Unit 2 Battery Construction Diagnosis Service and Safety

Unit 2 Battery Construction, Diagnosis, Service, and Safety

Introduction (1 of 2) • Electrical system relies on electrical power. – – – Engine and emissions systems Brakes and suspension Navigation Entertainment Information retrieval

Introduction (2 of 2) • Almost every system relies on: – Electrical and electronic components – Electronic control modules – Networking systems • Automobiles have: – 12 -volt storage battery – Engine starting system – Electrical charging system

The Battery (1 of 4) • Electricity is flexible and useful. – Drawback—not easily stored for later use • Must be stored in another form of energy and reconverted when needed

The Battery (2 of 4) • Developed in early 1800 s – Stores electricity in chemical form: • Electricity causes chemical reaction. • Changes chemical composition • Stored until electrical energy needed • When electricity needed, chemicals react and transform chemical back to electrical energy.
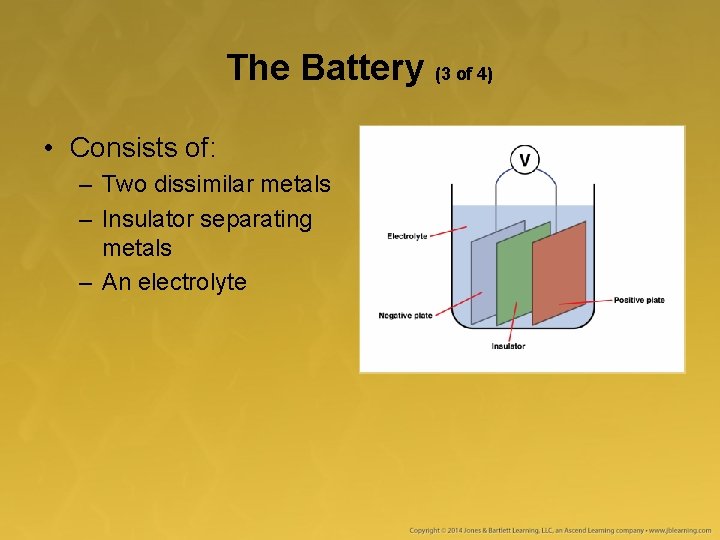
The Battery (3 of 4) • Consists of: – Two dissimilar metals – Insulator separating metals – An electrolyte

The Battery (4 of 4) • Lead-acid battery traditional type – Available in many shapes, sizes, and designs • Requires different design characteristics • Vehicle battery designed to give high current draws for short time periods • Deep cycle batteries—smaller, continuous loads over long periods of time

The Battery—Lead-Acid Flooded Cell and Gel Cell Batteries (1 of 8) • Wet cell lead-acid battery—main storage device in auto use – Lead plates immersed in water-acid electrolyte solution – High discharge currents with high voltage – High power output for compact size

The Battery—Lead-Acid Flooded Cell and Gel Cell Batteries (2 of 8) • 12 -volt car battery—six cells in series – 2. 1 volts in each cell; total 12. 6 volts – Each cell has: • 2 sets of plates in electrolyte solution – One set of lead dioxide

The Battery—Lead-Acid Flooded Cell and Gel Cell Batteries (3 of 8) • As battery discharges: – – Sulfuric acid is absorbed into lead plates. Both plates are turned into lead sulfate. Electrolyte strength becomes less acidic. Recharging battery reverses the process.

The Battery—Lead-Acid Flooded Cell and Gel Cell Batteries (4 of 8) • Conventional open wet cell battery – Charging generates hydrogen and oxygen gas by separating them from the electrolyte. – Highly explosive mix – Gassing from overcharging or rapid charging – Sulfuric acid highly corrosive and harmful • Use protective clothing and extra care.

The Battery—Lead-Acid Flooded Cell and Gel Cell Batteries (5 of 8) • Storage battery – Main device is a wet cell. – Nominal 2. 1 volts in each cell • Not dependent on cell size – Current capacity depends on size. – Plate surface area determines current capacity.

