Unit 2 Atomic Structure Nuclear Chemistry new book

Unit 2 Atomic Structure & Nuclear Chemistry ‘new’ book: Ch 2

Early Theories of Matter o Democritus (460 -370 B. C. ) proposed & believed that n n n Matter was not infinitely divisible Matter is made up of tiny particles called atomos (“uncuttable”) Atoms could not be created, destroyed, or further divided Democritus’ model of atom Solid and INDESTRUCTABLE “Billiard Ball” Model

Development of Chemistry Robert Boyle o Beginnings of modern chemistry were seen in the 16 th and 17 th centuries, where great advances were made in metallurgy, the extraction of metals from ores. o In the 17 th century, Robert Boyle described the relationship between the pressure and volume of air and defined an element as a substance that cannot be broken down into two or more simpler substances by chemical means.

Development of Chemistry o During the 18 th century, Priestley discovered oxygen gas and the process of combustion where carbon-containing materials burn vigorously in an oxygen atmosphere. Priestley

Development of Chemistry Lavoisier o In the late 18 th century, Lavoisier wrote the first modern chemistry text. His most important contribution was the law of conservation of mass, which states that in any chemical reaction, the mass of the substances that react equals the mass of the products that are formed. n He is known as the father of modern chemistry.

Development of Chemistry o In the 19 th century, John Dalton revised Democritus's ideas based upon the results of scientific research he conducted n Led to Dalton’s atomic theory Dalton

Dalton’s Atomic Theory 1. Elements are composed of tiny indivisible particles called atoms 2. Atoms of the same element are identical. The atoms of any one element are different from those of any other element.

Dalton’s Atomic Theory 3. Atoms of different elements can physically mix together or can chemically combine with one another in simple whole-number ratios to form compounds.

Dalton’s Atomic Theory 4. Chemical reactions occur when atoms are separated, joined, or rearranged. n Atoms of one element, however, are never changed into atoms of another element as a result of a chemical reaction.
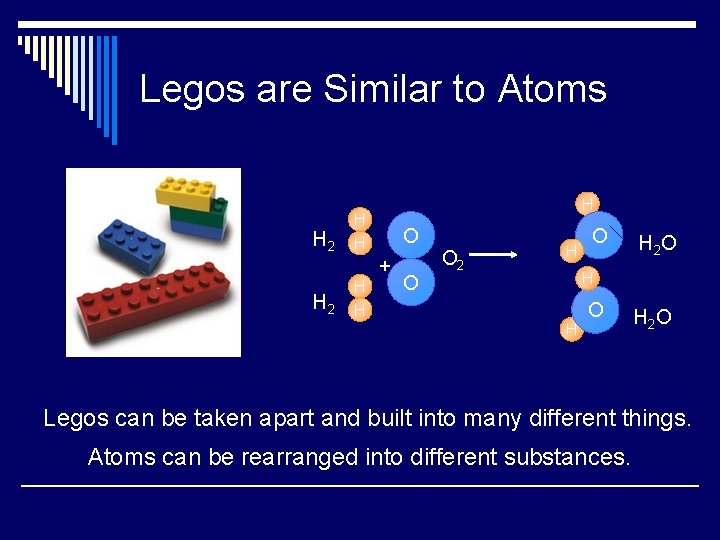
Legos are Similar to Atoms H 2 H H H O + H 2 H H O O 2 H O H 2 O H H O H 2 O Legos can be taken apart and built into many different things. Atoms can be rearranged into different substances.

Foundations of Atomic Theory Law of Conservation of Mass is neither destroyed nor created during ordinary chemical reactions. Law of Definite Proportions The fact that a chemical compound contains the same elements in exactly the same proportions by mass regardless of the size of the sample or source of the compound.

