Unit 2 Biochemistry 1 Life depends on chemistry

Unit 2 Biochemistry 1. Life depends on chemistry 2. There are 4 elements that make up 96% of the human body. 3. Elements are pure substances that can’t be broken down into other substances. They can be found listed on the Periodic table of elements.
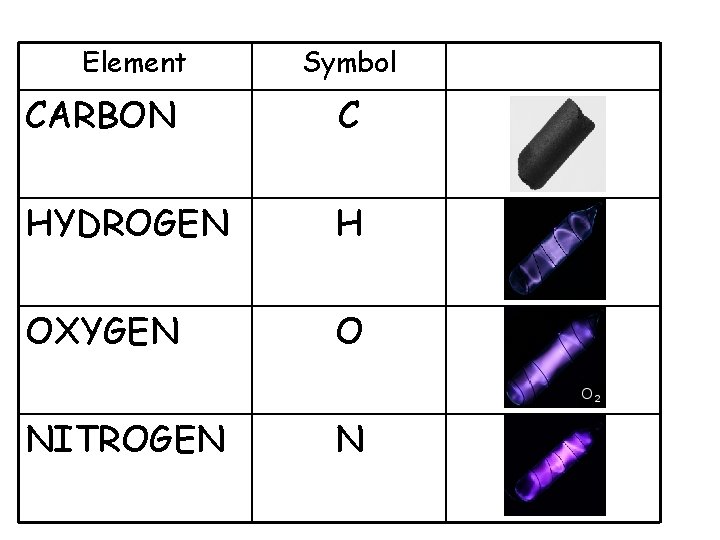
Element Symbol CARBON C HYDROGEN H OXYGEN O NITROGEN N

There are many important Compounds in Living Organisms 1. A compound is a substance that is made up of 2 or more elements that are chemically combined in a fixed ratio. 2. Every compound has a chemical formula even baking soda! Na. HCO 3 sodium bicarbonate

Most Common Compounds in Living Environment Formula Name H 20 Water C 6 H 1206 Glucose CO 2 Na. Cl Elements Present Hydrogen Oxygen Carbon Dioxide Carbon Oxygen Sodium Chloride Sodium Chlorine

Special Compounds Water 1. It is a neutral molecule which means it has no charge on it. 2. It has a p. H of 7. 3. It is known as the universal solvent because more things dissolve in water than any other compound. 4. In organisms, this allows for transport of substances throughout the cells.

