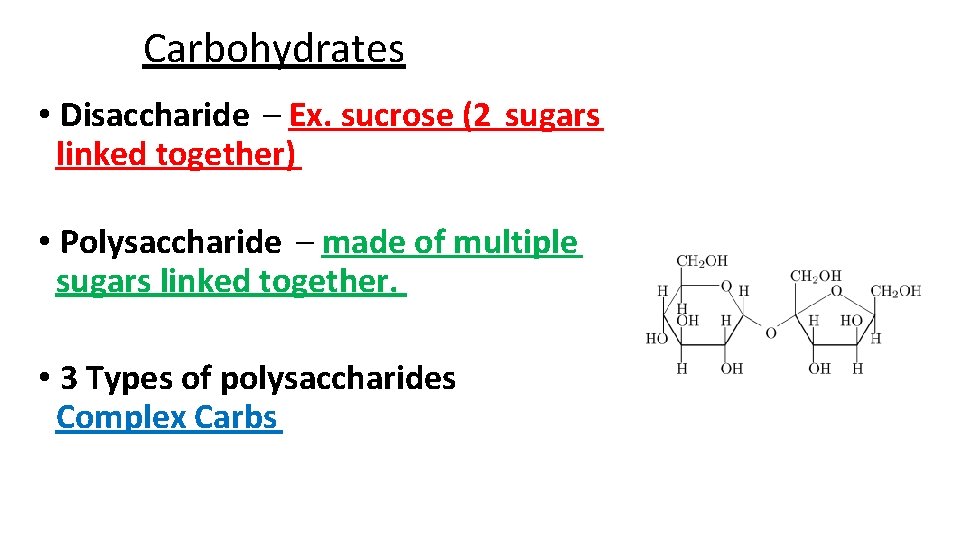
Carbohydrates • Disaccharide – Ex. sucrose (2 sugars linked together) • Polysaccharide – made of multiple sugars linked together. • 3 Types of polysaccharides Complex Carbs
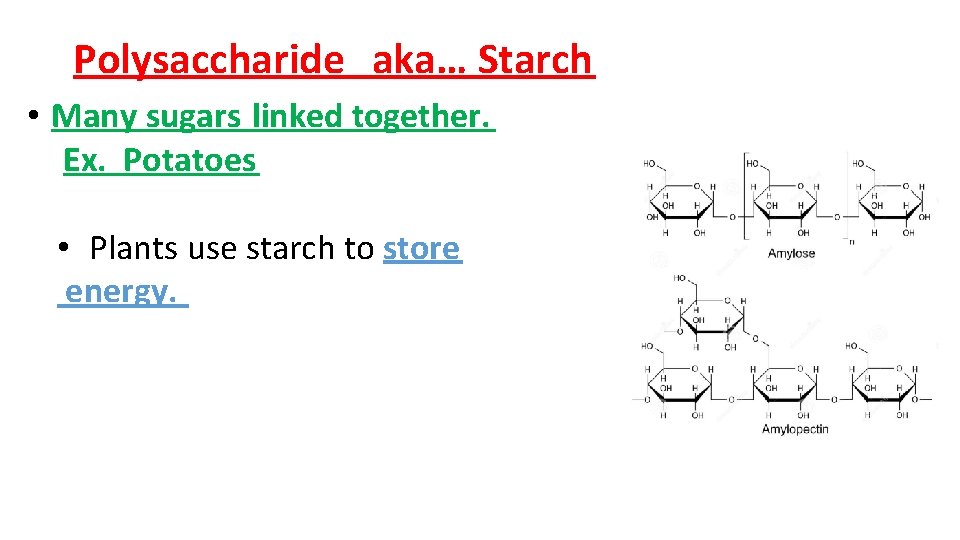
Polysaccharide aka… Starch • Many sugars linked together. Ex. Potatoes • Plants use starch to store energy.
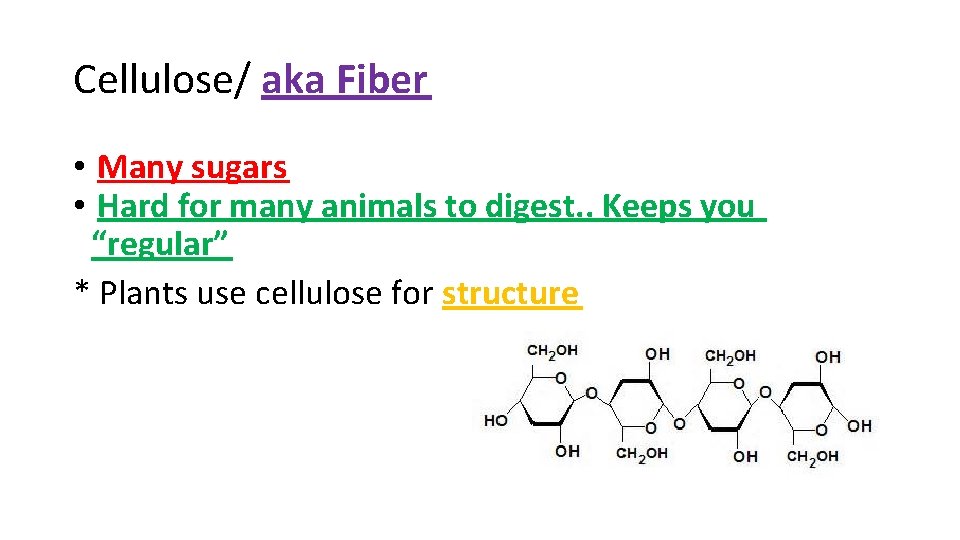
Cellulose/ aka Fiber • Many sugars • Hard for many animals to digest. . Keeps you “regular” * Plants use cellulose for structure

