UNIT 14 Acids and Bases and p H

UNIT 14: Acids and Bases, and p. H acidic solution basic solution Arrhenius model Brønsted - Lowry model conjugate acid conjugate base conjugate acid-base pair amphoteric Lewis model strong acid weak acid ionization constant strong base weak base ionization constant acid-base indicator end point salt hydrolysis buffer capacity

Acids and Bases: An Introduction All aqueous solutions contain hydrogen ions (H+) and hydroxide ions (OH-). An acidic solution contains: + more (H ) ions than (OH ) ions, whereas a basic solution contains: more (OH-) ions than (H+) ions A neutral solution contains: equal + concentrations of (H ) ions and (OH ) ions

Aqueous Solutions = acid + base hydrogen ions (H+) + hydroxide ions (OH-) hydronium ion (H 3 O+) is a hydrated hydrogen ion. The acidic symbols (H+) and (H 3 O+) can be used interchangeably in chemical equations.

Pure water self-ionizes slightly to form H 30+ and OHions, as shown in this equation: + H 20(l) → H 30 (aq) + OH (aq) The equation for the equilibrium can be simplified by removing one water molecule from each side. H 20(l) → H+(aq) + OH-(aq)

Submicroscopic Behavior of Acids • Hydronium ion formation: HCl + H 2 O H 3 O+ + Cl. HNO 3 + H 2 O H 3 O+ + NO 3 -

Acids react with bases – the reaction of acids and bases are central to the chemistry of living systems, the environment, and many important industrial processes
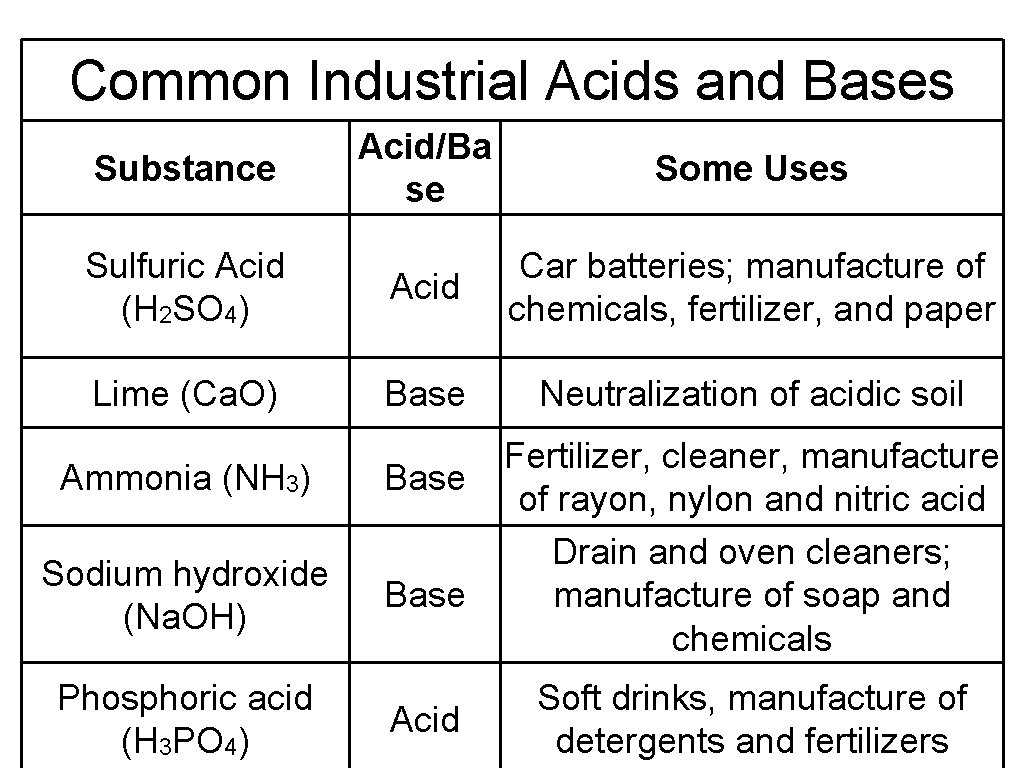
Common Industrial Acids and Bases Acid/Ba se Some Uses Sulfuric Acid (H 2 SO 4) Acid Car batteries; manufacture of chemicals, fertilizer, and paper Lime (Ca. O) Base Neutralization of acidic soil Substance Ammonia (NH 3) Base Sodium hydroxide (Na. OH) Base Phosphoric acid (H 3 PO 4) Acid Fertilizer, cleaner, manufacture of rayon, nylon and nitric acid Drain and oven cleaners; manufacture of soap and chemicals Soft drinks, manufacture of detergents and fertilizers