The Battery—Lead-Acid Flooded Cell and Gel Cell Batteries (6 of 8) • Positive/negative plates alternated in each cell – Both positive and negative plates connected in parallel – More plates, greater the current capacity

The Battery—Lead-Acid Flooded Cell and Gel Cell Batteries (7 of 8) • Six 2. 1 -volt cells connected in series – – Nominal voltage of 12. 6 volts Cells sealed from each other Filled with dilute sulfuric acid Battery case made of plastic or hard rubber

The Battery—Lead-Acid Flooded Cell and Gel Cell Batteries (8 of 8) • With cells in series – One end connected to negative posts – Other end connected to positive post

The Battery—Ratings and Sizing (1 of 6) • Different ratings and sizes to fit needs – Ratings—electrical specs of battery – Sizing—physical attributes of the battery • Size of battery case • Arrangement of battery terminals • Size or type of battery terminal post

The Battery—Ratings and Sizing (2 of 6) • Size designated by BCI, given group number – Individual groups specified by dimension in: • Length • Width • Height – Physically bigger, not necessarily higher capacity

The Battery—Ratings and Sizing (3 of 6) • Other designations— placing of positive and negative terminals – Varies with layout

The Battery—Ratings and Sizing (4 of 6) • Different battery post types: – Lug terminals – Side terminals – Standard posts

The Battery—Ratings and Sizing (5 of 6) • Electrical capacity determined by total surface area and plate thickness – More plate surface area = higher electrical capacity – Plates are standard size. • Increase number of plates per cell to increase surface area.

The Battery—Ratings and Sizing (6 of 6) • To rate automotive battery capacity, use: – Cold cranking amps (CCA) – Cranking amps (CA) – Reserve capacity • Ratings typically marked on batteries

The Battery—Low-Maintenance and Maintenance-Free Batteries and Cells (1 of 4) • Many battery and cell design variations for: – Starting – Extended-load marine usage • Low-maintenance batteries – Require little or now water topping in electrolyte – Plates and venting system do not gas – Removable caps

The Battery—Low-Maintenance and Maintenance-Free Batteries and Cells (2 of 4) • Maintenance-free batteries – – Fully sealed Do not require topping off of electrolytes Some use gel-type electrolyte instead of liquid. Absorbed glass mat-electrolyte absorbed within mat of fine glass fibers

The Battery—Low-Maintenance and Maintenance-Free Batteries and Cells (3 of 4) • Plates can be flat or wound in cylindrical cell. • Electrolyte a gel—handy for rough handling • Can be mounted on side • Suited for off-road and racing vehicles

The Battery—Low-Maintenance and Maintenance-Free Batteries and Cells (4 of 4) • Sealed or low-maintenance battery with no removable cell covers – Cannot adjust or test fluid levels – Some have visual indicators for status of charge and condition of one of the battery cells.

The Battery—Battery Charging and Discharging Cycle (1 of 6) • In discharged lead-acid cell: – – Active material of both plates becomes lead sulfate. Electrolyte becomes mostly water. Acid absorbed into the plate Results in very week sulfuric acid solution

The Battery—Battery Charging and Discharging Cycle (2 of 6) • When charging, battery connects to a DC electrical supply with voltage higher than the battery’s total cell voltage. – Acts like an electron pump—forces electrons to move from positive to negative plates

The Battery—Battery Charging and Discharging Cycle (3 of 6) • At the negative plates: – Sulfate is discharged. – Changes chemical composition of plates – Creates stronger sulfuric acid • Lead peroxide forms at positive plates, restoring voltage.

The Battery—Battery Charging and Discharging Cycle (4 of 6) • Charging increases acid amount in electrolyte. – Makes it stronger • When it no longer makes the electrolyte stronger, charging is complete.

The Battery—Battery Charging and Discharging Cycle (5 of 6) • Connecting a lead-acid battery to a load causes chemical changes at discharge. – Positive plate—sulfate joins with lead to form lead sulfate. – Negative plate—lead sulfate forms as sponge lead forms with sulfate.

The Battery—Battery Charging and Discharging Cycle (6 of 6) • Overall – – – Percentage of acid in electrolyte falls. Percentage of water rises. Strength of electrolyte falls. Plates develop same composition as cell discharges. Recharging restores the difference.

