Unit 2 3 Carbohydrates and Lipids Outline the

Unit 2. 3: Carbohydrates and Lipids

• • • Outline the role of condensation and hydrolysis in the relationships between monosaccharides, disaccharides and polysaccharides List 3 examples each of monosaccharides, disaccharides and polysaccharides • Compare the structure and function of cellulose and starch in plants and glycogen in humans. • Outline the variation in the molecular structure of fatty acids, cis- and trans- unsaturated fatty acids, monounsaturated and polyunsaturated fatty acids. • Outline the role of condensation and hydrolysis in the relationship between fatty acids, glycerols and triglyceride • • Evaluate the health consequences of diets rich in the various types of fatty acid Calculate body mass index (BMI) from the body mass and height of a person. • Distinguish, using the body mass index, between being underweight, normal weight, overweight and obese. • Compare the use of carbohydrates and lipids in energy storage

Functions of CHOs Energy Storage: monosaccharides: glucose, fructose disaccharides: sucrose, maltose polysaccharides: starch, glycogen Structural molecules: Polysaccharides: cellulose, chitin Recognition or signaling molecules: Oligosaccharides: glycoproteins, glycolipids

Basic structure of CHOs a result of three factors: 1. Number of carbon atoms 2. Location of the functional groups attached to each carbon 3. Orientation of functional groups around the carbons