Law of Definite Proportions Joseph Louis Proust (1754 – 1826) o Each compound has a specific ratio of elements o It is a ratio by mass o EX: Water is always 8 grams of oxygen for every one gram of hydrogen

Law of Definite Proportions o Whether synthesized in the laboratory or obtained from various natural sources, copper (II) carbonate always has the same composition. o Analysis of this compound led Proust to formulate the law of definite proportions. + 103 g of copper (II) carbonate 53 g of copper + 40 g of oxygen 10 g of carbon

Structure of Atoms o Scientists began to wonder what an atom was like. o Was it solid throughout with no internal structure or was it made up of smaller, subatomic particles? o It was not until the late 1800’s that evidence became available that atoms were composed of smaller parts.

Radioactivity o One of the pieces of evidence that atoms are made of smaller particles came from the work of Marie Curie (1876 - 1934). o She discovered radioactivity, the spontaneous disintegration of some elements into smaller pieces. Marie Curie

Discovery of the Electron o J. J. Thomson (1856 – 1940) performed experiments that involved passing electric current through gases at low pressure n Thomson n He sealed the gases in glass tubes fitted at both ends with metal disks called electrodes Electrodes were connected to a source of high-voltage electricity

Cathode-Ray Experiment o One electrode, the anode, became positively charged o The other electrode, the cathode, became negatively charged o A glowing beam formed between the 2 electrodes (called a cathode ray)
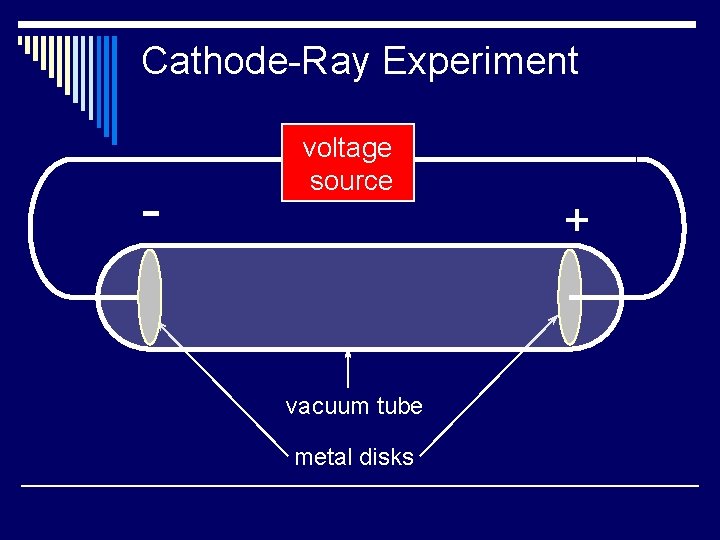
Cathode-Ray Experiment - voltage source vacuum tube metal disks +
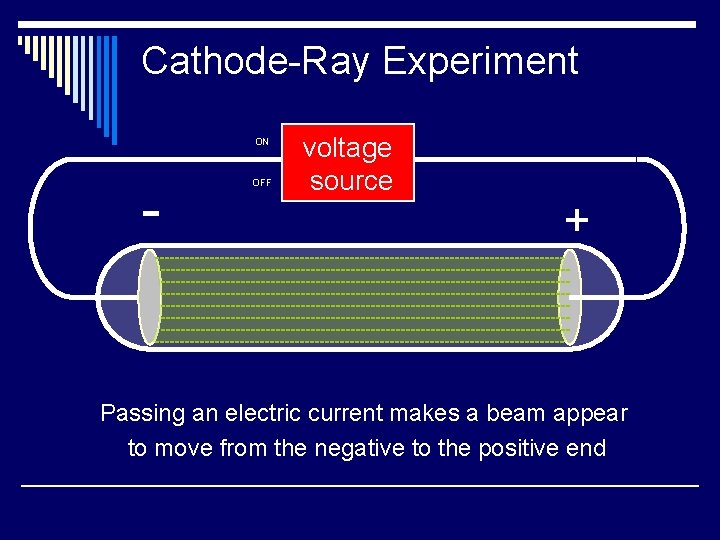
Cathode-Ray Experiment ON - OFF voltage source + Passing an electric current makes a beam appear to move from the negative to the positive end
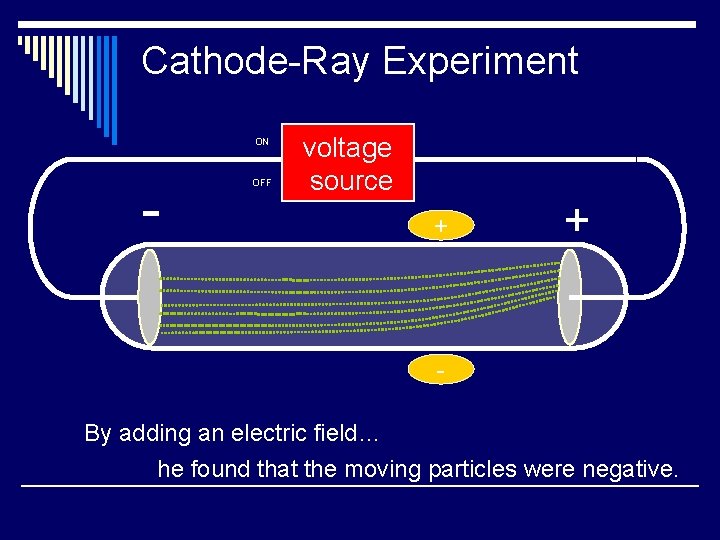
Cathode-Ray Experiment ON - OFF voltage source + + By adding an electric field… he found that the moving particles were negative.