Glycogen • Stored in human liver until converted to fat. • Animals use glycogen for short term energy storage

Foods with Carbohydrates Simple Carbs = fruit , veggies, jelly beans (colorful) Complex Carbs = bread, pasta, rice (brown or whitish)

Carbs! https: //www. youtube. com/watch? v=wxzc_2 c 6 GMg

Carbohydrate Article (2. 03) • Read the article. It should be kept in your notebook. • Write the following questions in your notebook and then answer. 1. Where were the findings to this study published? 2. How does the article describe cognitive impairment? 3. What does this study suggest happened to the participants with a higher intake of carbohydrates at the beginning of the study? 4. What about those with a high fat diet? 5. How would you describe your diet? 6. Who funded the study?

Lipid Articles – As a group, answer the following questions • 1. What is HDL? • 2. What is LDL? • 3. Which one is better for you? HDL or LDL? Why ? • 4. What is one negative health effect of having a diet high in cholesterol? • 5. What are transfats? • 6. Why were transfats created? • 7. Name one “Do” and one “Don’t” concerning fats. Do= Don’t= Names ___________________________

Lipids… aka FATS • Lipids are made of Carbon and Hydrogen and small amounts of Oxygen. • Their main function is to store energy. • Other functions… Water proofing, in cell membranes, insulation • Types of lipids include: fats, waxes, and oils. • Energy is stored in the C-H bond and is released when the C-H-bond is broken.
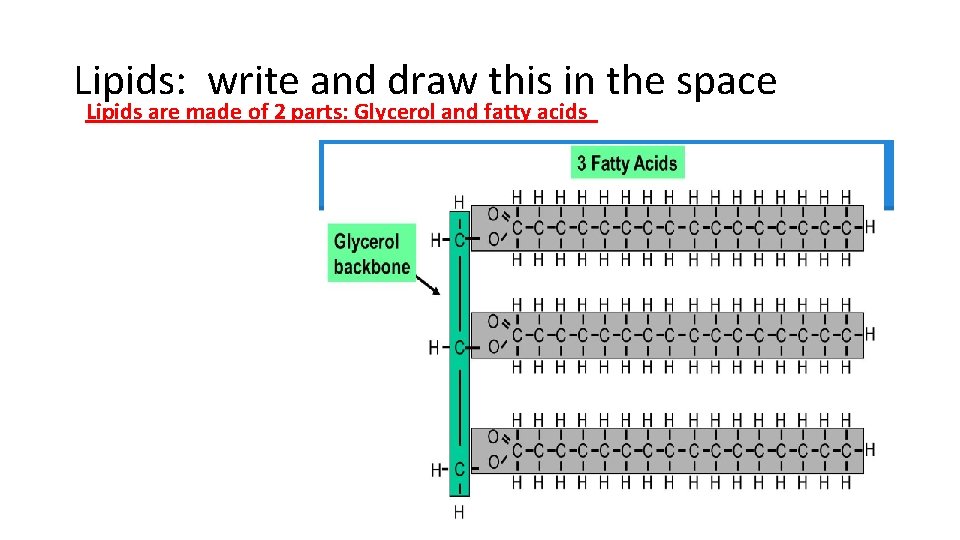
Lipids: write and draw this in the space Lipids are made of 2 parts: Glycerol and fatty acids