The Battery—Battery Temperature Monitoring (1 of 3) • Temperature affects battery performance. – – Ideal temperature range for each battery Cold weather—slows chemical reaction process Hot weather—overheating compromises lifetime Temperature probe in some batteries

The Battery—Battery Temperature Monitoring (2 of 3) • Power train control module controls levels of: – Current flow – Battery charge voltage • Colder temperature, higher rate of charging • Hotter temperature, slower rate of charging

The Battery—Battery Temperature Monitoring (3 of 3) • Smart chargers and battery management systems use feedback from temp probe. – Manages battery charge rate – Ensures battery at capacity and temperature within optimal range

The Battery—Battery Operating Conditions (1 of 2) • For best performance and lifetime – – Clean, dry, and fully charged Minimal vibration and correct level of electrolyte Moderate temperature Be well secured

The Battery—Battery Operating Conditions (2 of 2) • Lifetime shortened by: – – – – Fully discharged or deep discharge cycles Remaining over- or undercharged High discharge rates for extended periods Experiencing excessive vibration Exposed to temperature extremes Dirt or moisture around the case Developing corrosion

The Battery—Rechargeable Cell Batteries (1 of 8) • More efficient, higher-density batteries being developed – Nickel-cadmium (Ni-Cd) – Nickel-metal hydride (Ni-MH) – Lithium ion (Li-ion) • All are rechargeable cell batteries.

The Battery—Rechargeable Cell Batteries (2 of 8) • Ni-Cd batteries contain older technology. – – Have been replaced by Ni-MH batteries Both have nominal cell voltage of 1. 2 volts. Ni-MH— 2 or 3 times energy density of Ni-Cd Especially useful for drive motor applications

The Battery—Rechargeable Cell Batteries (3 of 8) • Lithium-ion newer type of rechargeable cell – Used in consumer electronic devices – Used in hybrid-electric or battery electrical vehicles with RESS – One of highest energy density ratios • Can store more energy • Low self-discharge rate • Shelf life up to 5 years

The Battery—Rechargeable Cell Batteries (4 of 8) • Cell voltage depends on material used to make the cell. – Typical voltage: • Lithium-ion battery— 3. 6 volts • Nickel-cadmium or nickel metal hydride— 1. 2 volts • Lead-acid battery— 2. 1 volts

The Battery—Rechargeable Cell Batteries (5 of 8) • Lithium-ion batteries have: – Anode – Cathode – Electrolyte • When discharging, lithium ions are removed from anode and added into the cathode.

The Battery—Rechargeable Cell Batteries (6 of 8) • Lithium-ion batteries have thermal runaway and cell rupture if overheated or overcharged. – May explode in extreme cases – Take extreme care in handling or charging.

The Battery—Rechargeable Cell Batteries (7 of 8) • In hybrid or electric vehicles: – Many small dry cell battery-sized cells are connected in series and in parallel arrangements to form a battery pack. – RESS battery packs develop voltage in excess of 200 volts.

The Battery—Rechargeable Cell Batteries (8 of 8) • Advantages of Li-ion batteries – – – High energy density Low self-discharge Low maintenance No memory Low internal resistance • Disadvantages of Li-ion batteries – Need for circuit protection – Limited shelf life – High temp sensitivity – Increased cost

The Battery—Battery Cables and Terminals (1 of 2) • Carry high discharge currents to crank engine • Number of designs: – Cone design – Side terminal – Flat terminal with hole in center

The Battery—Battery Cables and Terminals (2 of 2) • Cables made from copper wire strands bound together and insulated – High current capacity – Different cable sizes – Heat-shrink tubing and sealing adhesive over joints

Summary • The primary components of the battery are the case, cover, vent caps, plates, separators, electrolyte, and terminals. • Batteries operate by storing electrical energy in chemical form. • The discharge of electrolytes produces battery current.

Summary • The standard automotive battery has 2. 1 volts per cell, with six cells holding 12. 6 volts on a fully charged battery. • Batteries are classified into groups, and only certain groups and layouts will fit certain vehicle applications. • Batteries are rated by electrical capacity, cold cranking amps, and amp hour ratings.

Summary • There are two chemicals make a battery a safety hazard: hydrogen gas and sulfuric acid. • The absorbed-glass mat battery is good for rough handling and can be mounted on its side compared to the electrolyte battery. • Battery temperature plays a critical part in battery life. When a battery is cold, it takes more power to start the vehicle; when a battery is too hot, its life is diminished significantly.

Summary • Lithium-ion batteries are the future and are especially being used in hybrids. However, they have not replaced the conventional battery in most applications. • There are two ways to connect the battery to the vehicle: the top-post design and the side-post design.

Credits • Unless otherwise indicated, all photographs and illustrations are under copyright of Jones & Bartlett Learning.
- Slides: 51