Thomson’s Findings o Cathode rays are attracted to positively charged metal plates and repelled by negatively-charged plates o He proved that atoms contain tiny negative particles (electrons) and concluded that ALL atoms must contain these negative particles. o He knew that atoms were neutral in charge and deduced that there must be a positive charge within the atom.

Plum-Pudding Model

Discovery of the Proton o Goldstein discovered the proton using the cathode ray tube in a similar way as did Thomson and the electron

Discovery of the Nucleus o Ernest Rutherford (1871 - Rutherford 1937) learned physics in J. J. Thomson’ lab. o Noticed that ‘alpha’ particles were sometimes deflected by something in the air. n n Alpha particles are helium nuclei Alpha particles are positivelycharged o Gold-foil experiment

Rutherford’s Gold Foil Experiment o Alpha particles were fired at a thin sheet of gold foil o Particle-hits on the detecting screen (film) are recorded
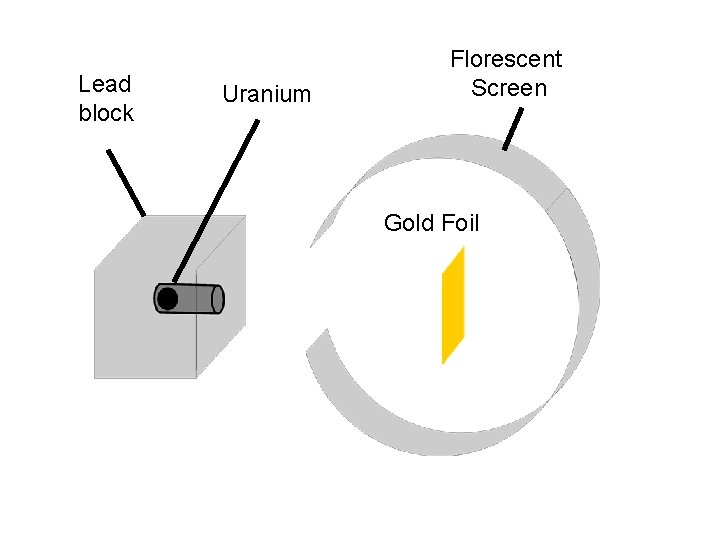
Lead block Uranium Florescent Screen Gold Foil