Number of Carbons/Location of Functional Groups
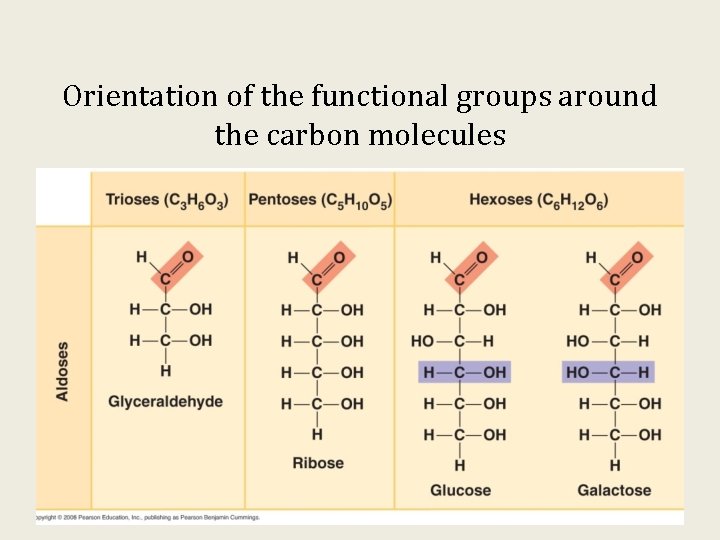
Orientation of the functional groups around the carbon molecules
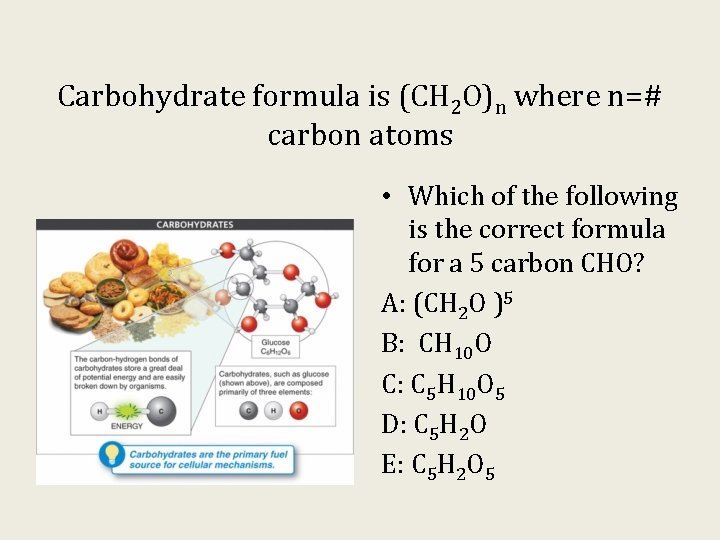
Carbohydrate formula is (CH 2 O)n where n=# carbon atoms • Which of the following is the correct formula for a 5 carbon CHO? A: (CH 2 O )5 B: CH 10 O C: C 5 H 10 O 5 D: C 5 H 2 O E: C 5 H 2 O 5
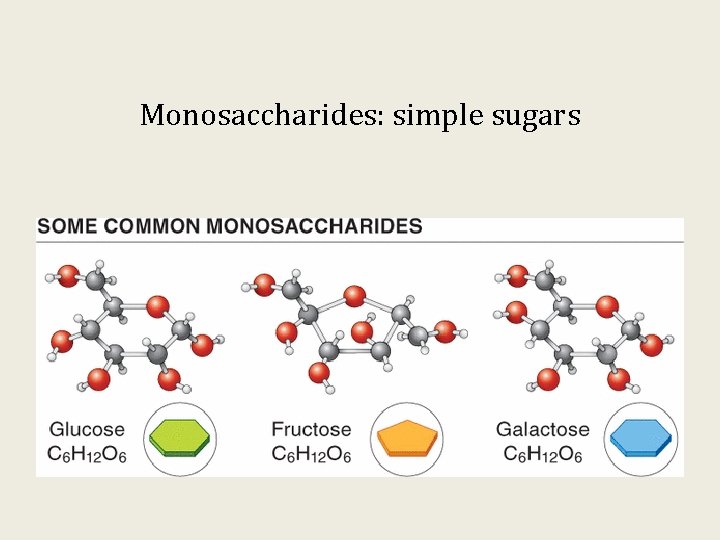
Monosaccharides: simple sugars
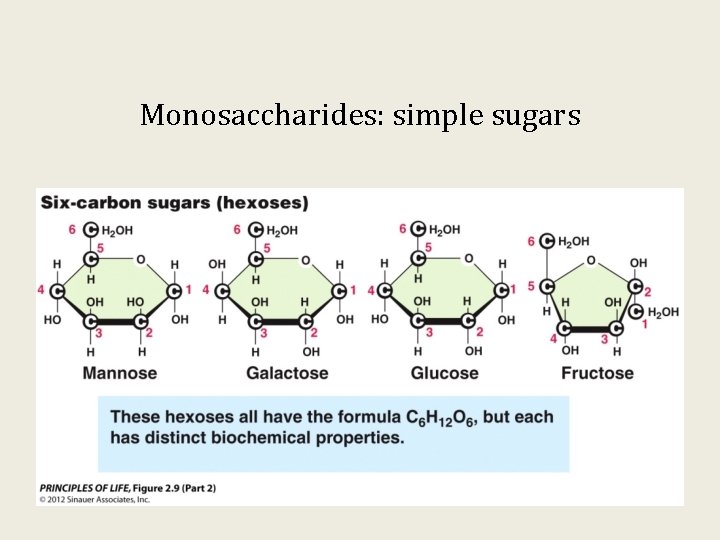
Monosaccharides: simple sugars
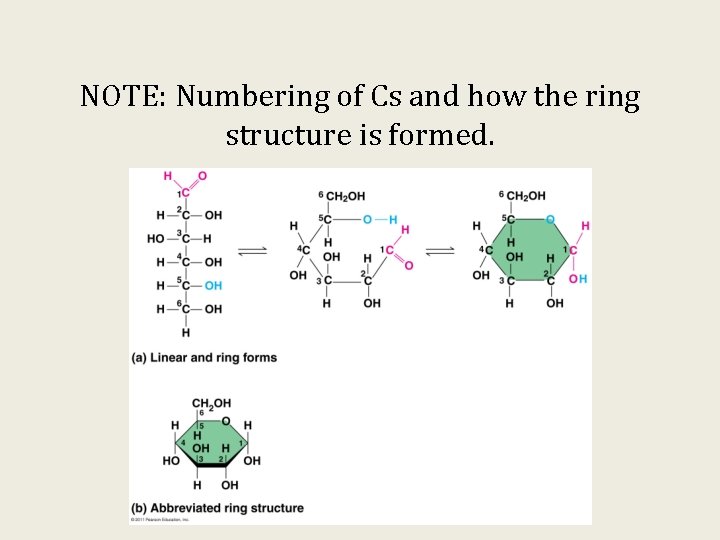
NOTE: Numbering of Cs and how the ring structure is formed.
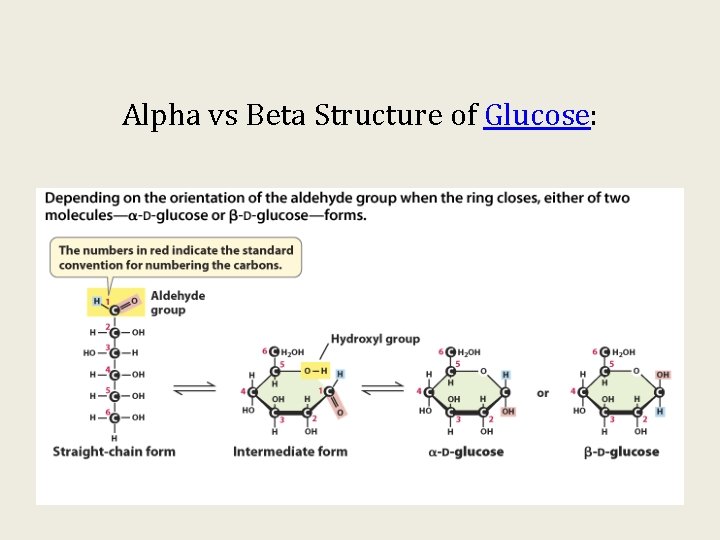
Alpha vs Beta Structure of Glucose:
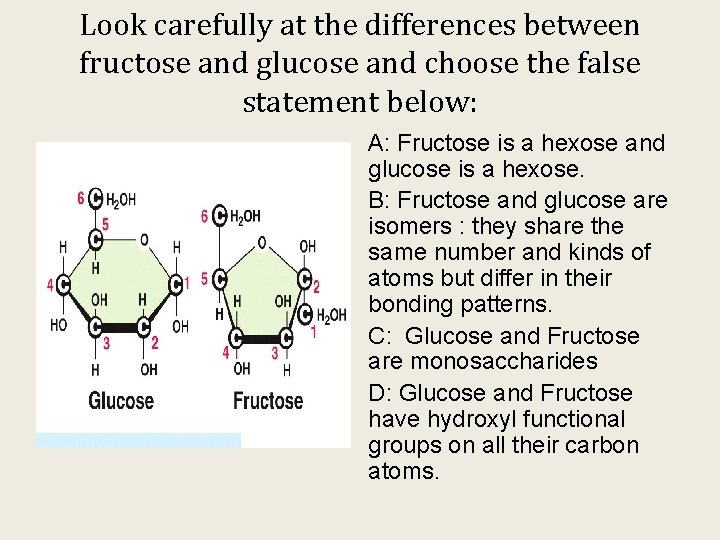
Look carefully at the differences between fructose and glucose and choose the false statement below: A: Fructose is a hexose and glucose is a hexose. B: Fructose and glucose are isomers : they share the same number and kinds of atoms but differ in their bonding patterns. C: Glucose and Fructose are monosaccharides D: Glucose and Fructose have hydroxyl functional groups on all their carbon atoms.
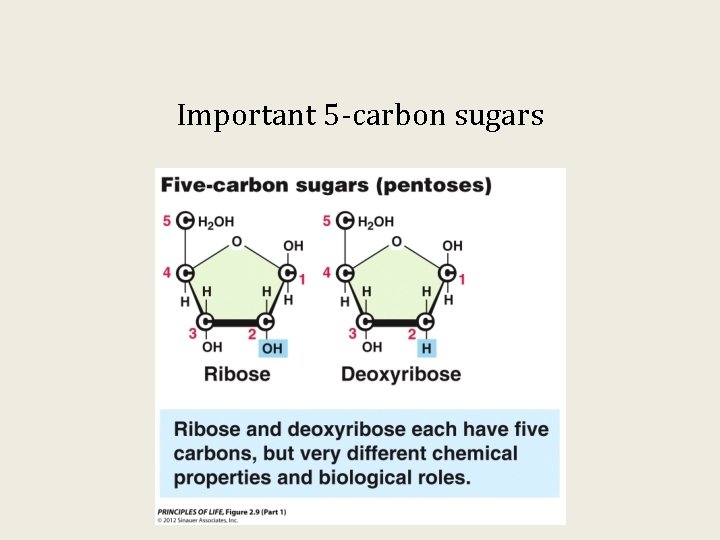
Important 5 -carbon sugars
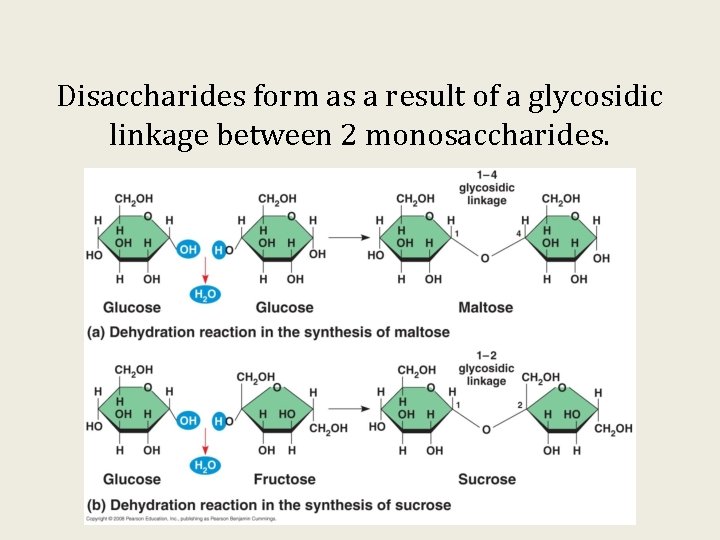
Disaccharides form as a result of a glycosidic linkage between 2 monosaccharides.
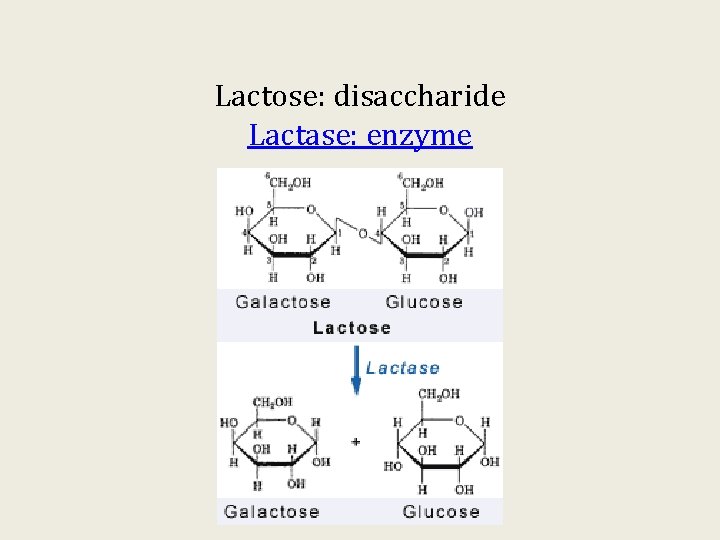
Lactose: disaccharide Lactase: enzyme