Special Compounds Acids, Bases & p. H 1. The p. H scale is a way to measure how acidic or basic a substance is. 2. * The lower the p. H , the greater the acidity * The higher the p. H, the more basic the solution 3. Acids have a p. H less than 7 4. Bases have a p. H greater then 7 5. A p. H of 7 exactly is neutral.
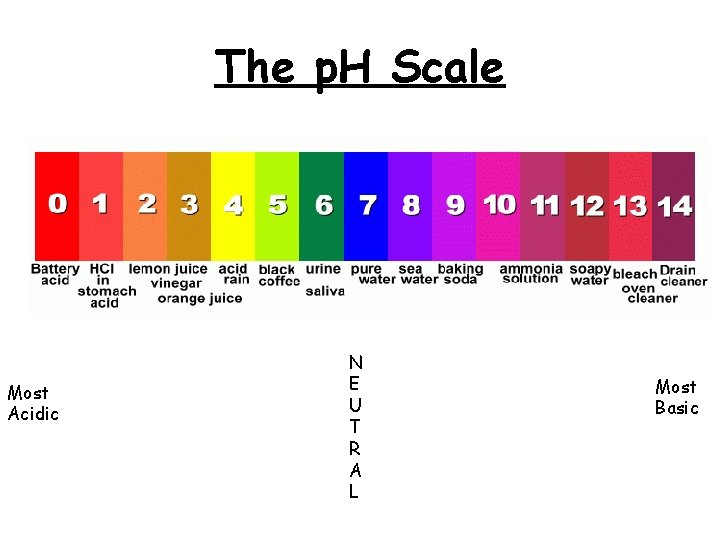
The p. H Scale Most Acidic N E U T R A L Most Basic
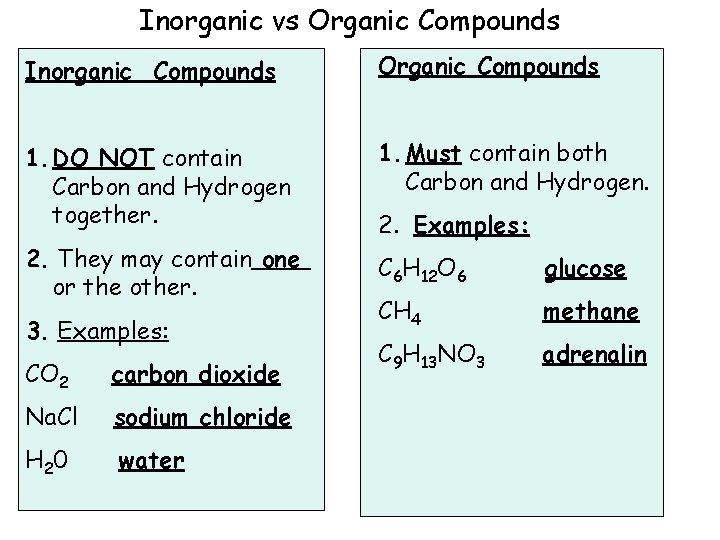
Inorganic vs Organic Compounds Inorganic Compounds Organic Compounds 1. DO NOT contain Carbon and Hydrogen together. 1. Must contain both Carbon and Hydrogen. 2. They may contain one or the other. C 6 H 12 O 6 glucose CH 4 methane C 9 H 13 NO 3 adrenalin 3. Examples: CO 2 carbon dioxide Na. Cl sodium chloride H 2 0 water 2. Examples:
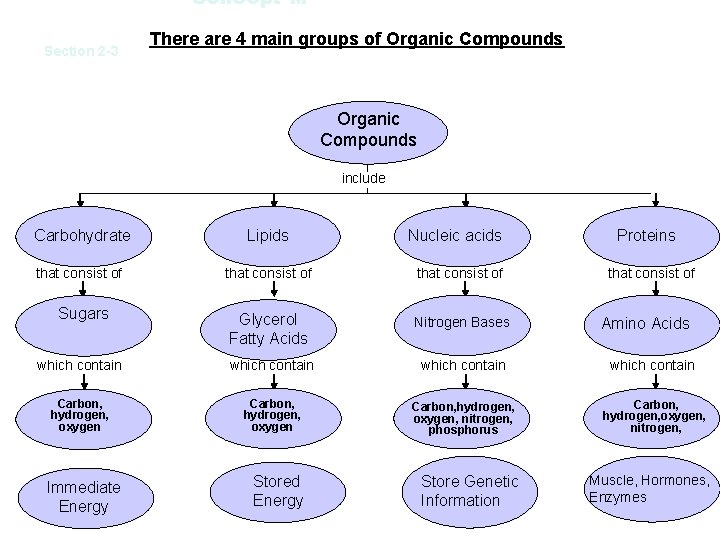
Concept M Section 2 -3 There are 4 main groups of Organic Compounds include Carbohydrate Lipids that consist of Glycerol Fatty Acids Nitrogen Bases which contain Carbon, hydrogen, oxygen, nitrogen, phosphorus Carbon, hydrogen, oxygen, nitrogen, Sugars Immediate Energy Stored Energy Nucleic acids Store Genetic Information Proteins that consist of Amino Acids Muscle, Hormones, Enzymes

