Unit 2 Behavior of Gases Notes Quick Demo

Unit 2 - Behavior of Gases Notes
Quick Demo: Pumpkin Spice

Quick Demo: Pumpkin Spice • Think about what you already know: How do they move? What effects their movement? What is a gas?

Quick Demo: Pumpkin Spice Good models should be consistent with our Model So Far from Unit 1 and should… • Key Concept 1: The Pumpkin Spice demo illustrated that particles move. • We also can not forget about________ particles. • Diffusion of a gas 3 D. • • Random Fact for Today: Guess how fast a typical air particle moves at room temperature.

Quick Demo: Pumpkin Spice • Key Concept 2: Diffusion is the process of a substance spreading out to evenly fill its container or environment. • In a solution (liquid), a concentrated solute diffuses to spread evenly in its solvent. • In air, gas molecules diffuse to mix thoroughly. • Substances diffuse from areas of high concentration to low concentration.
Quick Demo: Green vs Blue Key Concept 3: Key Water Dye

Quick Demo: Blue vs. Green • Key Concept 4: A particles rate of diffusion is linked to the temperature of the system; • HOT- Fast COLD- Slow • Diffusion through a liquid.

Do solids diffuse? “Diffusion is not confined to gases; it can take place with matter in any state. For example, salt diffuses (dissolves) into water; water diffuses (evaporates) into the air. It is even possible for a solid to diffuse into another solid; e. g. , pure gold will diffuse into pure lead, two pieces of chalk tightly wrapped together are found to become homogeneous after some years; although at room temperature this diffusion is very slow. Generally, gases diffuse much faster than liquids, and liquids much faster than solids. ”

Before You Go… Are all particles always moving? How can you draw particles that are in motion?

End of Day

Unit 2 Purpose • We are going to investigate how gases behave and what factors affect their behavior. • By the end of this unit you will be able to explain the following scenario: The interior of the tank car washed out & cleaned with steam. Then all the outlet valves were shut and the tank car was sealed. All the workers went home for the evening and when they returned, this is what they found that the tank car crushed…

Before

After https: //www. youtube. com/watch? v=Zz 95_Vv. Tx. ZM

What is happening? ? Before • During After Brainstorming Session: with your partner • HOW/WHY did the Tanker Crush? Represent this at the Particle Level

Initial Group Models ? Before • • During After With your lab table share your ideas about how/why the tanker crushed and come up with a group model of what happened to the tanker. Make sure to include: • Diagram of water and air particles inside/outside the tanker with a key • Written explanation for each step • Model So Far and new key concepts

End of Day 2

Opener Gallery Walk • Take a walk (get up) and look at examples of your classmates initial models for why the tanker is crushing. • What are some new ideas (at least 2) you have about why/how the tanker crushes? • What ideas (at least 2) do you want to have explained more?

Big Idea: How gases behave and what factors affect their behavior. Question: WHow. Y did the tanker crush? Today: What is pressure?

Discussion Demos • “Sucking” Straw • Blowing A Student Up

Partner Talk If pressure is a force, what type of force might it be? a) Force is a push or pull b) Force cause physical change c) Net force = Mass times acceleration (F = ma) d) A force is that which changes or tends to change the state of rest or motion of a body.
Today: What is pressure? Question: WHow. Y did the tanker crush? Big Idea: How gases behave and what factors affect their behavior.

End of Day

Big Idea: How gases behave and what factors affect their behavior. Question: WHow. Y did the tanker crush? Today: What is pressure?

Last Class…. If pressure is a force, what type of force might it be? a) Force is a push or pull b) Force cause physical change c) Net force = Mass times acceleration (F = ma) d) A force is that which changes or tends to change the state of rest or motion of a body.

Discussion Demos • Under Pressure (what type of force) • Under Pressure Original

Discussion Demos Key Concept 5: Through exploring pressure demos, we determined that a particle can only push. At the particle level there is no way a particle can pull. •

Shoulder Partner Talk If pressure is a force, what type of force might it be? a) Force is a push or pull b) Force is the capacity to do work or cause physical change c) Force= Mass times acceleration (F = ma) d) A force is that which changes or tends to change the state of rest or motion of a body.

Drawing WHow. Y… How can we represent pressure in our models?

Pressure Key Concept 6: Pressure is caused by the pushing of particles on an object. Key Concept 7: Pressure is a measurement of the force (number of pushes) per unit of area. Pressure is a numerical value that represents the number of pushes per unit area. 1 atm = 760 mm. Hg = 14. 7 psi = 101. 3 k. Pa

Vacuum---- Sucking Force

“Sucking” Force vs. Pressure Force 1. Are these the same thing? 2. Is there really a thing called “sucking” force? The “Sucking” straw demo

By changing the shape of your lungs, you're actually getting the air pressure to push the liquid to your month.

Key Concept 8: How does sucking a straw work? 1. You expand/ or increase the volume of your lungs which decrease the air pressure in your lungs. 2. Therefore, the pressure in the straw is now greater compared to the pressure of your lungs. 3. Air particles in the straw move from high pressure in the straw to the low pressure of your lungs. 4. This creates lower pressure in the straw allowing the air pressure to PUSH the liquid up the straw.

• There is NO such thing as a sucking force. • Particles can only push. • How does that help us explain the tanker?

Pressure REVIEW: Key Concept 6: Pressure is caused by the pushing of particles on an object. Key Concept 9: Units of pressure can be. measured in atmospheres (atm) millimeters of mercury (mm. Hg), pounds per square inch (psi), or kilopascals (kpal).

Atmospheric Pressure • Key Concept 10: Atmospheric (Air) pressure is the pressure (PUSHES) exerted by the Earth’s atmosphere (AIR PARTICLES). Air pressure is measured by a barometer. • How does a barometer work?

Temperature • Key Concept 11: Temperature is a measure of the motion of the particles in an object. • Temperature is a measure of the average kinetic energy of particles in matter. • Kinetic energy is the energy an object has because of its motion.

Vacuum and Gravity • Key Concept 12: A Vacuum is a space where no particles or matter exist. • Key Concept 13: Gravity is the force of attraction by which terrestrial bodies tend to fall toward the earth. • Pass Out Practice Problems 2 A and #16 Particle Diagrams.

