Unit 15 Electrochemistry Chapter 20 Problem Set p

Unit 15 Electrochemistry Chapter 20 Problem Set: p. 890 -898 3, 11, 13, 17, 23, 27, 31, 35, 43, 45, 47, 53, 61, 63, 69, 72, 75, 79, 85, 87, 95

Oxidation Numbers Please assign oxidation numbers to the elements in each substance. 1) HNO 3 2) HNO 2 3) Pb. SO 4 4) (NH 4)2 Ce(SO 4)3 5) Al(C 2 H 3 O 2)3

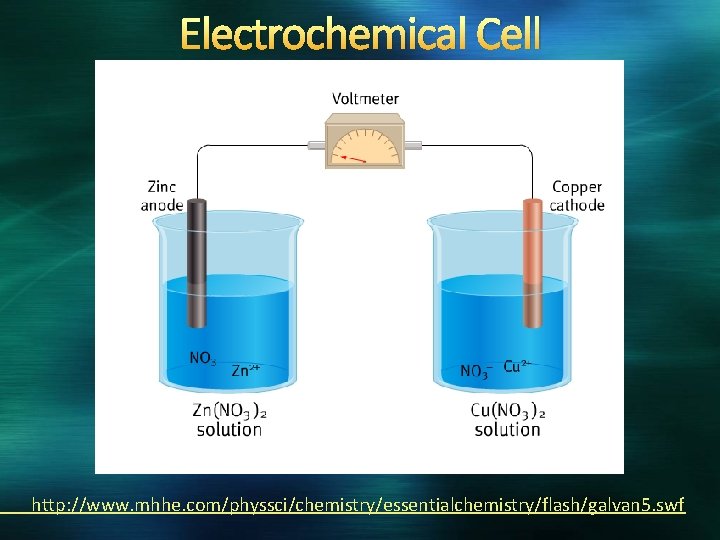
Electrochemistry: Relationship between electricity and redox reactions Electricity = movement of electrons Voltaic Cells The energy released in a spontaneous redox reaction that is used to perform electrical work is harnessed in Voltaic Cells Also called Galvanic Cells Electrons transfer through an external pathway rather than directly between reactants Electrodes: metals used in the circuit
Electrochemical Cell http: //www. mhhe. com/physsci/chemistry/essentialchemistry/flash/galvan 5. swf

Using a Porous Disk as a Salt Bridge

Components of Electrochemical Cells Anode: electrode at which oxidation occurs Cathode: electrode at which reduction occurs Salt Bridge: a connection between two half-cells that allows for the flow of ions Composed of a strong electrolyte Must be a solution of a compound that will not produce a precipitate with any other ion in solution Salt anions migrate toward the anode; cations migrate toward the cathode **Electrons flow from the anode to the cathode**

Pneumonic Devices Keep your vowels together and your consonants together Anode = oxidation Cathode = reduction An ox and Red cats Anode is oxidation, Cathode is reduction Electrons flow from A to C (in alphabetical order)

Standard Cell Potentials Used to determine which metal will be the cathode/anode for the reaction to be spontaneous Cell EMF: Electromotive Force Caused by a difference in potential energy between the different electrodes. Allows electrons to be pushed Denoted Ecell , measured in volts, V Also called cell potential

Cell Potential Cell potential is positive for spontaneous reactions negative for nonspontaneous reactions Depends on 1. Reactions Occurring (Metals Used) 2. Concentrations of Solutions (Molarity) 3. Temperature (normally 25 o. C) Eocell potential at standard conditions

EMF can then be used to determine what will happen in a chemical equation The more positive the Eored value for a half reaction, the greater the tendency for the reactant of the half reaction to be reduced and, therefore, to oxidize another species The half reaction with the smallest reduction potential is most easily reversed as an oxidation The Eored table acts as an activity series for which substances act as oxidizers and reducers

Calculating Cell Potential o E cell based on standard reduction potentials Potential associated with each electrode is the ability for reduction to occur at the electrode. Eored = standard reduction potential for metal (based on Standard Hydrogen Electrode, SHE – use Appendix E p. 1128!) Eocell = Eored (cathode) – Eored (anode) If the stoichiometry of the reaction changes DO NOT multiply the value by the cell potential for that metal The more positive the Eored for a metal, the greater the ability for reduction Thus the metal with a larger Eored will be the better

Example: A voltaic cell is based on the following half cells: 2+ Cd + 2 e � Cd 2+ Sn + 2 e � Sn Determine voltage produced in spontaneous reaction.