Carbohydrates 1. Contains C, H, O 2. Elements are in a 1: 2: 1 ratio example: C 6 H 1206 glucose 3. Represented by a hexagon symbol 4. They are commonly simple sugars. 5. Main source of energy for living things. 6. Their name usually has an –ose ending.
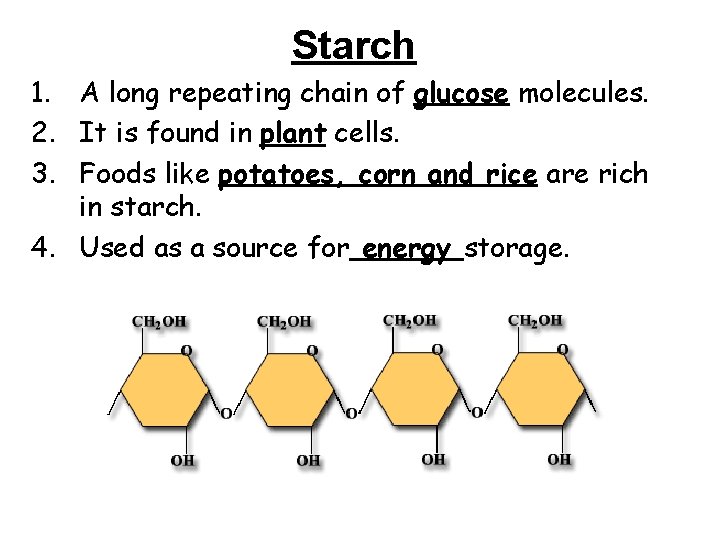
Starch 1. A long repeating chain of glucose molecules. 2. It is found in plant cells. 3. Foods like potatoes, corn and rice are rich in starch. 4. Used as a source for energy storage.
Lipids 1. Contains C, H, O 2. Commonly called fats 3. Important part of cell membranes. 4. Long term energy storage in organisms. 5. Insulates and cushions organs.
Nucleic Acids 1. Contain C, H, O, N, P (N=nitrogen and P=Phosphorous) 2. Examples: DNA and RNA DNA = deoxyribonucleic acid RNA = ribonucleic acid 3. Storage site for genetic information.
1. Contain C, H, O, N Proteins 2. Make-up hair, fur, hooves, nails, muscles. 3. Serve as a long –term (last resort) energy storage system. 4. They are represented by rectangles linked together. 5. Different amino acids link together to create what is known as a polypeptide chain. Then, these chains twist and fold together to make a larger protein molecule. AA AA

Chemical Reactions • Everything that happens in an organism – growth, development, response to environment – is a result of chemical reactions inside the organism. 2. A chemical reaction is a process that changes one set of chemicals into another set of chemicals 3. It always involves the breaking of bonds between atoms and the formation of new ones. 4. Chemists call the energy that is needed to get a reaction started the activation energy. 5. Some biochemical reactions have activation energies that are too high to make them practical for living tissue.

• Enzymes & Catalysts A catalyst is a substance that speeds up the rate of a chemical reaction. 2. Enzymes are the specialized proteins found in living things that act as biological catalysts. They speed up the chemical reactions that take place in cells. 3. They work because they lower the activation energy required to get the chemical reaction started.

Enzyme Names • Names of enzymes often end in “ ase”. • They also end in “in”. 3. Examples: lipase acts on lipids protease acts on proteins lactase acts on lactose pepsin acts on proteins

Enzyme Basics 1. The way that enzymes lower the “activation energy” that is required for a reaction is they physically provide a place for the reaction to occur. 2. Enzymes are very specific and catalyze only one specific reaction.
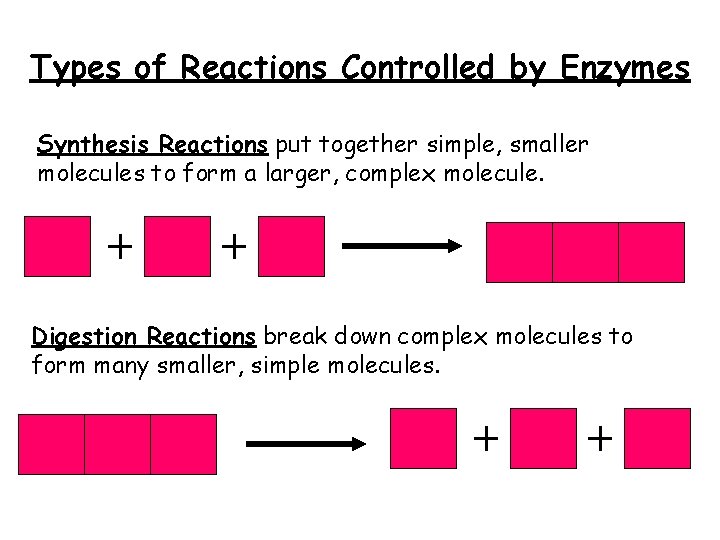
Types of Reactions Controlled by Enzymes Synthesis Reactions put together simple, smaller molecules to form a larger, complex molecule. Digestion Reactions break down complex molecules to form many smaller, simple molecules.
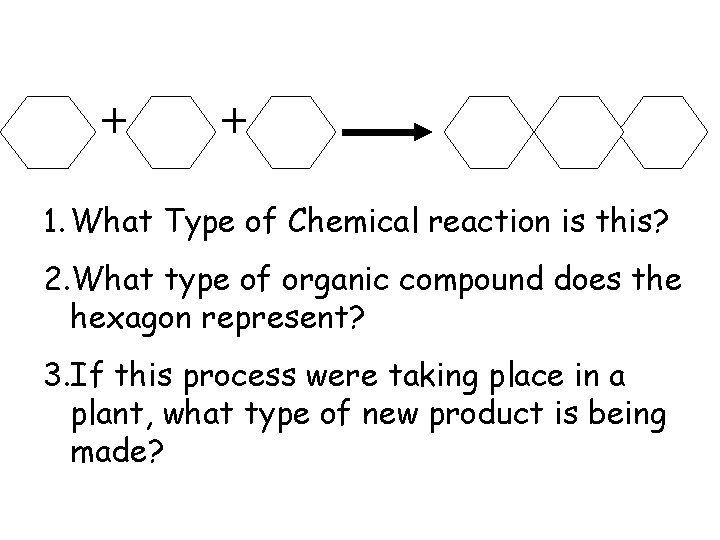
1. What Type of Chemical reaction is this? 2. What type of organic compound does the hexagon represent? 3. If this process were taking place in a plant, what type of new product is being made?