Practice Problems • Planning Ahead- Prove It Quiz 1 over Unit 2 will be on Tuesday Oct 9 th, over JUST practice problems 2 A. Quiz 2 retakes will be done after Fall Break.

Soda Can Lab—Part 1 • Goal: To be dicovered! (You’ll write this in later. )

Soda Can Lab— Procedures 1. Fill the plastic container up with ice water so the soda can will be submerged at least half way up the can. 2. Fill the dry soda can up with 15 m. L of water. 3. Using the tongs, place the soda can on the hot plate then turn the hot plate on. 4. Wait until the water inside starts boiling (you will see steam, and hear bubbling). 5. Let the water boil for a minute. 6. Using the tongs, CAREFULLY pick up the pop can and place it in the ice water. Make sure the opening of the can has been placed in the ice water ‘head first’ 7. Turn off the hot plate. 8. Pull out the can and make qualitative observations.

Safety • Apron • Goggles

End of Day 5 Soda Day 1

Big Idea: How gases behave and what factors affect their behavior. Question: WHow. Y did the tanker crush? Today: Soda Can Lab Continued

Break into your groups and complete the Conclusion Questions. This includes writing in the Goal!

What variables can we investigate to see what affected the amount the can crushed?
Today: Soda Can Lab Question: WHow. Y did the tanker crush? Big Idea: How gases behave and what factors affect their behavior.

End of Day

Big Idea: How gases behave and what factors affect their behavior. Question: WHow. Y did the tanker crush? Today: Soda Can Lab Discussion Part 2 - Pop Can Challenge

What variables can we investigate to see what affected the amount the can crushed?
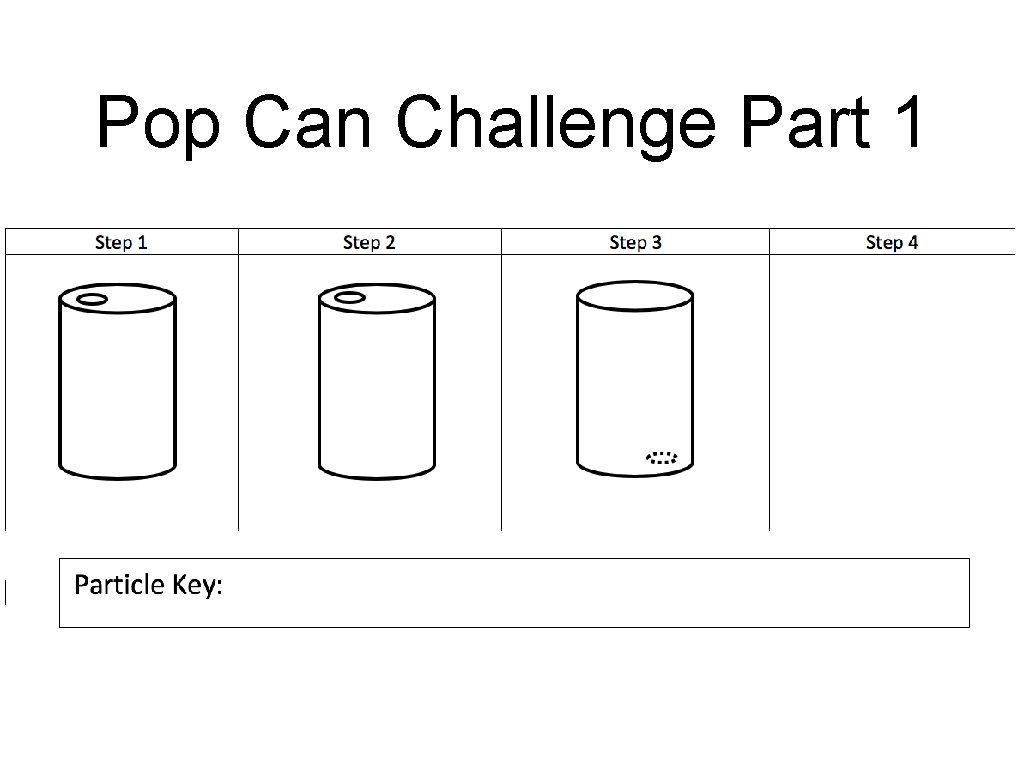
Pop Can Challenge Part 1

Pop Can Challenge: Part 2 Goal: Determine what factors affect the crushing of the can. • Size of can • Temp of “Ice Bath” • Soda can heating time (first determine the control heating time) • Amount of water in Soda Can • Material • Open vs. Closed System • **Opposite/ Cooling Time Te s Dif ting f Va eren ria t ble s

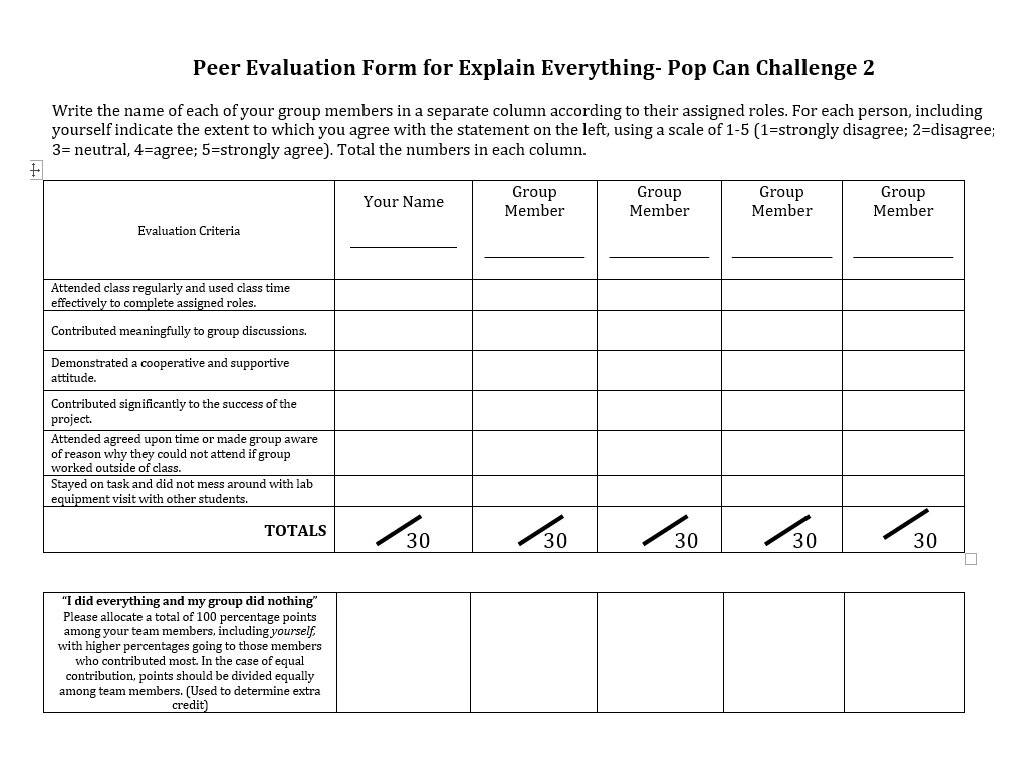
Explain Everything Project You will be working with your Lab Table to test your assigned variable and make a video to communicate your results. Video nd and handout must be done October 22 at 3: 00 pm. th 19 If your video is done by October your group will be rewarded- We need to watch two of the videos during class on the 19 th.

