Unit 16 AcidsBasesSalts General properties Taste sour Turn

Unit 16 - Acids/Bases/Salts

General properties • Taste sour • Turn litmus blue to red • React with active metals • React with bases • Taste bitter • Turn litmus red to blue • Feel soapy or slippery (react with fats to make soap) • React with acids
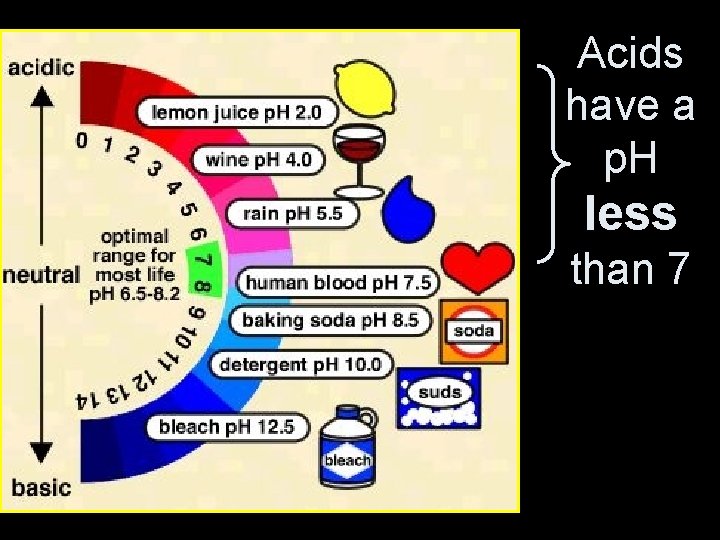
Acids have a p. H less than 7
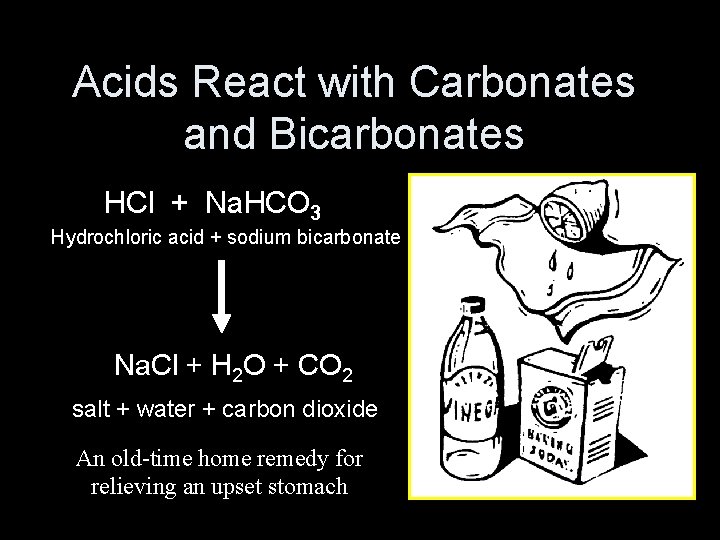
Acids React with Carbonates and Bicarbonates HCl + Na. HCO 3 Hydrochloric acid + sodium bicarbonate Na. Cl + H 2 O + CO 2 salt + water + carbon dioxide An old-time home remedy for relieving an upset stomach

Effects of Acid Rain on Marble (marble is calcium carbonate) George Washington: BEFORE acid rain George Washington: AFTER acid rain

Bases Neutralize Acids Milk of Magnesia contains magnesium hydroxide, Mg(OH)2, which neutralizes stomach acid, HCl. 2 HCl + Mg(OH)2 Mg. Cl 2 + 2 H 2 O Magnesium salts can cause diarrhea (thus they are used as a laxative) and may also cause kidney stones.

Naming • Acids are composed of hydrogen (H+) followed by an anion (negative ion). • If the acid formula contains oxygen in the anion, such as in H 2 SO 4, it is known as an oxyacid.

3 Rules To Naming Acids • If H + anion ending in –ide: Acid name is “hydro_____ic acid” – Take the root from the anion name and fill in the blank. • H + anion ending in –ate: Acid name is “_____ic acid” – Take the root from the anion name and fill in the blank. – “What I ATE was ICky” • H + anion ending in –ite: Acid name is “_____ous acid” – Take the root from the anion name and fill in the blank. – “Don’t b. ITE; it’s infecti. OUS”

Exceptions • Sulfate (SO 4 2 -) – Root is not sulf, but sulfur • Sulfuric acid • Phosphate (PO 4 3 -) – Root is not phosph, but phosphor • Phosphoric acid

Svante Arrhenius • He was a Swedish chemist (18591927), and a Nobel prize winner in chemistry (1903) • one of the first chemists to explain the chemical theory of the behavior of acids and bases

Arrhenius Definition - 1887 • Acids produce hydrogen ions (H 1+) in aqueous solution (HCl → H 1+ + Cl 1 -) • Bases produce hydroxide ions (OH 1 -) when dissolved in water. (Na. OH → Na 1+ + OH 1 -) • Limited to aqueous solutions. • Only one kind of base (hydroxides) • NH 3 (ammonia) could not be an Arrhenius base: no OH 1 - produced.