What he expected…
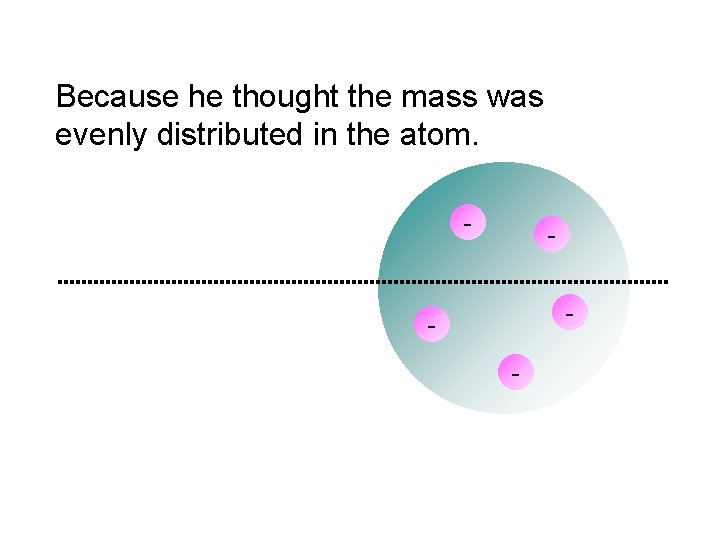
Because he thought the mass was evenly distributed in the atom. - - -

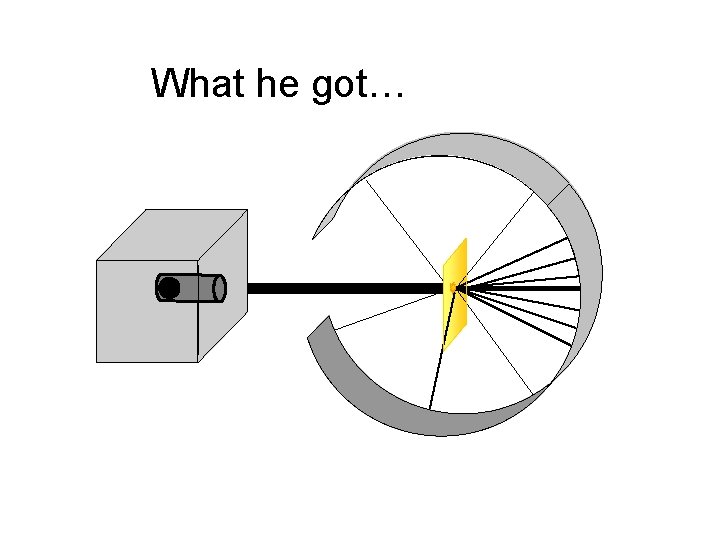
What he got…
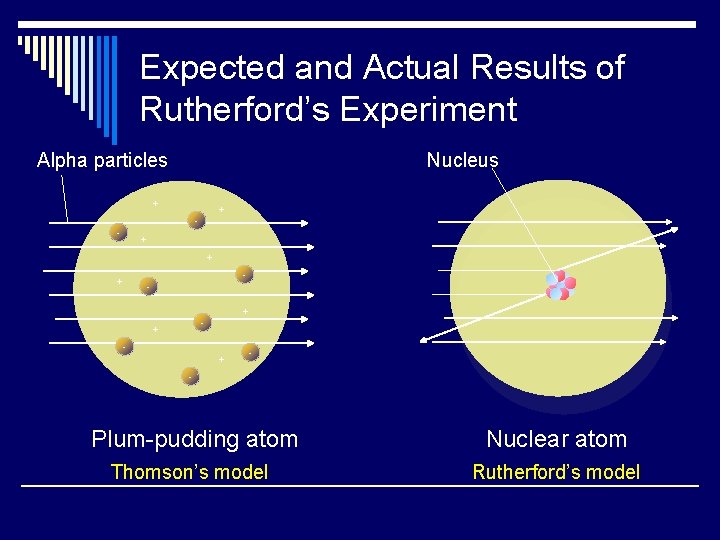
Expected and Actual Results of Rutherford’s Experiment Alpha particles Nucleus + + - - + + - + - - Plum-pudding atom Nuclear atom Thomson’s model Rutherford’s model