Lipids – Types of Fats • Saturated Fats • Long chains with no double bonds • Solid at room temp. • Ex. Cheese, butter, whole milk, high fat cuts of meat • Unsaturated Fats • Monounsaturated fats – only one double bond • Liquid at room temp • Ex. Olive oil, vegetable oil, avocado • Polyunsaturated fats – many double bonds • Ex. Salmon, sesame seeds, plant based oils

Lipids – Types of Fats continued! • Phospholipids – important for cell membrane structure • Steroids – serve as hormones… chemical messengers • Cholesterol – Prevents cell membrane from becoming too stiff. Foods containing Fats: Butter, oil, cream • Lipid Video

Lipid Articles – As a group, answer the following questions

Proteins • Proteins form a variety of functions in the body…. Speed up reactions(enzymes) • carry things, move things • they are the “workers” • Proteins are made up of amino acids. • Each amino acid contains: • 1. Amino group (-NH 2) • 2. Carboxyl group (-COOH) • 3. A different “R” group.

Protein Video
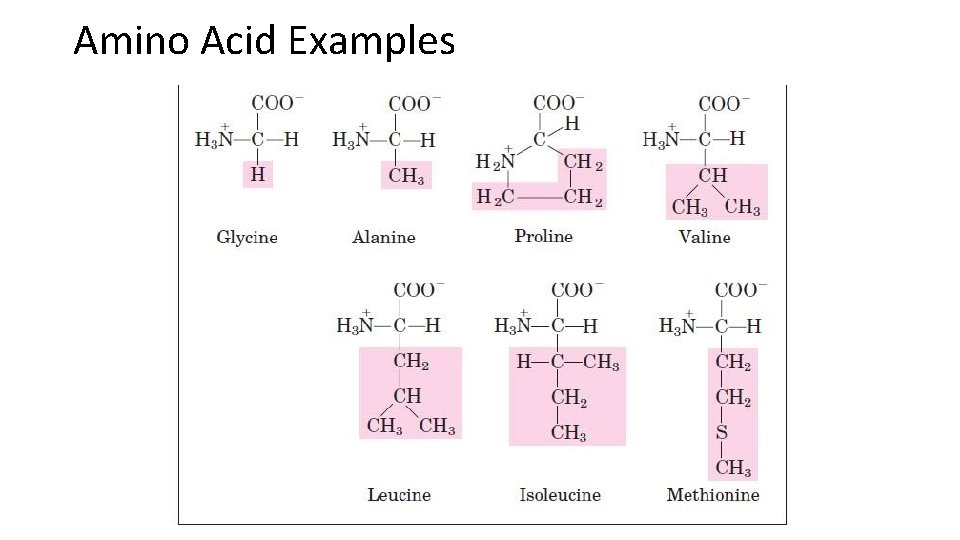
Amino Acid Examples
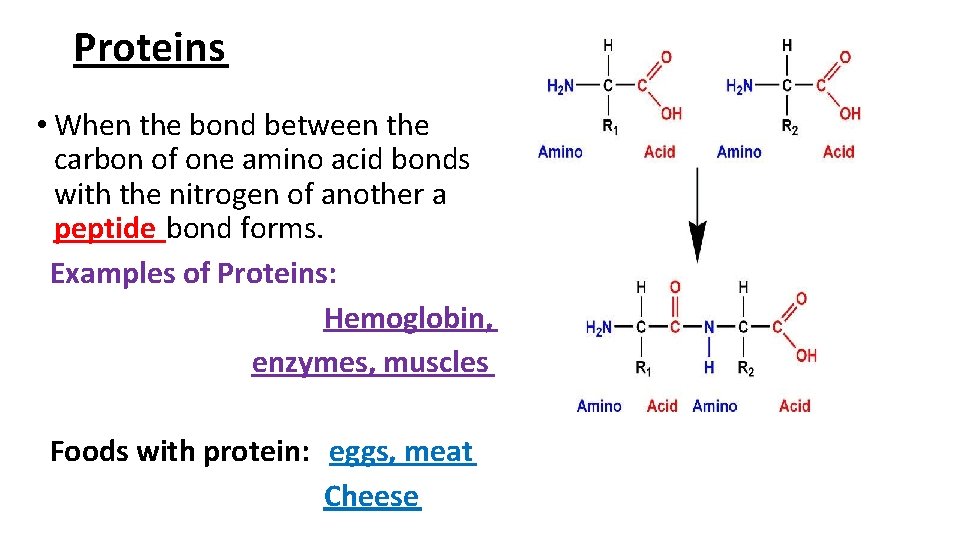
Proteins • When the bond between the carbon of one amino acid bonds with the nitrogen of another a peptide bond forms. Examples of Proteins: Hemoglobin, enzymes, muscles Foods with protein: eggs, meat Cheese