Example: A voltaic cell is based on the following half cells: Cd 2+ + 2 e- � Cd Sn 2+ + 2 e- � Sn Determine voltage produced in spontaneous reaction. Eored (Cd 2+ /Cd ) = -0. 403 V Eored (Sn 2+ /Sn ) = -0. 136 V Cathode: Sn 2+ + 2 e- �Sn Anode: Cd �Cd 2+ + 2 e. Eocell = Eocathode – Eoanode = -0. 136 V – (-0. 403 V) = 0. 267 V

Example: What would be the electrical potential for the reaction: 2+ + Pb. O 2 + Na � Pb + Na

Example: What would be the electrical potential for the reaction: 2+ + Pb. O 2 + Na � Pb + Na Na �Na+ + 1 e. Pb. O 2 �Pb 2+ + 2. 71 V + 1. 50 V (Na �Na+ + 1 e-)x 2 *DO NOT MULTIPLY Eored by 2* 4 H+ + 2 e- + Pb. O 2 �Pb 2+ + 2 H 2 O 4 H+ + Pb. O 2 + 2 Na � 2 Na+ + Pb 2+ + 2 H 2 O Overall potential for the battery = 2. 71 + 1. 50 = 4. 3 V

Spontaneous Redox Reactions Reduction Potentials can be used to calculate energies of reactions when using redox processes Remember: E=+ Spontaneous E=Nonspontaneous E can be converted into G for Gibbs Free Energy or othermochemical quantities ∆Go = -n. FEo n= number of electrons transferred F = Faraday’s constant = 96500 C/mol or J/Kmol Eo = Calculated from Eored values

Calculate ∆G for the following reaction: 2+ 2+ Cu (aq) + Fe(s) �Cu(s) + Fe (aq)

Calculate ∆G for the following reaction: Cu 2+(aq) + Fe(s) �Cu(s) + Fe 2+(aq) Start with Half-Reactions Fe �Fe 2+ + 2 e. Cu �Cu 2+ + 2 e- Flip the copper reaction so it matches the overall equation Fe �Fe 2+ + 2 e 0. 44 V Cu 2+ + 2 e- �Cu 0. 34 V Cu 2+(aq) + Fe(s) �Cu(s) + Fe 2+(aq) 0. 78 V Solve for ∆Go = -n. FEo ∆Go = -(2)(96500)(0. 78) = -150517 J/mol = -151 k. J/mol What is the Keq for this reaction? ∆Go = -RTln. Keq

Warm-Up : Do another page on the net ionic equations! The whole packet is due TODAY! The 2008 FRQ was your Homework. Please be ready to grade it and the crossword page.

Effect of Concentration on Cell EMF As voltage is produced, concentrations vary. Reactants are consumed and products are produced. When E = 0, the cell is dead, the concentrations cease to change due to equilibrium being reached. For different concentrations, the Nernst equation is used:

To calculate voltage, use the Nernst Equation Will be zero Cell EMF and Equilibrium ∆G = 0, ∆E = 0 at Equilibrium Using Nernst Solving for the equilibrium constant

Example: What is the electrical potential for the cell with the following concentrations? VO 21+ + Zn � Zn 2+ + VO 2+ [VO 21+] = 2. 0 M [H+] = 0. 50 M [VO 2+] = 1. 0 x 10 M [Zn 2+] = 0. 10 M

Example: What is the electrical potential for the cell with the following concentrations? VO 21+ + Zn � Zn 2+ + VO 2+ [VO 21+] = 2. 0 M [H+] = 0. 50 M [VO 2+] = 1. 0 x 10 -2 M [Zn 2+] = 0. 10 M Start with the half reactions 2(2 H+ + 1 e- + VO 21+ �VO 2+ + H 2 O ) Zn �Zn 2+ +2 e 0. 76 V 4 H+ + 2 VO 21+ + Zn �Zn 2+ + 2 VO 2+ + 2 H 2 O Write the equilibrium expression 1. 00 V 1. 76 V 4 x 10 -5

Solve for E =1. 76 – (-0. 13) E = 1. 89 V The voltage went up! Use Le. Chatelier’s Principle to explain why

Concentration Cells: an electrochemical cell where the same electrodes (same half-reactions) are used, but the solutions have different concentrations What will the voltmeter read for the following electrochemical cell? Anode Reaction – Oxidation dilute solution becomes more concentrated. Ag(s) → Ag+ (0. 1 M) + e- +0. 8 V Cathode Reaction – Reductionconcentrated solution becomes more dilute. Ag+ (1. 0 M) + e- → Ag (s) -0. 8 V Net Cell Reaction Ag+ (1. 0 M) → Ag+ (0. 1 M) 0. 0 V V

Use the Nernst Equation to solve for the potential You DON’T need your calculator for this one!!! E = 0. 0592 V