How can we communicate our results? Everyone will have a job! • Recorder/Time Keeper • Particle Artist • Tech Director • Experimenters (x 2)

Soda Can Lab—Part 2 Testing Different Variables Table ____Size of can Table ____Temp of “Ice Bath” Table _____Soda can heating time (first determine the control heating time) Table _____Amount of water in Soda Can Table _____ Material Table _____ Open vs. Close System Table _____ Opposite/ Cooling Time

Recorder/Time Keeper

Soda Can Lab—Part 2 On your board write your variable. Then copy the trials below. Trial 1 - Control Trial 2 Trial 3 Trail 4 -

Recorder/Time Keeper On ne the xt pa ge
Why/How Does the Can Crush?

Soda Can Lab—Part 2 Now, that your variables have been determined, as a group compose your hypothesis (if/then the can will crush…/because) on your whiteboards. Be ready to share with the class.

Recorder/Time Keeper

Procedure Everyone will do this step. 1) Trial 1 - We will put 15 m. L of water in a 12 oz can, we will boil the water for 1 minute, and completely invert the can into an ice bath. This will serve as our group’s ______

Class Discussion What is a control?

Recorder/Time Keeper 1. Trial 1 - We will put 15 m. L of water in a 12 oz can, we will boil the water for 1 minute, and completely invert the can into an ice bath. 2. Trial 23. Trial 34. Trial 4 -
Today: Part 2 - Soda Can Lab Question: WHow. Y did the tanker crush? Big Idea: How gases behave and what factors affect their behavior.

End of Day

Big Idea: How gases behave and what factors affect their behavior. Question: WHow. Y did the tanker crush? Today: Part 2 - Soda Can Lab How to use the app

Explain Everything Project You will be working with your Lab Table to test your assigned variable and make a video to communicate your results. Video and handout must be done October 9 th at 3: 00 pm. th 8 If your video is done by October your group will be rewarded- We need to watch two of the videos during class on the 9 th.

Explain Everything Project • Everyday your Recorder/Timekeeper will be given a list of tasks. • THEY ARE RESPONISIBLE FOR COMMINCATING WHAT NEEDS DONE. • IT IS YOUR RESPONSIBLEITY TO GET YOUR TASK DONE. • If it does not get done during class you must come in after school or before school to finish it.


Explain Everything Project Your lab group will be using the assigned I-pad number that correspond to your hour and lab table number.

Explain Everything Project • Take a picture of the periodic table and a very short (3 sec video). • Explain Everything Tutorial

Explain Everything Project Example video for Day 2 • How will we be graded? • 20 pts for complete handout • 36 pts for video which is grade on a checklist (Last page of the Pop Can Challenge: Part 2 handout) • 10 pts peer evaluations

Quick meeting with each job group 1. Recorder/Time Keeper 2. Experimenters 3. Tech Director 4. Particle Artist

Today: Part 2 - Soda Can Lab Question: WHow. Y did the tanker crush? Big Idea: How gases behave and what factors affect their behavior.

End of Day 8

Big Idea: How gases behave and what factors affect their behavior. Question: WHow. Y did the tanker crush? Today: Part 2 - Soda Can Lab

Today: Part 2 - Soda Can Lab Question: WHow. Y did the tanker crush? Big Idea: How gases behave and what factors affect their behavior.

End of Day

Big Idea: How gases behave and what factors affect their behavior. Question: WHow. Y did the tanker crush? Today: Part 2 - Soda Can Lab

Today: Part 2 - Soda Can Lab Question: WHow. Y did the tanker crush? Big Idea: How gases behave and what factors affect their behavior.

End of Day

Big Idea: How gases behave and what factors affect their behavior. Question: WHow. Y did the tanker crush? Today: Part 2 - Soda Can Lab

Today: Part 2 - Soda Can Lab Question: WHow. Y did the tanker crush? Big Idea: How gases behave and what factors affect their behavior.

End of Day

Big Idea: How gases behave and what factors affect their behavior. Question: WHow. Y did the tanker crush? Today: Experimental Design (ACT) & Model Revision with rubrics

Experimental Design What is a control? or a controlled experiment?

Procedures 1. Trial 1 - We will put 15 m. L of water in a 12 oz can, we will boil the water for 1 minute, and completely invert the can into an ice bath. The Control Sets the Standard By definition the control in a science experiment is a sample that remains the same throughout the experiment. The control must remain the same or equal at all times in order to receive accurate results. You can have as many controls as necessary to achieve results.

Experimental Design Key Concept 14: What is a control? In Pop Can Challenge 2 - Trial 1 was our control group. This can allowed us to be able to compare if our other trials crushed more or less. It was used as the standard of comparison.

Experimental Design How Does a Control Compare to Other Variables?

Experimental Design Key Concept 15: What is an independent variable? An independent variable is the variable that YOU the experimenter change or manipulate. In Pop Can Challenge 2 our independent variables was. . .

Soda Can Lab—Part 2 Testing Different Variables Table ____Size of can Table ____Temp of “Ice Bath” Table _____Soda can heating time (first determine the control heating time) Table _____Amount of water in Soda Can Table _____ Material Table _____ Open vs. Close System Table _____ Opposite/ Cooling Time

Experimental Design Key Concept 16: What is a dependent variable? A dependent variable is the variable that responds to independent variable (being measured) In Pop Can Challenge 2 our dependent variables was. . .