Nutritional labels provide information on relative content of CHOs This food item is most likely… A: Ice Cream B: Bacon C: Candy Bar D: Tofu Dog E: French Fries
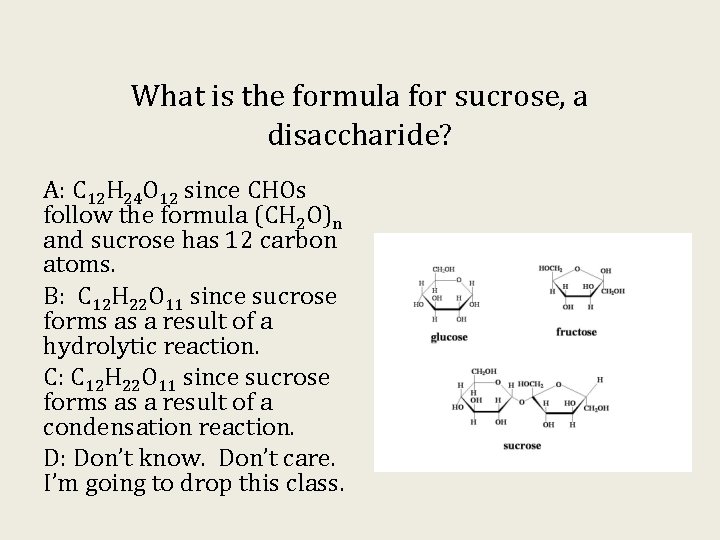
What is the formula for sucrose, a disaccharide? A: C 12 H 24 O 12 since CHOs follow the formula (CH 2 O)n and sucrose has 12 carbon atoms. B: C 12 H 22 O 11 since sucrose forms as a result of a hydrolytic reaction. C: C 12 H 22 O 11 since sucrose forms as a result of a condensation reaction. D: Don’t know. Don’t care. I’m going to drop this class.