Batteries Self-contained electrochemical power source consisting of 1 or more galvanic cells. Voltages are additive when batteries are connected due to a continuation of the flow of electrons Fuel Cells A voltaic cell that converts chemical energy into electrical energy Not self-contained Produces current by combustion and electronexcitation

Corrosion A spontaneous redox reaction in which a metal is attacked by some substance in the environment and converted to an unwanted compound Is often prevented, or reversed, by use of a sacrificial anode (a more active metal) Electrolysis The use of electricity to cause nonspontaneous reactions to occur by driving the reaction in the opposite direction (basis of rechargeable batteries Take place in electrolytic cells Cathode is connected to – terminal to accept e-, anode is attached to + terminal to donate e(opposite of voltage)
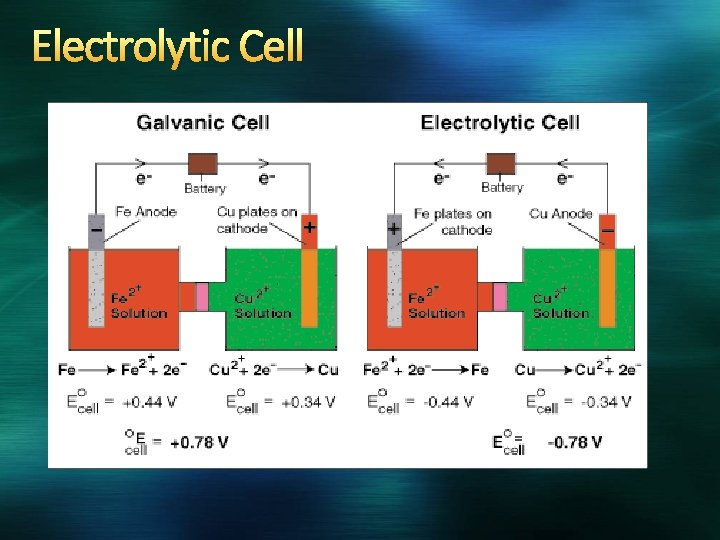
Electrolytic Cell

Calculations with Electrolytic Cells Ampere = units to measure current in Coulombs/second Measures the rate of electron flow Faraday’s Constant, F = 96485 Coulomb/mol Example: Calculate the amount of time required to produce 1000 g of magnesium metal by electrolysis of molten Mg. Cl 2 using a current of 50 A.

Calculations with Electrolytic Cells Ampere = units to measure current in Coulombs/second Measures the rate of electron flow Faraday’s Constant, F = 96485 Coulomb/mol Example: Calculate the amount of time required to produce 1000 g of magnesium metal by electrolysis of molten Mg. Cl 2 using a current of 50 A. Use a t-table! Time = 158823 sec = 2647 min = 44 hours = 1. 84 days

Example: a Cr 3+ (aq) solution is electrolyzed using a current of 7. 60 A. What mass of Cr (s) is plated out after 2. 00 days?
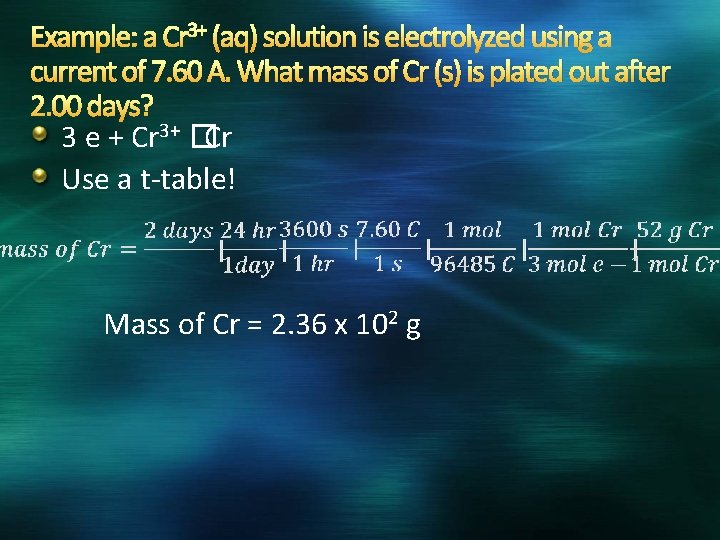
Example: a Cr 3+ (aq) solution is electrolyzed using a current of 7. 60 A. What mass of Cr (s) is plated out after 2. 00 days? 3 e + Cr 3+ �Cr Use a t-table! Mass of Cr = 2. 36 x 102 g

Electrolysis can be used to electroplate metals Thin coating is plated onto an existing metal to help protect it Metal to be plated is attached to the cathode. Metal coating is produced by the metal attached to anode and a “like” solution
- Slides: 34