Brønsted-Lowry - 1923 • A broader definition than Arrhenius • Acid is hydrogen-ion donor (H+ or proton); base is hydrogen-ion acceptor. • Acids and bases always come in pairs. • HCl is an acid. – When it dissolves in water, it gives it’s proton to water. HCl(g) + H 2 O(l) ↔ H 3 O+(aq) + Cl-(aq) • Water is a base; makes hydronium ion.

Acids and bases come in pairs • A “conjugate base” is the remainder of the original acid, after it donates it’s hydrogen ion • A “conjugate acid” is the particle formed when the original base gains a hydrogen ion • Thus, a conjugate acid-base pair is related by the loss or gain of a single hydrogen ion.

Definitions Arrehenius only in water • Acids – produce H+ • Bases - produce OH- • Acids – donate H+ Bronsted-Lowry + • Bases – accept H any solvent
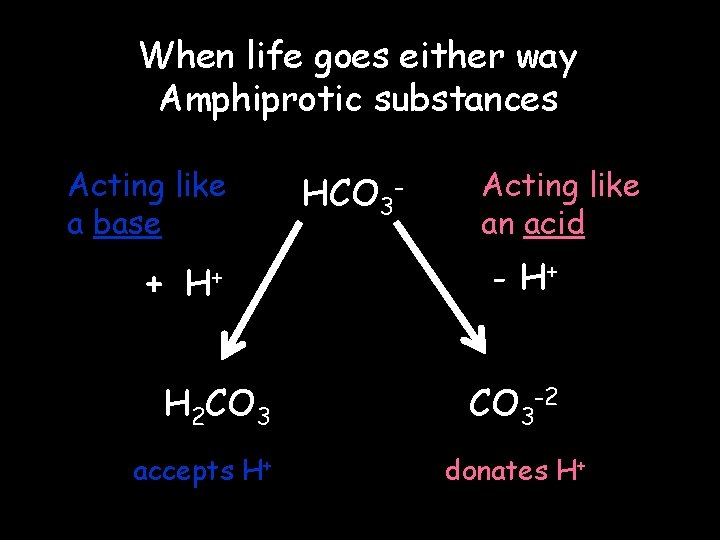
When life goes either way Amphiprotic substances Acting like a base + H+ H 2 CO 3 accepts H+ HCO 3 - Acting like an acid - H+ CO 3 -2 donates H+
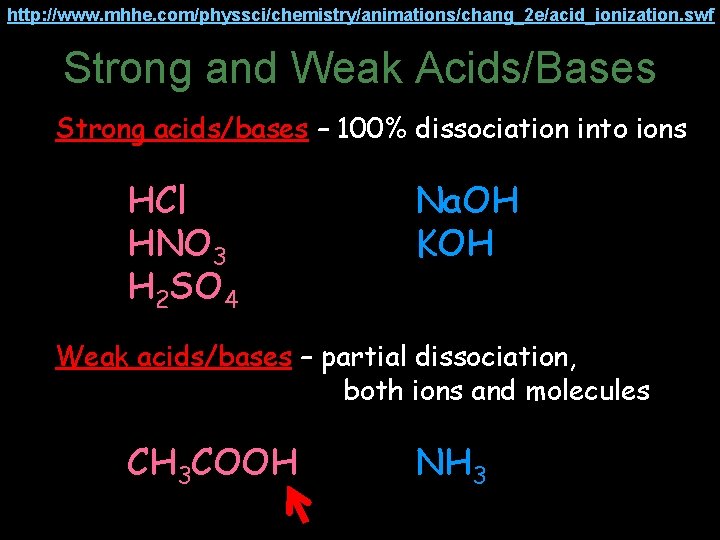
http: //www. mhhe. com/physsci/chemistry/animations/chang_2 e/acid_ionization. swf Strong and Weak Acids/Bases Strong acids/bases – 100% dissociation into ions HCl HNO 3 H 2 SO 4 Na. OH KOH Weak acids/bases – partial dissociation, both ions and molecules CH 3 COOH NH 3

Strong Acids • • • Prechloric Acid HCl. O 4 Sulfuric Acid H 2 SO 4 Hydriodic Acid HI Hydrobromic Acid HBr Hydrochloric Acid HCl Nitric Acid HNO 3