Soda Can Lab—Part 2 Testing Different Variables Table ____Size of can Table ____Temp of “Ice Bath” Table _____Soda can heating time (first determine the control heating time) Table _____Amount of water in Soda Can Table _____ Material Table _____ Open vs. Close System Table _____ Opposite/ Cooling Time

Experimental Design As the experimenter changes the independent variable, the change in the dependent variable is observed and recorded.

How/Why Poster Update

Model Update Mid-Unit Model Updates - turn over your initial models - work collaborative and quickly - follow class key on the board - follow the rubric & fill it out at the end - once completed, give it to Mrs. Young Finalize Project -Proof (play each slide) & Upload to Dropbox

Projects • Handout and Particle Diagram must be stapled and turned into the bin. • When project is done– it must be uploaded to Dropbox (this will take about 5 minutes)
Today: Opposite/ Cooling Time Demo Model Revision with rubrics Question: WHow. Y did the tanker crush? Big Idea: How gases behave and what factors affect their behavior.

End of Day

Big Idea: How gases behave and what factors affect their behavior. Question: WHow. Y did the tanker crush? Today: Presentations

Explain Everything Presentations. Pop Can Challenge 2 • • Complete peer evaluations. Turn in your evaluations into the turn in bin along the back wall. • 1 st hour bin----Table 1 • 2 nd hour bin---Table 2 • 3 rd hour bin—Table 3 ect.

Explain Everything Presentations. Pop Can Challenge 2 Key Concept 14: Class Claims and Reasoning Poster

Today: Presentations Question: WHow. Y did the tanker crush? Big Idea: How gases behave and what factors affect their behavior.

End of the Day

Big Idea: How gases behave and what factors affect their behavior. Question: WHow. Y did the tanker crush? Today: Define Mathematical Relationships

Science 101 Review • Key Concept 15: What is the independent variable verse the dependent variable in an experiment?

Science 101 Review • Key Concept 16: What is a positive verse negative mathematical relationship?

Rotation Station Lab Temperature vs Volume • Station A: Ballon Flask • Station B: Balloon in Dry Ice Pressure vs Volume • Station C: Marshmallow & A Bell Jar • Station D: Reversible Balloon Pressure vs Temperature • Class Demo: Egg in a Flask
Today: Practice Mathematical Relationships Question: WHow. Y did the tanker crush? Big Idea: How gases behave and what factors affect their behavior.

End of the Day

Big Idea: How gases behave and what factors affect their behavior. Question: WHow. Y did the tanker crush? Today: Define Mathematical Relationships

Rotation Station Lab From Yesterday… Temperature vs Volume • Station A: Balloon Flask • Station B: Balloon in Dry Ice Pressure vs Volume • Station C: Marshmallow in a Bell Jar • Station D: Reversible Balloon Pressure vs Temperature • Class Demo: Egg in a Flask Post-it Note Survey Stick your Post-it on what your think is “bigger” in science a theory or a scientific law.

Pressure vs. Temperature Demo

Pressure vs. Temperature Demo How can we get the egg out using gas law relationships? Complete the sentence by circling your choices and filling in the blank. • To remove the egg I believe that I have to (increase)/ (decrease) the variable (temperature)/ (pressure) by _______.

Pressure vs. Temperature Demo

Where are we Going? Defining Mathematical Relationships Pressure Temperature Volume # of Particles

Temperature vs Volume Key Concept 19: • 1 Volume 2 Temperature What has to be held constant? 3
Pressure vs Volume Key Concept 20 : • 1 Volume 2 Pressure What has to be held constant? 3

Pressure vs Number of Particles Key Concept 21: • 1 Pressure 2 # of Particles (n) What has to be held constant? 3