Polysaccharides are complex carbohydrates: Cellulose, Starch, and Glycogen
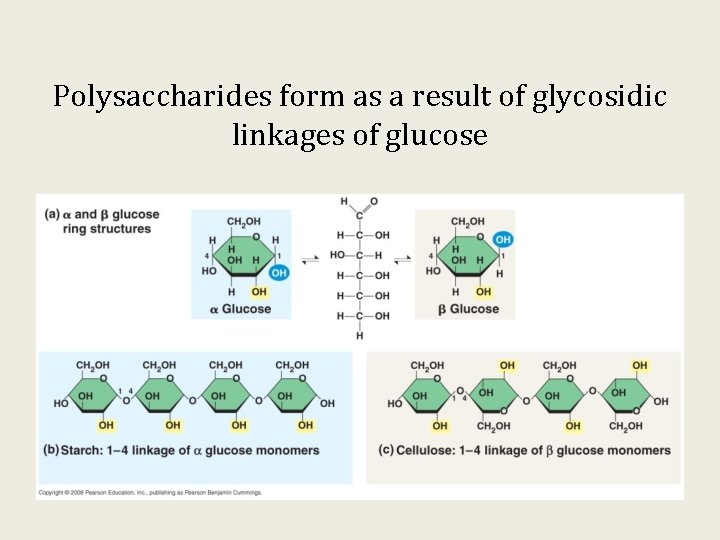
Polysaccharides form as a result of glycosidic linkages of glucose
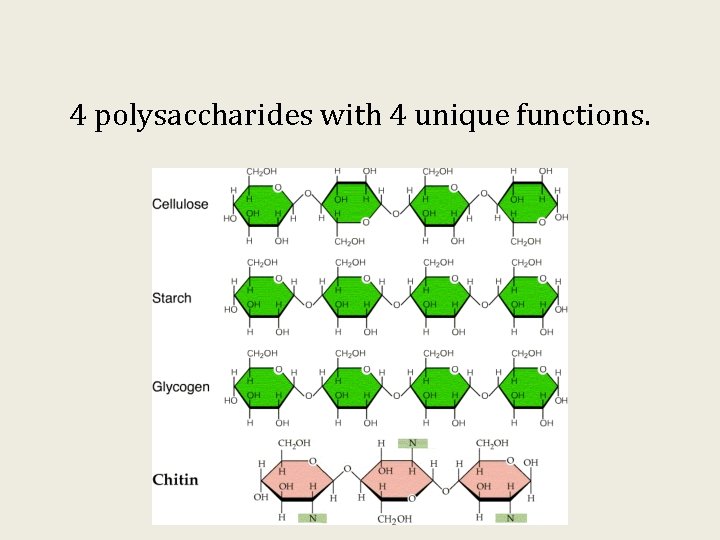
4 polysaccharides with 4 unique functions.

Glycogen: Animal polysaccharide Starch: Plant polysaccharide
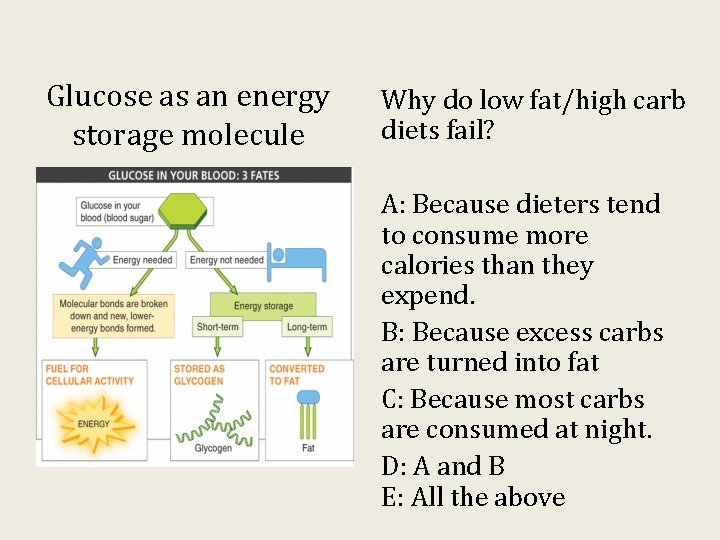
Glucose as an energy storage molecule Why do low fat/high carb diets fail? A: Because dieters tend to consume more calories than they expend. B: Because excess carbs are turned into fat C: Because most carbs are consumed at night. D: A and B E: All the above

Cellulose and Chitin: Structural polysaccharides

Cellulose is indigestible and is called fiber in our diet.