Try it Yourself! o In the following pictures, there is a target hidden by a cloud. To figure out the shape of the target, we shot some beams into the cloud and recorded where the beams came out. Can you figure out the shape of the target?
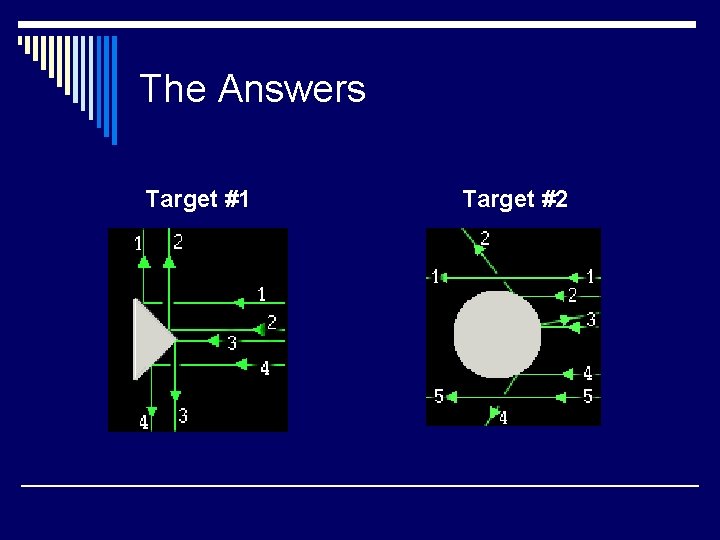
The Answers Target #1 Target #2

Rutherford’s Findings o Most of the particles passed right through o A few particles were deflected o VERY FEW were greatly deflected “Like howitzer shells bouncing off of tissue paper!” Conclusions: q. The atom is mostly empty space q. The nucleus is small q The nucleus is dense q The nucleus is positively charged

Size of the Nucleus o If an atom is as large as a stadium, then the nucleus is about the size of a fly in the center of the stadium!!!
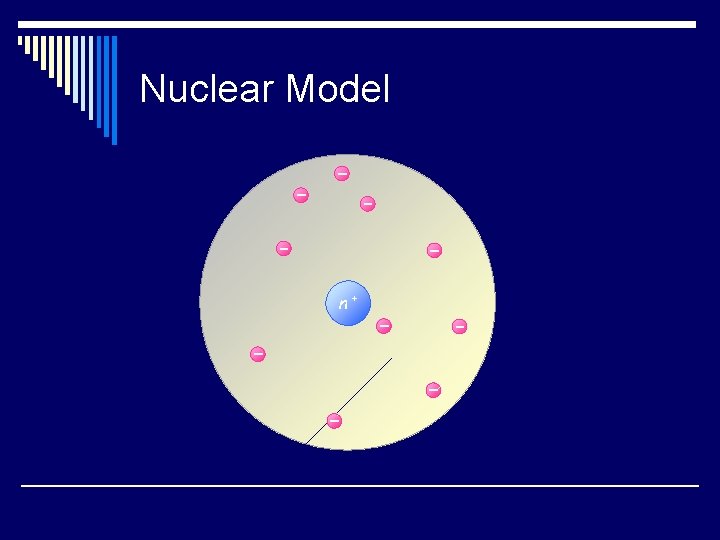
Nuclear Model n+
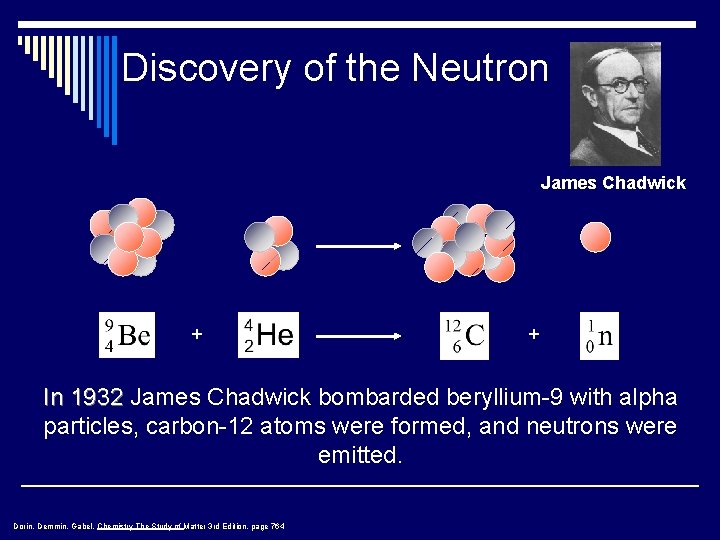
Discovery of the Neutron James Chadwick + + In 1932 James Chadwick bombarded beryllium-9 with alpha particles, carbon-12 atoms were formed, and neutrons were emitted. Dorin, Demmin, Gabel, Chemistry The Study of Matter 3 rd Edition, page 764