Pressure vs Temperature Key Concept 22: 1 2 Pressure • Temperature o. C What has to be held constant? 3
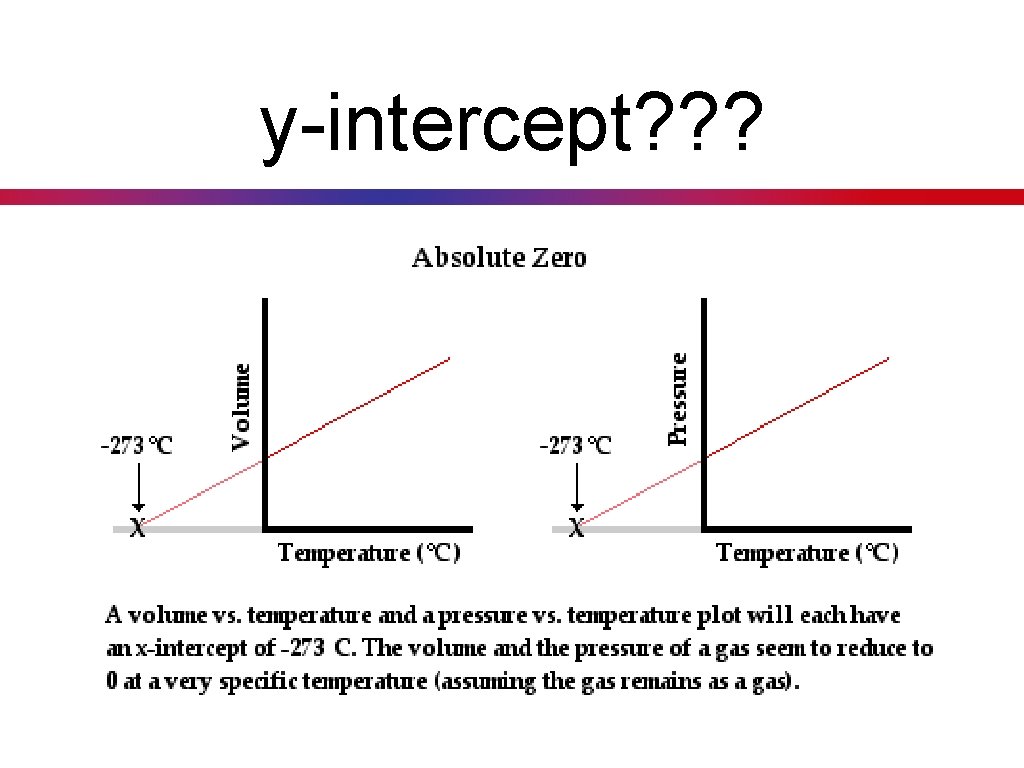
y-intercept? ? ?
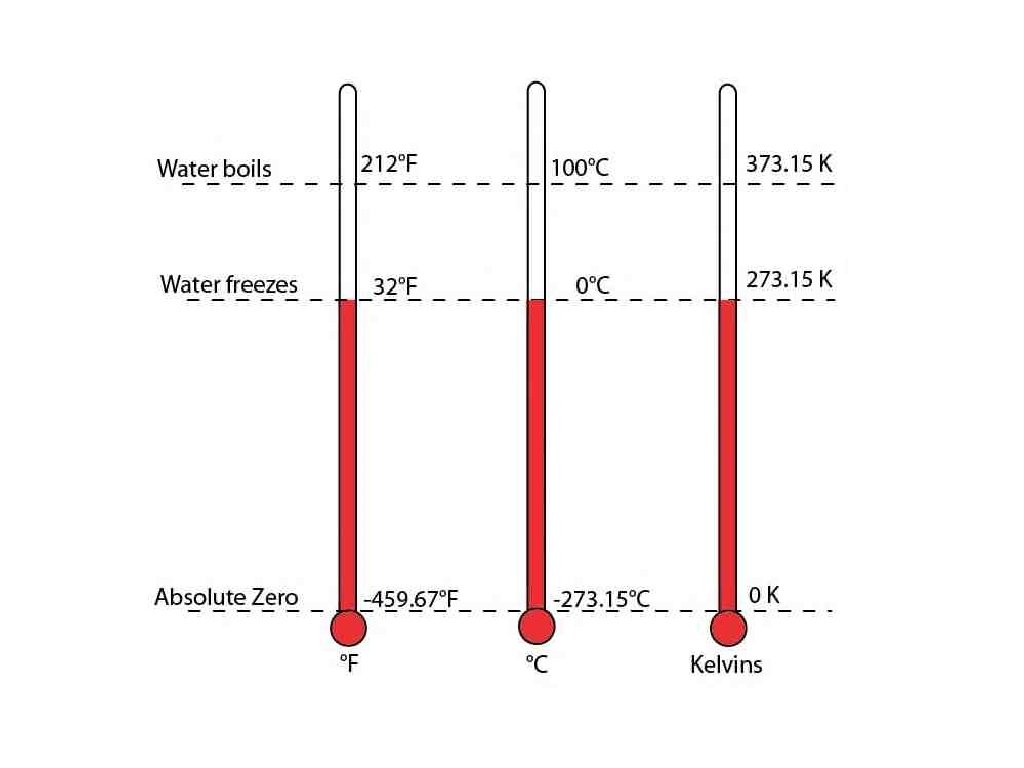
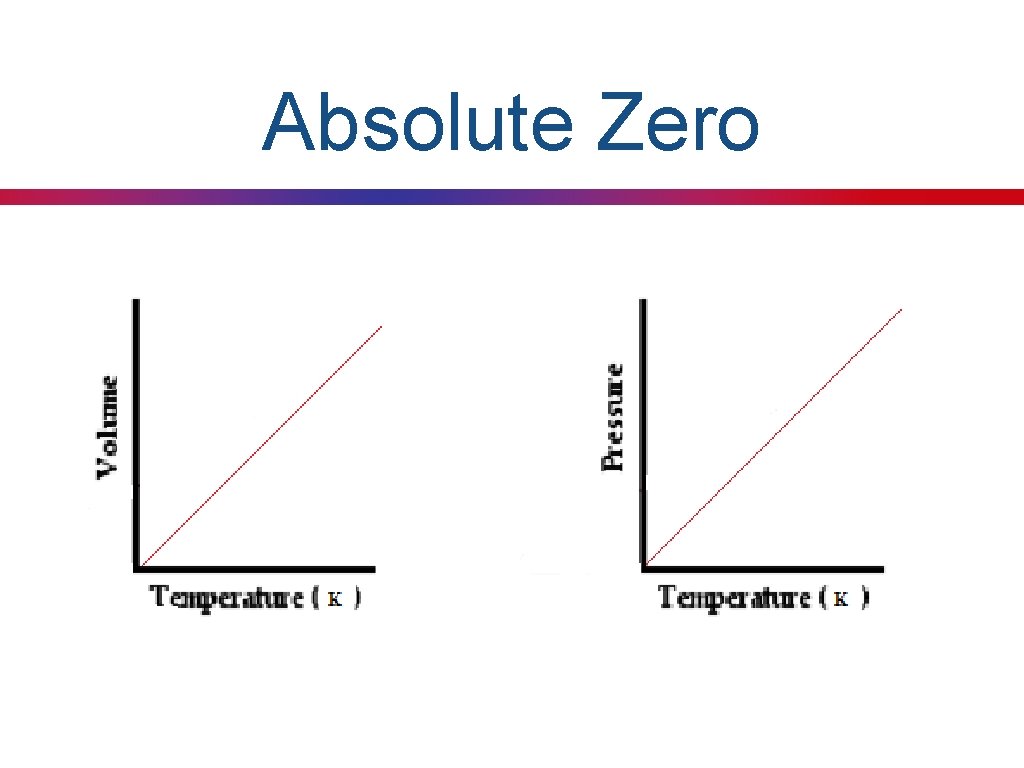
Absolute Zero

Absolute Temperature Key Concept 23: When solving gas law problems the Kelvin scale must be used for all temperatures. I need to convert all units of temperature to Kelvin. Practice: Convert 43 o. C to degrees Kelvin.