Chitin: a structural polysaccharide
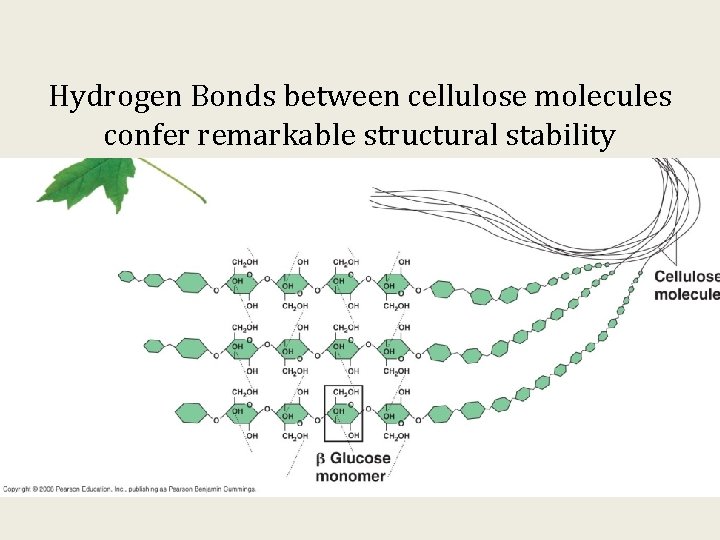
Hydrogen Bonds between cellulose molecules confer remarkable structural stability
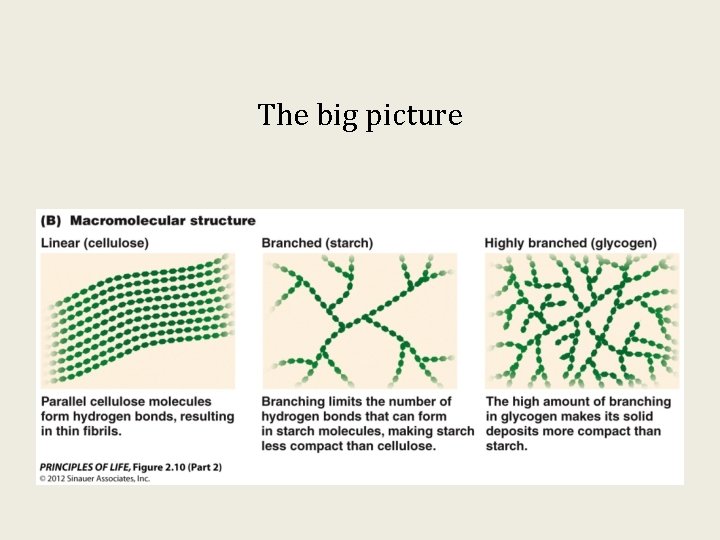
The big picture
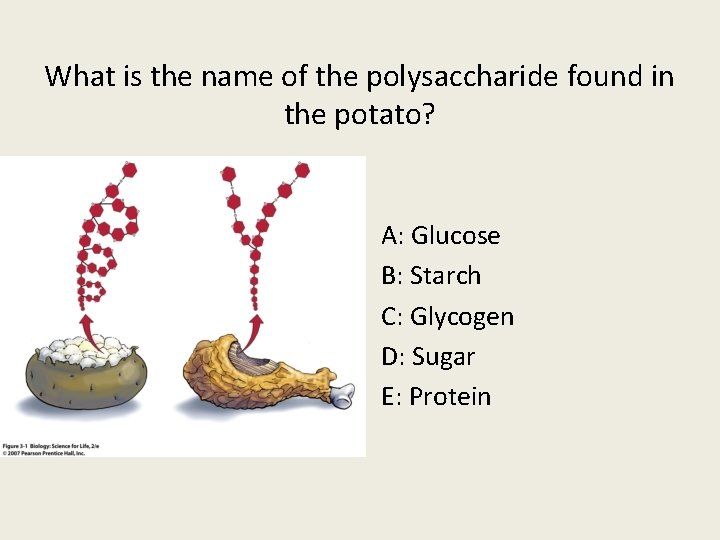
What is the name of the polysaccharide found in the potato? A: Glucose B: Starch C: Glycogen D: Sugar E: Protein
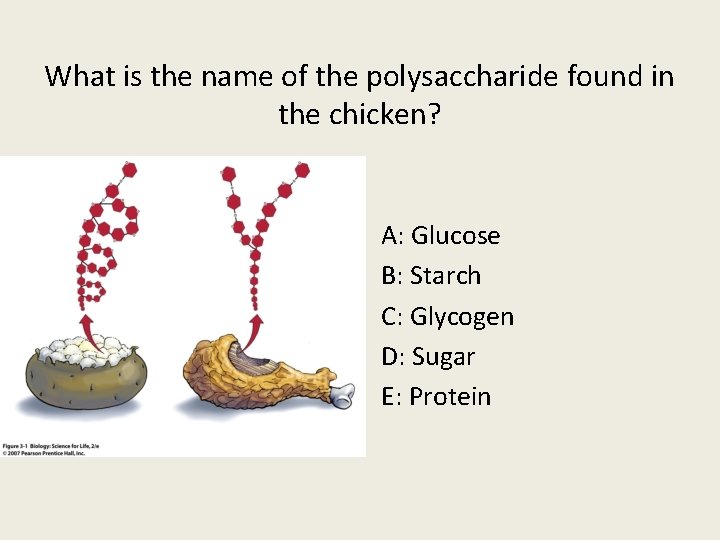
What is the name of the polysaccharide found in the chicken? A: Glucose B: Starch C: Glycogen D: Sugar E: Protein

Carbohydrates a. can have the same chemical formula, but distinct chemical properties and different biological roles. b. such as polysaccharides are formed when monosaccharides are ionically bonded by condensation reactions. c. are made of carbon, hydrogen, and oxygen. d. are always linear, unbranched molecules. e. Both a and c

Lipids: Fats, Sterols and Phospholipids • Long term energy storage: fats and oils • Regulate growth and Development: sterols • Structural role in cell membranes: phospholipids and cholesterol

Using the data from this nutrional label, calculate the energy storage capacity (in Cal) of fats versus CHOs. A: CHOs have about 4 cal/gram whereas fats contain about 9 cal/gram. B: CHOs are about 8% calories whereas fats are about 18% calories. C: CHOs have about 25 calories whereas fats contain about 12 calories. D: I have no idea!