The Modern View of Atomic Structure o The atom contains: n electrons n protons: found in the nucleus, they have a positive charge equal in magnitude to the electron’s negative charge. n neutrons: found in the nucleus, virtually same mass as a proton but no charge.

The Mass and Charge of the Electron, Proton, and Neutron

The Chemists’ Shorthand: Nuclear Symbols Mass number Atomic number 39 K 19 Element Symbol

Atomic Number o Equal to the number of protons o Equal to the number of electrons in an atom o Determines the element!

Mass Number o mass # = protons + neutrons o always a whole number o NOT on the Periodic Table!

Ions o Atoms that have lost or gained electrons n Cation – positive ion (lost electrons) p Example: How many electrons does Na 1+ have? n Anion – negative ion (gained electrons) p Example: have? How many electrons does S 2 -

PRACTICE WITH IONS Find the n n number of protons = 55 number of neutrons = 78 number of electrons = 54 Atomic number = 55 Mass number = 133 Cs 133 55 Cesium ion 1+

Isotopes o Atoms of the same element with different mass numbers. 12 6 C carbon-12 14 6 C carbon-14 o Isotopes are atoms with the same number of protons but different number of neutrons

PRACTICE o Atomic Number & Mass Number WS

Calculating Relative Atomic Mass https: //www. youtube. com/watch? v =Sdh. LTfma_Eg

Relative Atomic Mass https: //www. youtube. com/watch? v=vq. Tg 4 c. Yw. HXY o 12 C atom = 1. 992 × 10 -23 g (*watch above video!) o atomic mass unit (amu) o 1 amu o 1 p 1 n 1 e- = 1/12 the mass of a 12 C atom = 1. 007276 amu = 1. 008665 amu = 0. 0005486 amu