Practice Thumbs Up= Increase Thumbs Down= Decrease A. If I have a gas and I increase its temperature, what will happen to its pressure? B. If I have a gas at a certain volume and I decrease the pressure what happens to the volume? C. If I take a gas and decrease the temperature, what will happen to its volume?
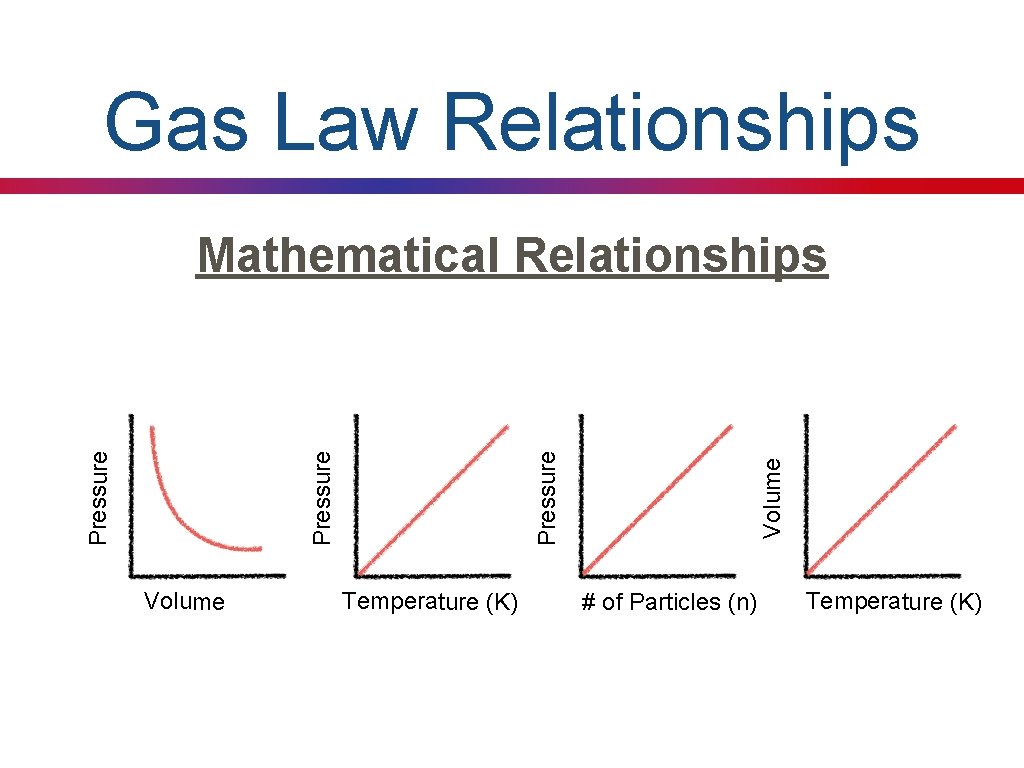
Gas Law Relationships Volume Temperature (K) Volume Pressure Mathematical Relationships # of Particles (n) Temperature (K)

Proportional Relationships A. If I have a gas and double its temperature, what will happen to its pressure? B. If I have a gas at a certain volume and I cut the pressure by a third what happens to the volume? C. If I take a gas and decrease the temperature by 10%, what will happen to its volume?

Practice B. of 25 m. L B. If II have aa gas at at aa volume certain volume and Ithe decrease thewhat pressure and 1 I atm, decrease pressure to 0. 72 toatm happens to the happens thewhat volume? P Initial Final Effect PP 2 B T V n

Practice A gas has a temperature of 14 0 C, and a volume of 4. 5 liters. If the temperature is raised to 29 0 C and the pressure is not changed, what is the new volume of the gas? P Initial Final Effect PP 2 C T V n

Speed Dating • Practice Problems 2 B • #8 • #9

Practice A balloon filled with CO 2 travels from Flag to Phoenix. The balloon has an initial pressure of 1. 08 atm, a temperature at 25 o. C, and volume equal to 2 L. What is the temperature in Phoenix if the balloon volume decreases to 1. 9 L and the pressure increases to 1. 2 atm? P Initial Final Effect T V n

Speed Dating • Practice Problems 2 B • # 11
Today: Practice Mathematical Relationships Question: WHow. Y did the tanker crush? Big Idea: How gases behave and what factors affect their behavior.

End of Day 16
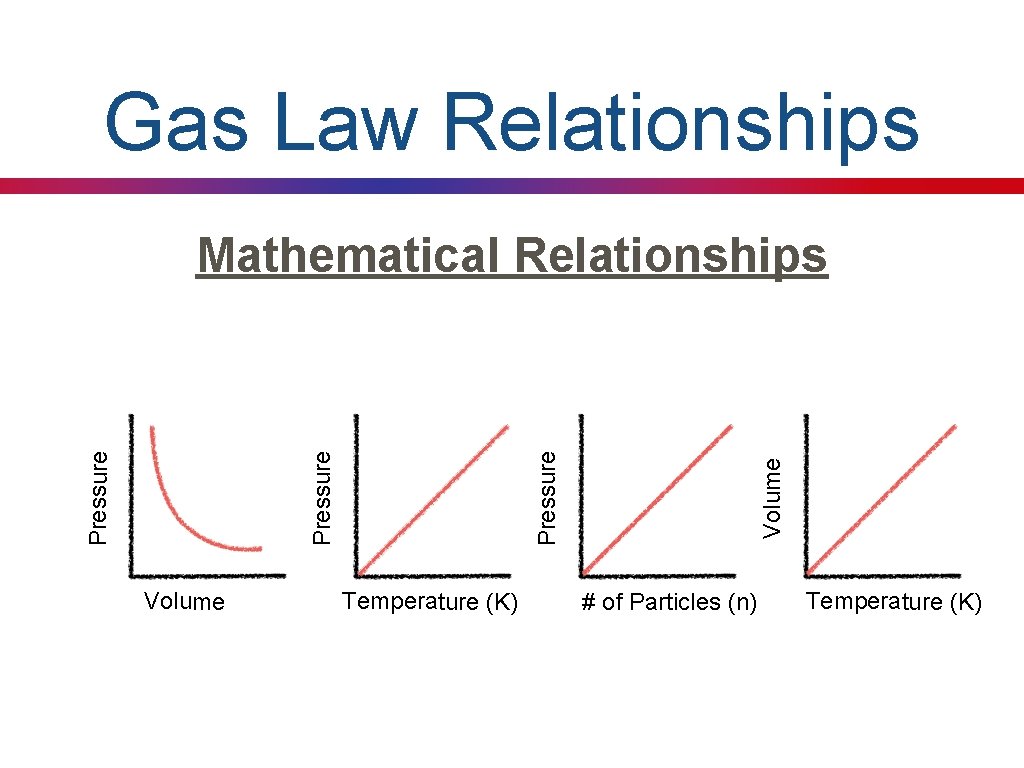
Gas Law Relationships Volume Temperature (K) Volume Pressure Mathematical Relationships # of Particles (n) Temperature (K)