Fats and Oils have more calories because they have more C-H bonds than glucose or starch.
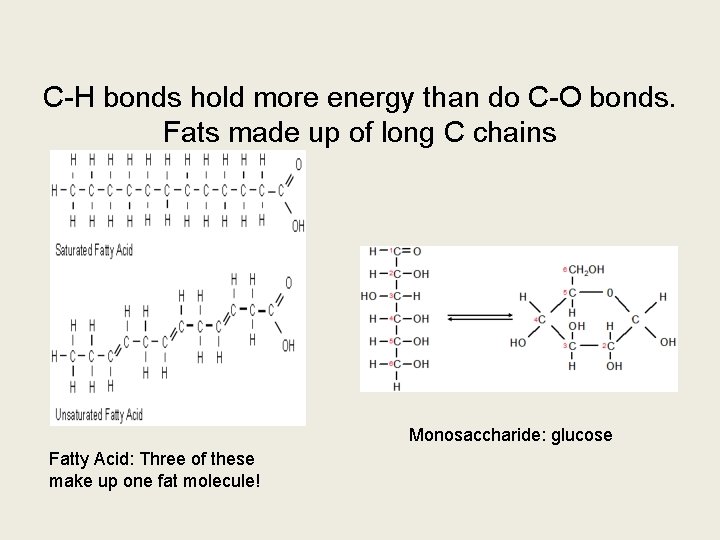
C-H bonds hold more energy than do C-O bonds. Fats made up of long C chains Monosaccharide: glucose Fatty Acid: Three of these make up one fat molecule!
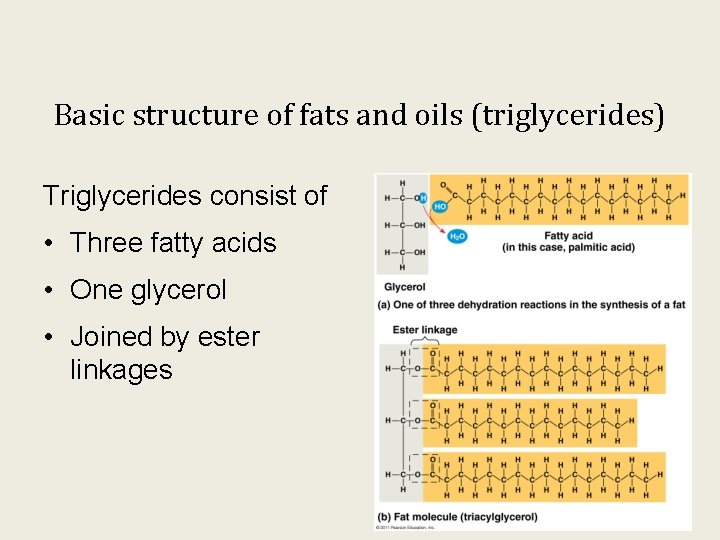
Basic structure of fats and oils (triglycerides) Triglycerides consist of • Three fatty acids • One glycerol • Joined by ester linkages

All fats are not the same!

Fatty acids differ in their length and number of C=C double bonds or saturation.
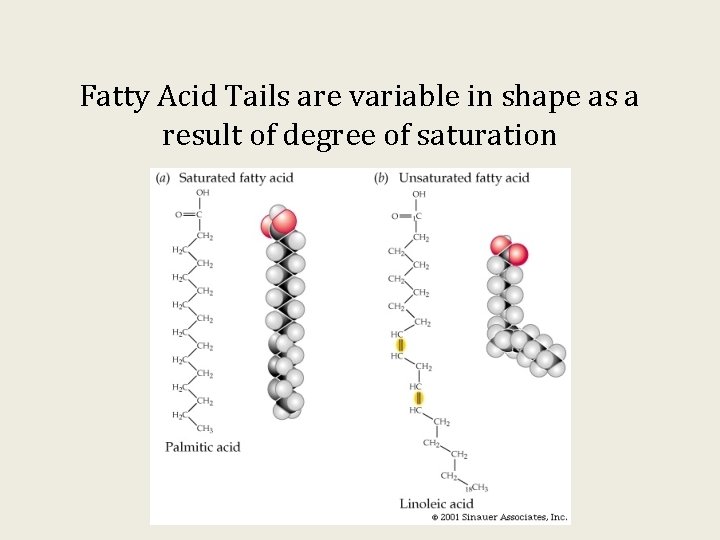
Fatty Acid Tails are variable in shape as a result of degree of saturation
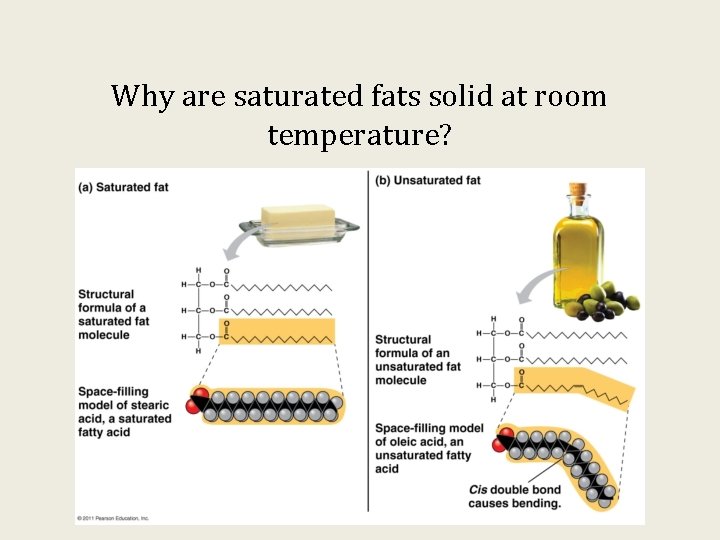
Why are saturated fats solid at room temperature?