What are enzymes? • Proteins made up of amino acids • They change the rate of a chemical reaction • Name ends in –ase (Ex. Amylase – breaks down starch into sugar) • Can be reused • Acts as a catalyst (meaning they speed up a reaction) • Dependent on p. H and temperature • If you change the p. H or temperature, you can denature the enzyme, meaning the shape is changed and it will no longer function as well.
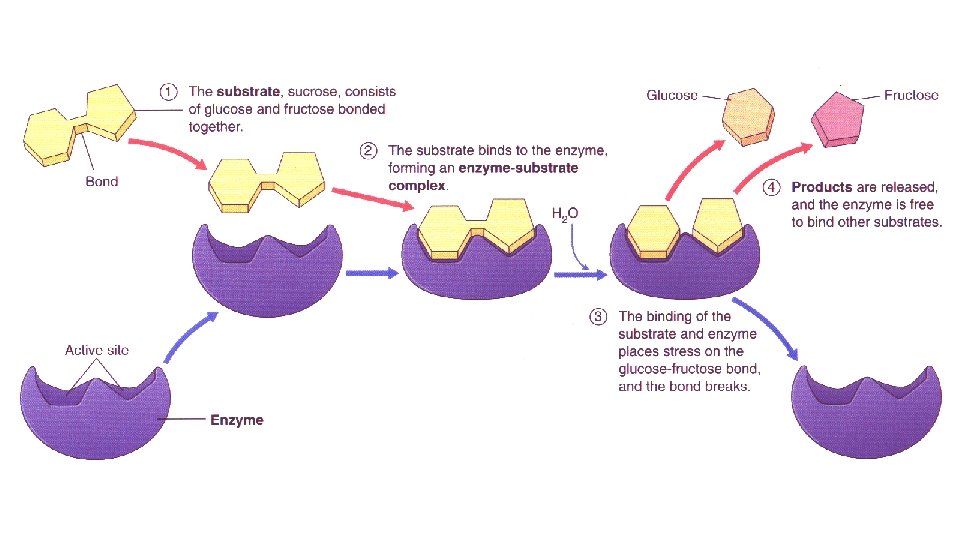
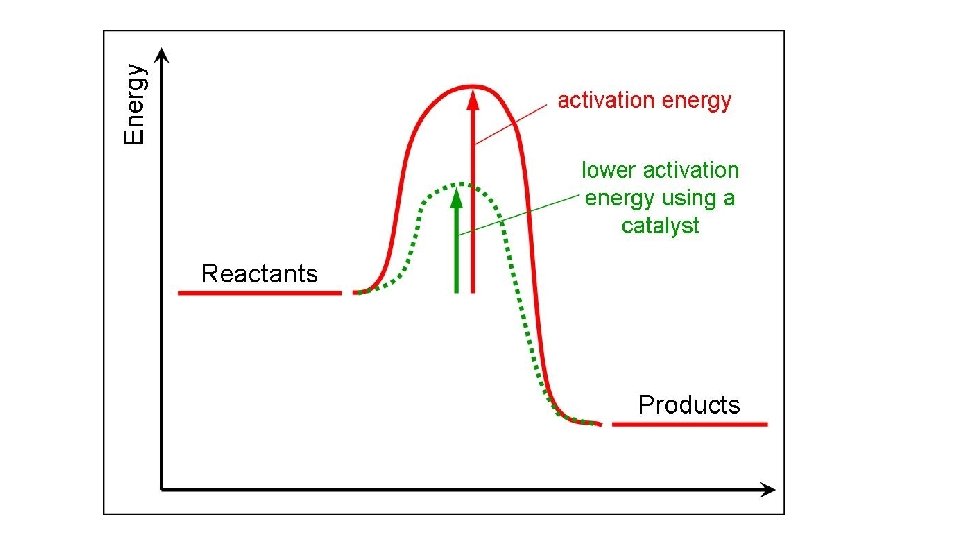