1. What type of reaction is this? 2. What type of organic compound is present at the start of the reaction 3. What do we call the products that are made by this reaction?
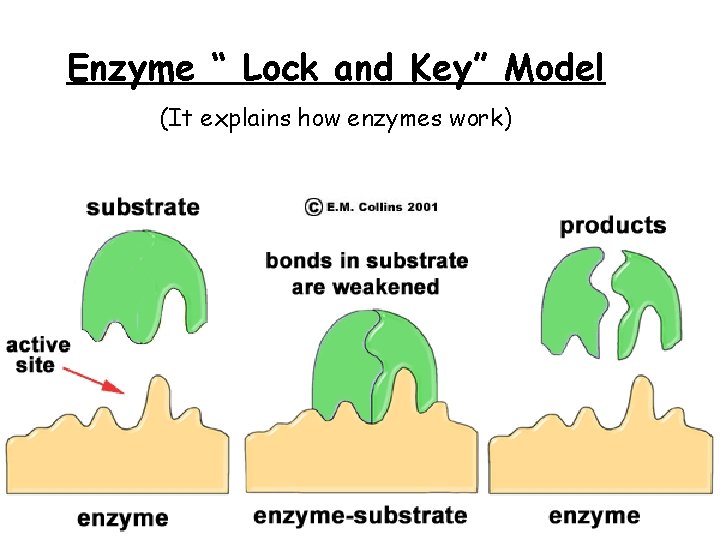
Enzyme “ Lock and Key” Model (It explains how enzymes work)

The active site on the enzyme attaches to a substrate molecule forming an enzyme-substrate complex. While attached to the substrate, the enzyme causes a weakening of certain chemical bonds in the substrate molecule, resulting in a breakdown of the substrate into two smaller product molecules. The enzyme is unaltered during the reaction and is free to catalyze the breakdown of another substrate molecule.

Definitions 1. Active site is the area on the enzyme where the substrate fits into. 2. Substrate is the reactant molecule(s) that attach to an enzyme. 3. Enzyme-Substrate Complex refers to what you have when the reactants are in place and attached to the enzyme at its active site.

Factors Affecting Enzyme Function 1. Temperature • When temperatures get too high the enzyme will become denatured. • Denatured means that it will lose its shape and its active site will no longer fit the substrate. • Most human body enzymes work best at 37°C (98. 6° F)- normal core body temperature.

2. p. H Each enzyme works at a specific p. H. Most inside cells work at a neutral p. H of 7. • Stomach enzymes such as pepsin work at a p. H of 2 • Pancreatic lipase work best at an optimal p. H of 12 • The human body has about 3000 different known types of enzymes.
- Slides: 26