Strong Bases • Lithium hydroxide, Li. OH • Barium hydroxide, • Calcium hydroxide, Ba(OH)2 Ca(OH)2 • Magnesium • Sodium hydroxide, Na. OH hydroxide, • Strontium hydroxide, Mg(OH)2 Sr(OH)2 • Potassium hydroxide, KOH

Salt Hydrolysis • A salt is an ionic compound that: – comes from the anion of an acid – comes from the cation of a base – is formed from a neutralization reaction – some neutral; others acidic or basic • “Salt hydrolysis” - a salt that reacts with water to produce an acid or base

Salt Hydrolysis • Hydrolyzing salts usually come from: 1. a strong acid + a weak base, or 2. a weak acid + a strong base • Strong refers to the degree of ionization A strong Acid + a strong Base = Neutral Salt • How do you know if it’s strong? – Refer the list on your notes

Salt Hydrolysis • To see if the resulting salt is acidic or basic, check the “parent” acid and base that formed it. Practice on these: HCl + Na. OH Na. Cl, a neutral salt H 2 SO 4 + NH 4 OH (NH ) SO , acidic salt CH 3 COOH + KOH CH COOK, basic salt 4 2 3 4

Ionization constant of Water • Water ionizes, or falls apart into ions: H 2 O ↔ H 1+ + OH 1 • Called the “self ionization” of water • Occurs to a very small extent: [H 1+ ] = [OH 1 -] = 1 x 10 -7 M • Since they are equal, a neutral solution results from water Kw = [H 1+ ] x [OH 1 -] = 1 x 10 -14 M 2 • Kw is called the “ion product constant” for water
![Ion Product Constant • Kw is constant in every aqueous solution: [H+] x [OH-] Ion Product Constant • Kw is constant in every aqueous solution: [H+] x [OH-]](http://slidetodoc.com/presentation_image_h2/4438539db16ffa6338e3573482956973/image-23.jpg)
Ion Product Constant • Kw is constant in every aqueous solution: [H+] x [OH-] = 1 x 10 -14 M 2 • If [H+] > 10 -7 then [OH-] < 10 -7 • If [H+] < 10 -7 then [OH-] > 10 -7 • If we know one, other can be determined • If [H+] > 10 -7 , it is acidic and [OH-] < 10 -7 • If [H+] < 10 -7 , it is basic and [OH-] > 10 -7 – Basic solutions also called “alkaline”
![• If [H 3 O+] > [OH–‑] the solution is acidic. • If • If [H 3 O+] > [OH–‑] the solution is acidic. • If](http://slidetodoc.com/presentation_image_h2/4438539db16ffa6338e3573482956973/image-24.jpg)
• If [H 3 O+] > [OH–‑] the solution is acidic. • If [H 3 O+] < [OH–‑] the solution is basic. • If [H 3 O+] = [OH–‑] the solution is neutral.

Acid/Base/Salts Part 2

p. H • Expressing hydrogen ion concentration in numbers can be cumbersome. • A widely used system for expressing [H 3 O+] is the p. H scale. Whether or not a solution is acidic, basic, or neutral depends on the balance of H+ and OH- ions: • Neutral: [H+] = [OH-] • Acid: [H+] > [OH-] • Base: [H+] < [OH-]

p. H • p. H is the negative base 10 logarithm of the hydronium ion concentration: p. H = - log [H 3 O+]


Measuring p. H • Why measure p. H? 4 Everyday solutions we use - everything from swimming pools, soil conditions for plants, medical diagnosis, soaps and shampoos, etc. • Sometimes we can use indicators, other times we might need a p. H meter
![p. H • Remember, for pure water, [H 3 O+] is 1 x 10 p. H • Remember, for pure water, [H 3 O+] is 1 x 10](http://slidetodoc.com/presentation_image_h2/4438539db16ffa6338e3573482956973/image-30.jpg)
p. H • Remember, for pure water, [H 3 O+] is 1 x 10 -7 M. So what’s the p. H of pure water? • Ex: What is the p. H of a solution with a hydronium ion concentration of 1. 0 x 10 -10 M?
![Note that as p. H increases, [H 3 O+] decreases and [OH–‑] increases. Note Note that as p. H increases, [H 3 O+] decreases and [OH–‑] increases. Note](http://slidetodoc.com/presentation_image_h2/4438539db16ffa6338e3573482956973/image-31.jpg)
Note that as p. H increases, [H 3 O+] decreases and [OH–‑] increases. Note the relationship between [H 3 O+] and [OH–‑]. Remember, the product of these must always equal 1 x 10 -14 for aqueous solutions. • [H+] x [OH-] = 1 x 10 -14 M 2 Note that as p. H increases, [H 3 O+] decreases and [OH–‑] increases. Note the relationship between [H 3 O+] and [OH–‑]. Remember, the product of these must always equal 1 x 10 14 for aqueous solutions.
![p. OH = - log [OH-] p. H + p. OH = 14 p. OH = - log [OH-] p. H + p. OH = 14](http://slidetodoc.com/presentation_image_h2/4438539db16ffa6338e3573482956973/image-32.jpg)
p. OH = - log [OH-] p. H + p. OH = 14