Enzymes • Substrate – The molecule that undergoes a change • Active site – where the enzyme and substrate come together like a “lock and key” • Product – the substrate is changed and products are released. • The enzyme does NOT change. It goes on to do the same reaction over and over. • Activation energy – The amount of energy required for the reaction to take place. Enzymes decrease the activation energy.

Nucleic Acids • Nucleic Acids are formed by nucleotides (the monomer). • Each nucleotide has: 1. a 5 – Carbon sugar 2. Nitrogenous base 3. phosphate group
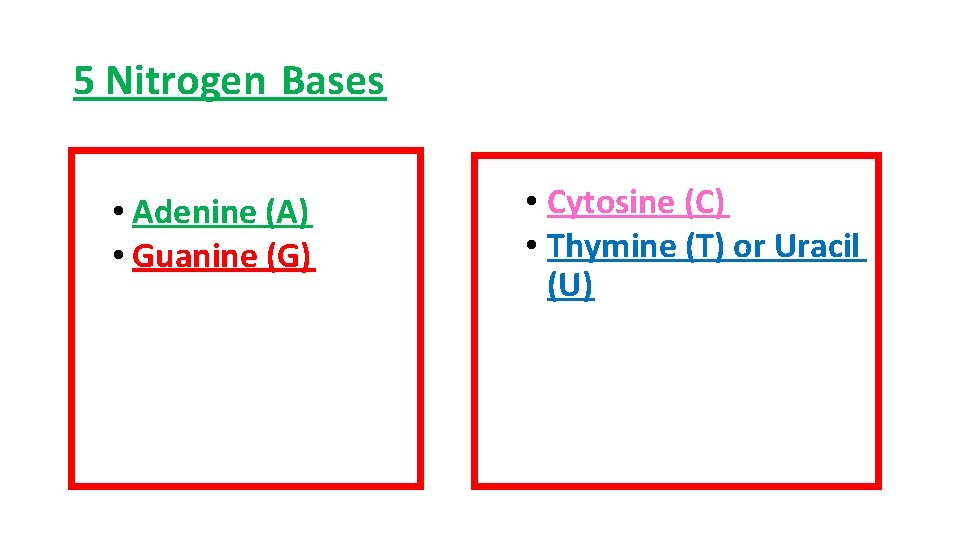
5 Nitrogen Bases • Adenine (A) • Guanine (G) • Cytosine (C) • Thymine (T) or Uracil (U)
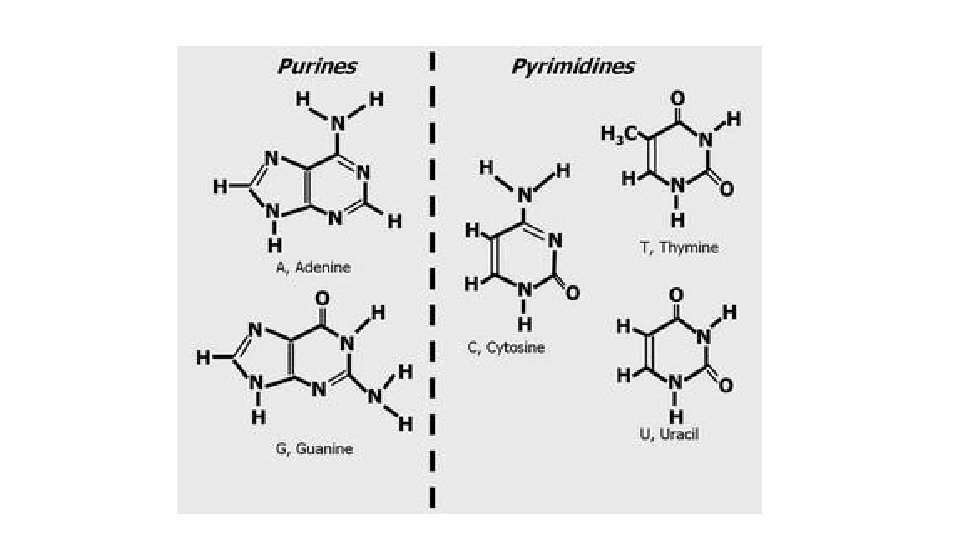
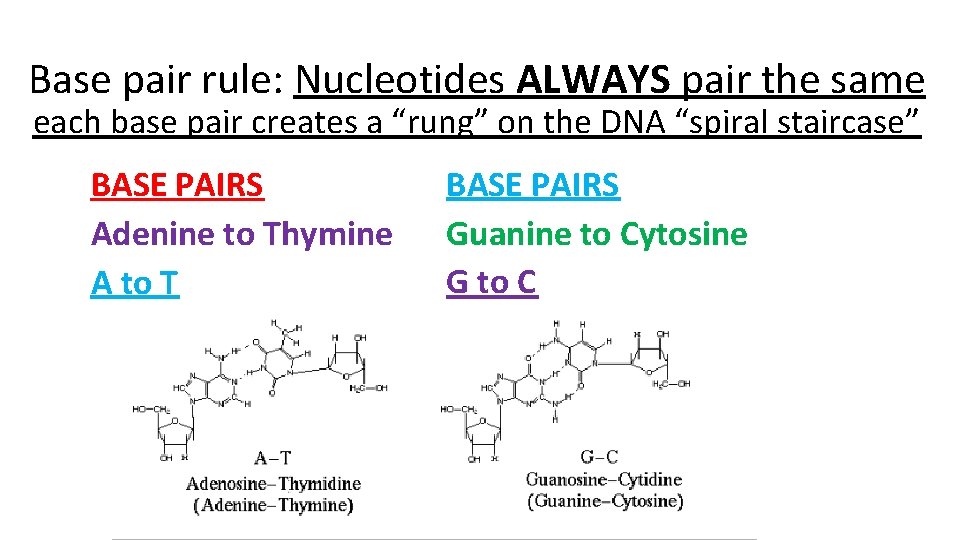
Base pair rule: Nucleotides ALWAYS pair the same each base pair creates a “rung” on the DNA “spiral staircase” BASE PAIRS Adenine to Thymine A to T BASE PAIRS Guanine to Cytosine G to C
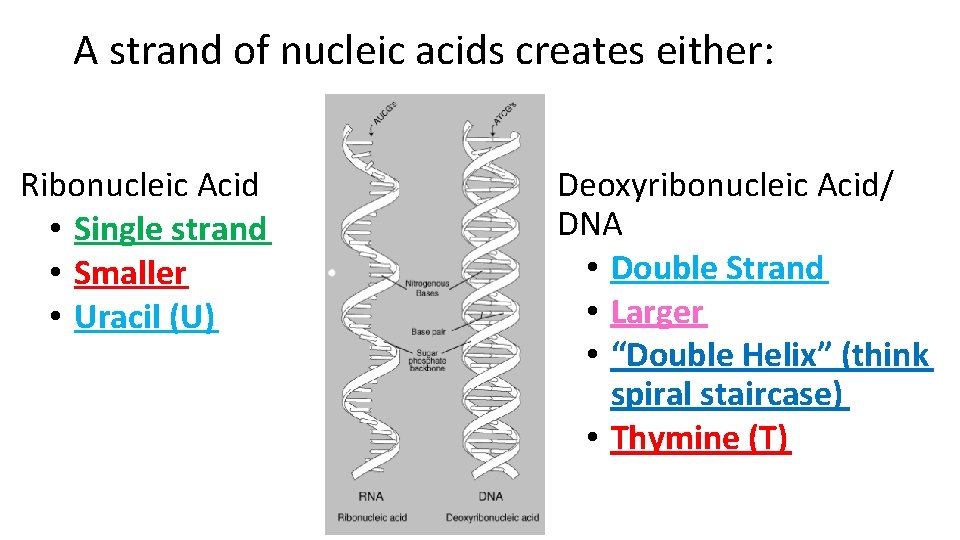
A strand of nucleic acids creates either: Ribonucleic Acid • Single strand • Smaller • Uracil (U) Deoxyribonucleic Acid/ DNA • Double Strand • Larger • “Double Helix” (think spiral staircase) • Thymine (T)