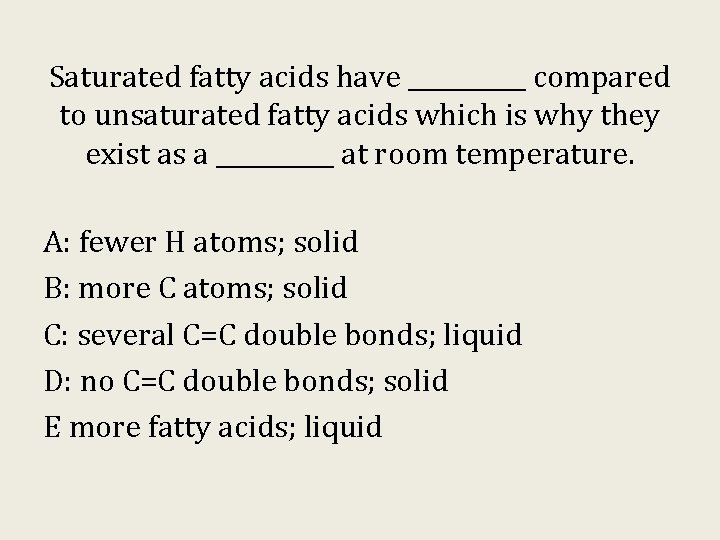
Saturated fatty acids have _____ compared to unsaturated fatty acids which is why they exist as a _____ at room temperature. A: fewer H atoms; solid B: more C atoms; solid C: several C=C double bonds; liquid D: no C=C double bonds; solid E more fatty acids; liquid

Trans fat: REALLY BAD
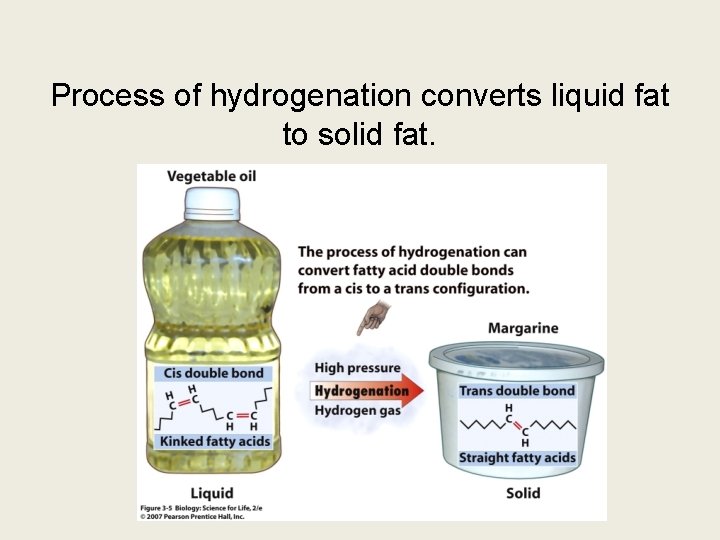
Process of hydrogenation converts liquid fat to solid fat.
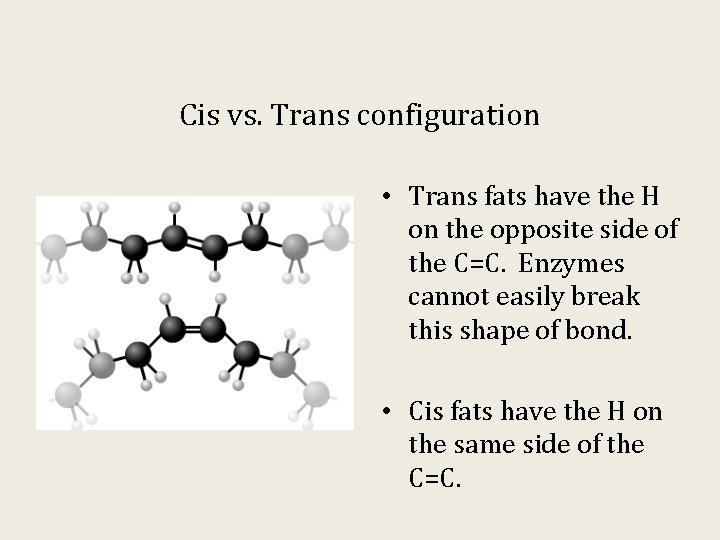
Cis vs. Trans configuration • Trans fats have the H on the opposite side of the C=C. Enzymes cannot easily break this shape of bond. • Cis fats have the H on the same side of the C=C.