Page 333

Boyle’s Law 1627 -1691
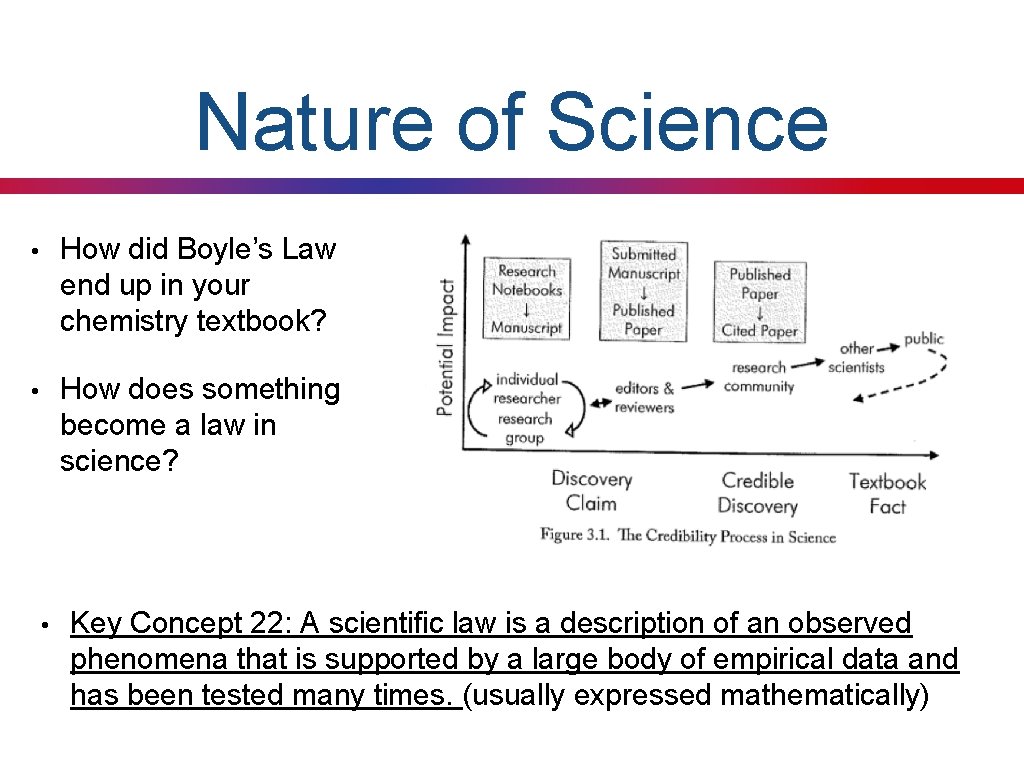
Nature of Science • How did Boyle’s Law end up in your chemistry textbook? • How does something become a law in science? • Key Concept 22: A scientific law is a description of an observed phenomena that is supported by a large body of empirical data and has been tested many times. (usually expressed mathematically)

Boyle’s Law • Key Concept 23: Boyle’s law states that, at a constant temperature, the volume of a gas is inversely proportional (negative relationship) to the pressure. • As pressure decreases, the volume will increase. P 1627 -1691 V P V
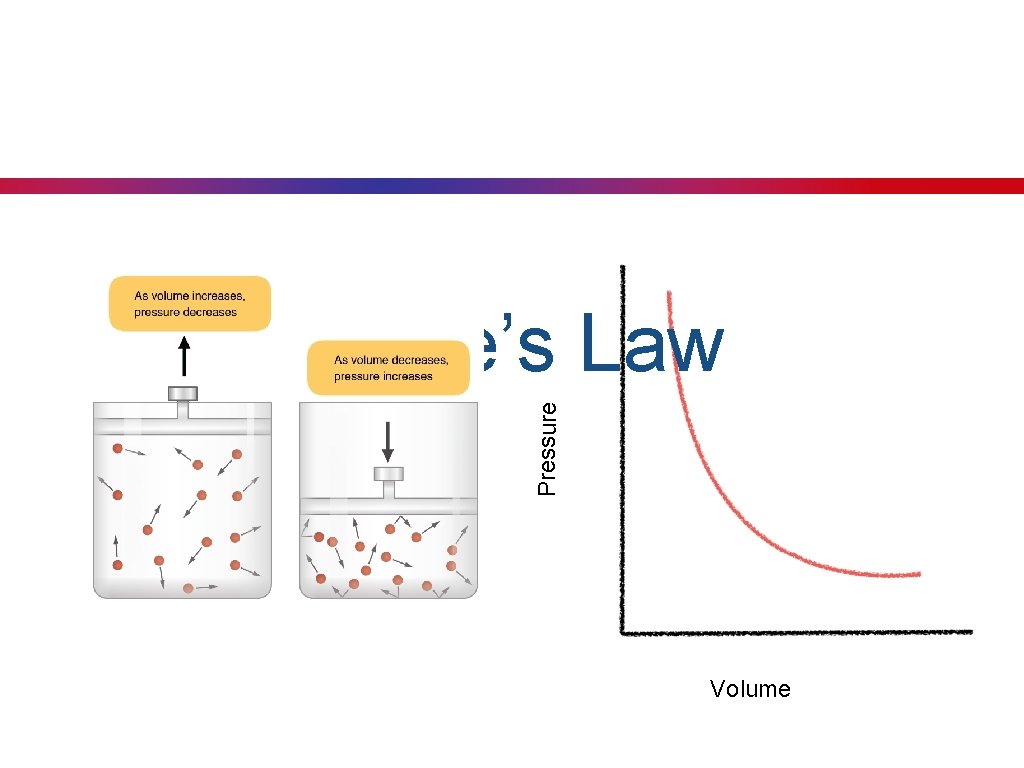
Pressure Boyle’s Law Volume