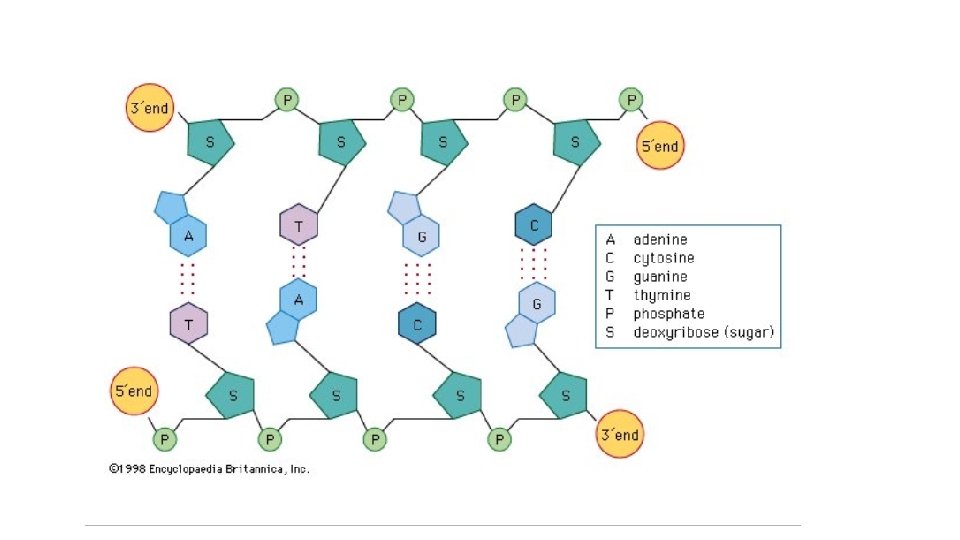

RNA = creates proteins from the DNA = is the Genetic Code… for your proteins Foods with Nucleic Acids: All living things contain DNA so all foods contain DNA. But Bananas and Strawberries • Nucleic Acid are easiest to see it in Video

Biomolecules Review!

Constructing a DNA Ladder - Instructions Questions: (for your notebook) 1. Describe the base-pair rule. 2. What three things make up a nucleotide? 3. What does anti-parallel mean? 4. Write YOUR sequence of base pairs.
- Slides: 40