Example • Find the p. H, the p. OH = 5. 3 Øp. H + p. OH = 14 p. H = 14 - 5. 3 p. H = 8. 7
![p. H calculations • Use the reverse of the equation to calculate the [H+] p. H calculations • Use the reverse of the equation to calculate the [H+]](http://slidetodoc.com/presentation_image_h2/4438539db16ffa6338e3573482956973/image-34.jpg)
p. H calculations • Use the reverse of the equation to calculate the [H+] when p. H is known. Ø[H 3 O+] =10(-p. H) *** 2 nd log on the calulator! = 10 -p. H
![p. H calculations • Use identical process for [OH-] when p. OH is known. p. H calculations • Use identical process for [OH-] when p. OH is known.](http://slidetodoc.com/presentation_image_h2/4438539db16ffa6338e3573482956973/image-35.jpg)
p. H calculations • Use identical process for [OH-] when p. OH is known. Ø[OH-] = 10(-p. OH)

Acid/Bases/Salts Neutralization/Titrations

A. Neutralization • Chemical reaction between an acid and a base. • Products are a salt (ionic compound) and water. • Note in the reaction above, the acid: base mole ratio is 1: 1. • However, in the reaction between H 2 SO 4 and Na. OH: • it takes 2 moles of base to neutralize 1 mole of acid. • The reacting ratios of acid and base will be important in solving problems related to neutralization reactions.

A. Neutralization ACID + BASE SALT + WATER HCl + Na. OH Na. Cl + H 2 O strong neutral HC 2 H 3 O 2 + Na. OH Na. C 2 H 3 O 2 + H 2 O weak strong basic – Salts can be neutral, acidic, or basic. – Neutralization does not mean p. H = 7.

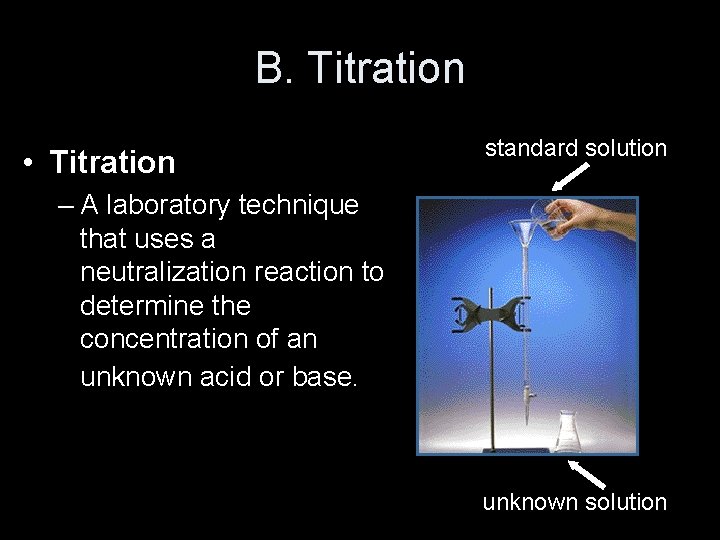
B. Titration • Titration standard solution – A laboratory technique that uses a neutralization reaction to determine the concentration of an unknown acid or base. unknown solution

• An indicator is used to show when neutralization has occurred. • changes color in response to changes in p. H. • actually weak acid or base that change color in response to p. H change. • In acidic solutions, indicators act as Bronsted-Lowry base. As the indicator molecules accept H+, they change color. • In basic solutions, indicators act as Bronsted-Lowry acid. As the indicator molecules donate H+, they change color.

B. Titration • End Point – – point at which an indicator changes color during a titration • Equivalence point – Point at which equal amounts of H 3 O+ and OH- have been added. – when mole ratio exactly equals mole ratio required by reaction – Determined by… • indicator color change • dramatic change in p. H

B. Titration + O moles H 3 = moles M V n = M V n M: Molarity V: volume n: # of H+ ions in the acid or OH- ions in the base OH

B. Titration • 42. 5 m. L of 1. 3 M KOH are required to neutralize 50. 0 m. L of H 2 SO 4. Find the molarity of H 2 SO 4. H 3 O + OH- M=? M = 1. 3 M V = 50. 0 m. L V = 42. 5 m. L n=2 n=1 MV# = MV# M(50. 0 m. L)(2)=(1. 3 M)(42. 5 m. L)(1) M = 0. 55 M H 2 SO 4
- Slides: 44