Sources of Fats: • Saturated Fats: animal fats, coconut, palm and palm kernel oils, fully hydrogenated oils • Unsaturated Fats: plant oils, fatty fish, • Trans Fats: partially hydrogenated oils

What is a major difference between saturated and unsaturated fats? A. At room temperature, saturated fats are liquid and unsaturated fats are solid. B. Unsaturated fats have a flat molecular structure, but saturated fats are kinked. C. Saturated fat molecules are packed together loosely, whereas unsaturated fats are tightly packed. D. Unsaturated fats contain carbon-to-carbon double bonds, but saturated fats do not.

Lipids: Fats, Sterols and Phospholipids • Store a lot of energy in C—H bonds: fats and oils • Regulate growth and development: sterols • Structural role in cell membranes: phospholipids and cholesterol
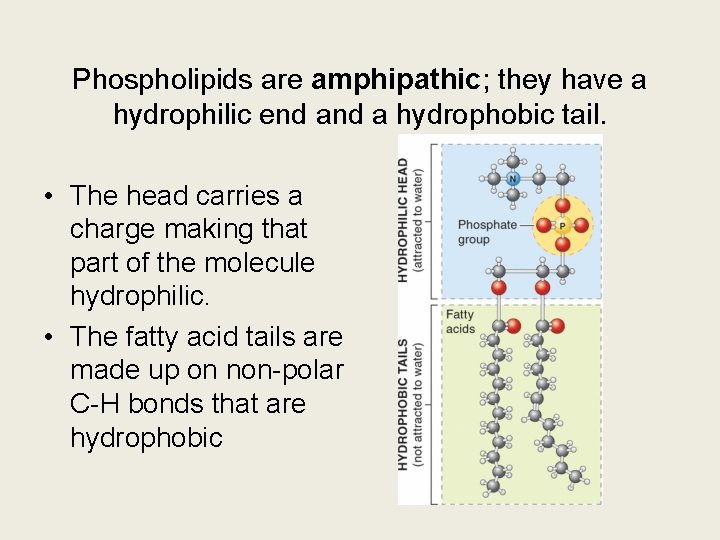
Phospholipids are amphipathic; they have a hydrophilic end a hydrophobic tail. • The head carries a charge making that part of the molecule hydrophilic. • The fatty acid tails are made up on non-polar C-H bonds that are hydrophobic
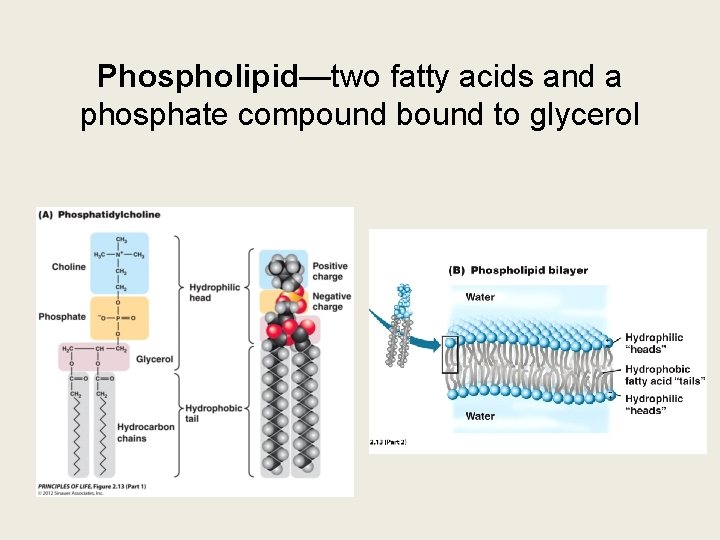
Phospholipid—two fatty acids and a phosphate compound bound to glycerol
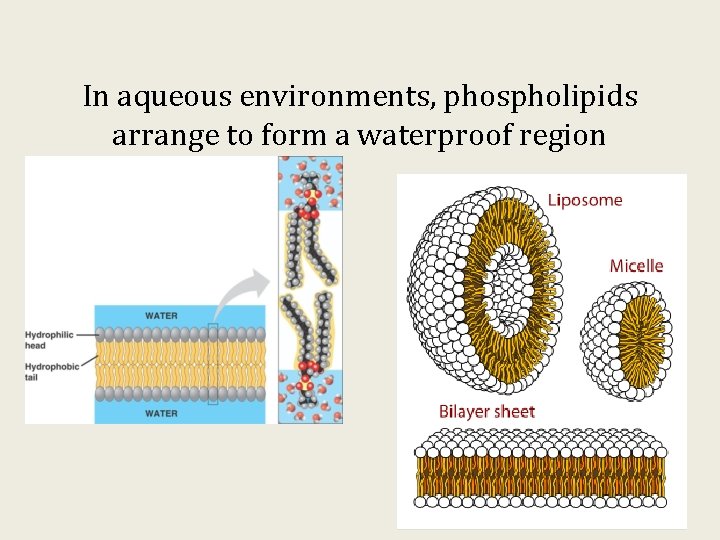
In aqueous environments, phospholipids arrange to form a waterproof region

Which of the following statements about phospholipids is false? a. The phosphate functional group and glycerol form the hydrophobic head of a phospholipid. b. A phospholipid has two fatty acids whereas a triglyceride has three fatty acids. c. Phospholipids are amphipathic (i. e. , they have two opposing chemical properties). d. The phosphate functional group and glycerol form the hydrophilic head of a phospholipid. e. Biological membranes are characterized by a phospholipid bilayer structure.
- Slides: 51