Average Atomic Mass For help: http: //www. docbrown. info/page 04/4_73 calcs 01 ram. htm o weighted average of all isotopes o on the Periodic Table o round to 2 decimal places! Avg. = (mass x relative abundance) + (mass Atomic x relative abundance) +…. Mass % divided by 100 or part over whole
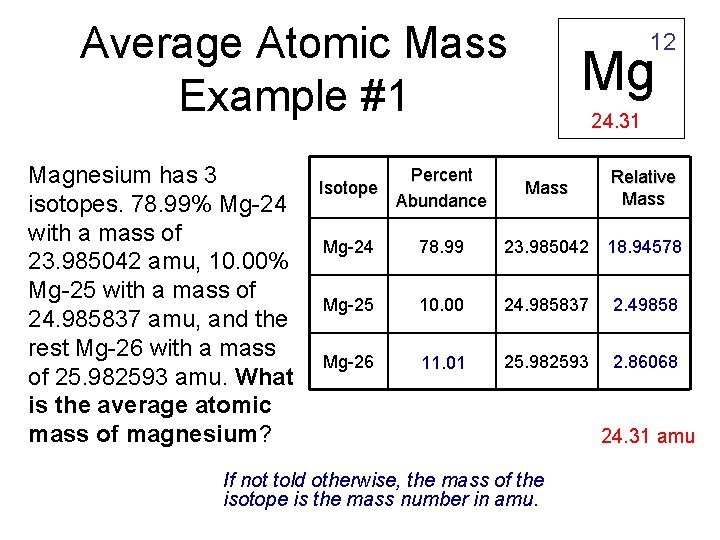
Average Atomic Mass Example #1 Magnesium has 3 isotopes. 78. 99% Mg-24 with a mass of 23. 985042 amu, 10. 00% Mg-25 with a mass of 24. 985837 amu, and the rest Mg-26 with a mass of 25. 982593 amu. What is the average atomic mass of magnesium? 12 Mg 24. 31 Isotope Percent Abundance Mg-24 78. 99 23. 985042 18. 94578 Mg-25 10. 00 24. 985837 2. 49858 Mg-26 11. 01 25. 982593 2. 86068 Mass If not told otherwise, the mass of the isotope is the mass number in amu. Relative Mass 24. 31 amu

Average Atomic Mass Example #2 o EX: Calculate the avg. atomic mass of oxygen if its abundance in nature is 99. 76% 16 O, 0. 04% 17 O, and 0. 20% 18 O. Avg. Atomic Mass = 16. 00 amu

What’s the difference between mass number and average atomic mass? o Mass number- specifically about one isotope o Average atomic mass- includes the masses of all the different isotopes for that atom

Atomic Structure o ATOMS n Differ by number of protons o IONS n Differ by number of electrons o ISOTOPES n Differ by number of neutrons

Intro to Periodic Table (Book: Section 2. 3) o Distinguish between: n Groups and periodsp Group # of ‘A’ elements gives valence e(available for bonding); Can use this to determine the ‘charge’ (oxidation #) the atom needs to form a perfect ‘octet’ p Period # gives the # of energy levels where electrons ‘reside’. n Metals and Nonmetals (and metalloids) Where are they on PT?

Intro to Bonding (Book: Section 2. 4) o Distinguish between: Molecule and Ion o Molecule: Covalent bond between atoms. Share e-. Usually between nonmetals and H. Represent with molecular formula which shows ratios using subscripts. (Ex: CH 4) o Molecular Structural formula: Each bond between atom represents a shared pair of e-. o Molecules are weak electrolytes (don’t ionize in solution). Ex- sugar and water o What is a binary compound or molecule?

Intro to Bonding- con’t o Ionic: Usually between metal and nonmetal. Give and take of e- to form cations and anions. Compounds held together by strong intermolecular forces. Most are strong electrolytes in water. Ex of ionic binary compound: Na. Cl o Some ionic compounds are ternary (more than 2 atoms): Ex-Na. OH. Na is 1+ and the (OH) is 1 -. The OH is actually covalently bonded and carries overall 1 - charge, BUT the Na+ is ionically bonded to the (OH)-.

Note: o You don’t have to know charges/ oxidation #’s for this test, except for the 5 polyatomic ions given on next slide

Polyatomic Ions o Group of covalently bonded (share e-) atoms carrying a charge. Will learn how this is done in the next unit o Start learning some common polyatomic ions and their charges. For this test- see table 2. 2 in your book and know the formula name and charge of these 5: ammonium, hydroxide, carbonate, sulfate, phosphate. You will have to know them all for the next test.

HW- ‘New’ Book o Go over Summary Problem p. 47 (omit h and i) o From p. 47 - 50 (end of Ch 2): #3, 6, 8, 10, 14, 16, 18, 20, 22, 26, 44, 49, 50, 52 o Make sure you do fill in notes from PPt (especially on the individuals and their contributions to chemistry) o Go over all worksheets and PPt’s
- Slides: 59