Charles’s Law • Key Concept 24: Charles’ law states that at a constant pressure, the volume of a given quantity of a gas varies directly (positive relationship) with the temperature. • temperature in Kelvin • As the temperature increases, the volume will also increase. V T 1747 - 1823
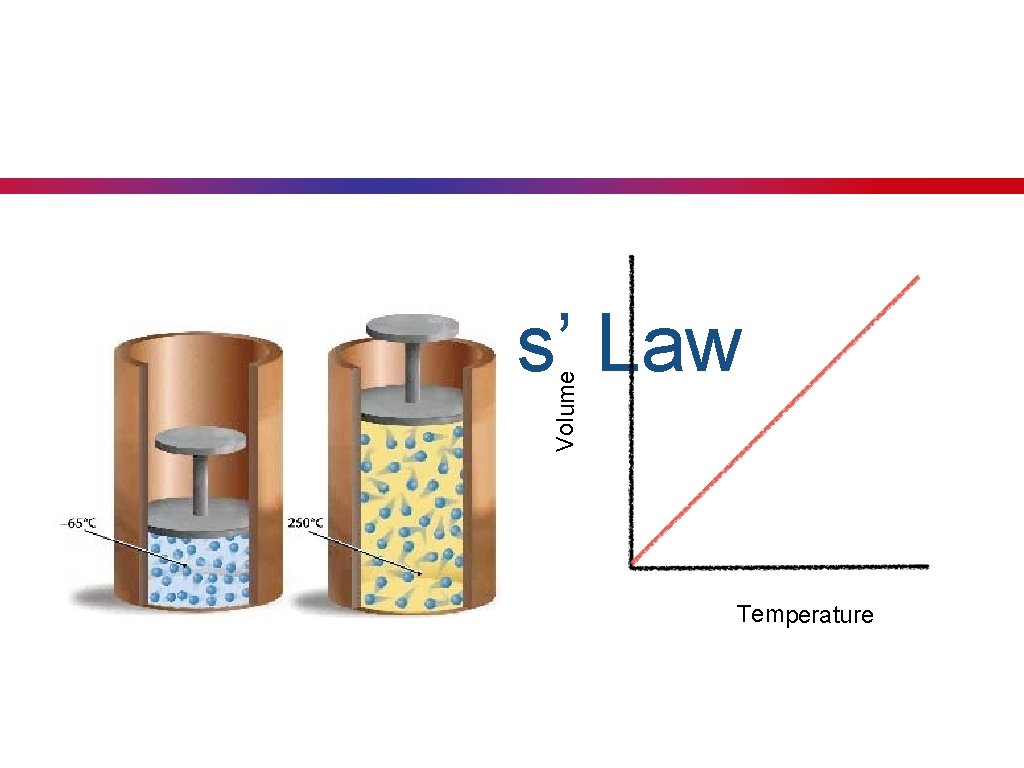
Volume Charles’ Law Temperature


Law of Gay-Lussac • Key Concept 25: The law of Gay. Lussac states that at a constant volume, the pressure exerted by a given mass of gas varies directly (positive relationship) with the absolute temperature. • As the temperature increases, the pressure will also increase. (directly proportional) P 1778 - 1850 T P T
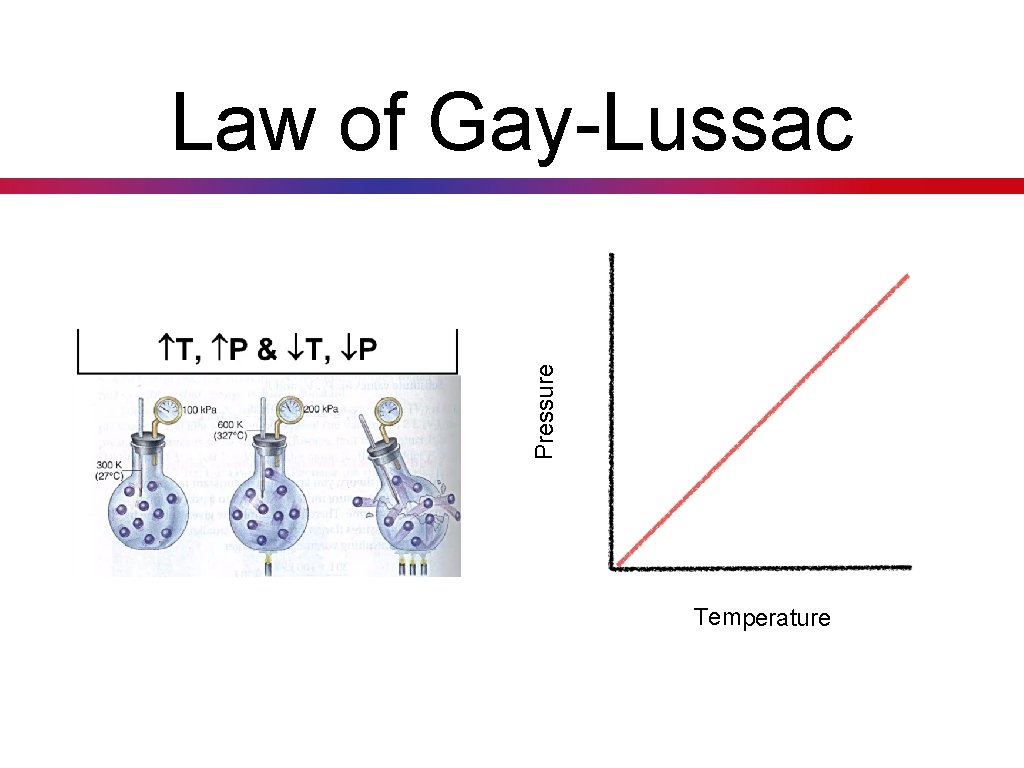
Pressure Law of Gay-Lussac Temperature


Key Concept 26: Theory • In science, a well-substantiated explanation of some aspect of the natural world that can incorporate facts, laws, inference, and tested hypotheses. • https: //en. wikipedia. org/wiki/Scientific_theory

Key Concept 27: Kinetic Molecular Theory • The molecules in a gas occupy no volume (that is, they are points) • These molecules are in constant, continuous, random, straightline motion • The molecules collide with one another, but the collisions are perfectly elastic (they result in no net loss of energy) • The pressure of a gas is the result of collisions between the gas molecules and the walls of the container • The temperature of a gas depends on its average kinetic energy

MUST BE KNOWN Boyles Law KC 18, 23 V. P. (Negative) You’re a little negative and Boyle with jealousy if you’re the V. P. Charles Law KC 17, 24 T. V. (Positive) Charlie Chapil was a positive TV character. Guy-Lussac Law KC 20, 25 T. P. (Positive) When you on the lu and run out of T. P. You can be positive and use a paper sacc.
Today: Practice Mathematical Relationships Question: WHow. Y did the tanker crush? Big Idea: How gases behave and what factors affect their behavior.

End of Day 17
- Slides: 159