Macroscopic Properties of Acids and Bases Taste and feel: Acids: taste sour, conduct electricity Examples: (lemon juice, vinegar) Bases: taste bitter, feel slippery, conduct electricity. Bases are slippery (soap)

Chemical Properties Litmus test: Indicatorschange colors in the presence of an acid or a base Litmus Paper: base = turns blue acid = turns red

Monoprotic and Polyprotic Acids • Hydrogen atoms that are bonded to electronegative elements are ionizable –Monoprotic acid can donate only one hydrogen Example- HCl –Polyprotic acids can donate more than one hydrogen atom • diprotic acid has two ionizable hydrogens Example: sulfuric acid • triprotic acids has three ionizable hydrogens Example: Boric acid 10

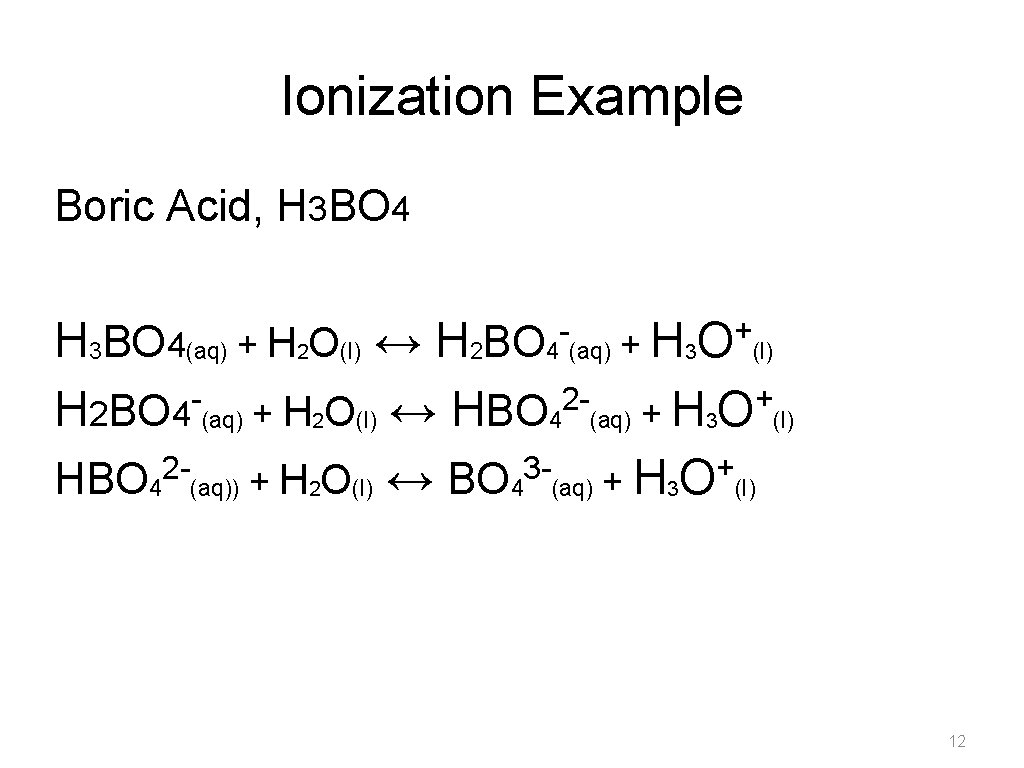
Ionization Example Boric Acid, H 3 BO 4 - + O H 3 BO 4(aq) + H 2 O(l) ↔ H 2 BO 4 (aq) + H 3 (l) 2+ H 2 BO 4 (aq) + H 2 O(l) ↔ HBO 4 (aq) + H 3 O (l) 23+ HBO 4 (aq)) + H 2 O(l) ↔ BO 4 (aq) + H 3 O (l) 12

MODELS • The Arrhenius Model • The Bronsted-Lowry Model 13

The Arrhenius Model • Acids – –produce hydrogen ions in aqueous solution HCl H+ + Cl- • Bases – –produce hydroxide ions in aqueous solution Na. OH Na+ + OH-

The Bronsted-Lowry Model Arrhenius concept is limited because it only allows for one kind of base. It expresses the substance. Acid – proton (H+) donor Base – proton (H+) acceptor

Acid – hydrogen ion donor Base – hydrogen ion acceptor Conjugate base – results when acid donates hydrogen ion Conjugate acid – results when a base accepts a hydrogen ion 16
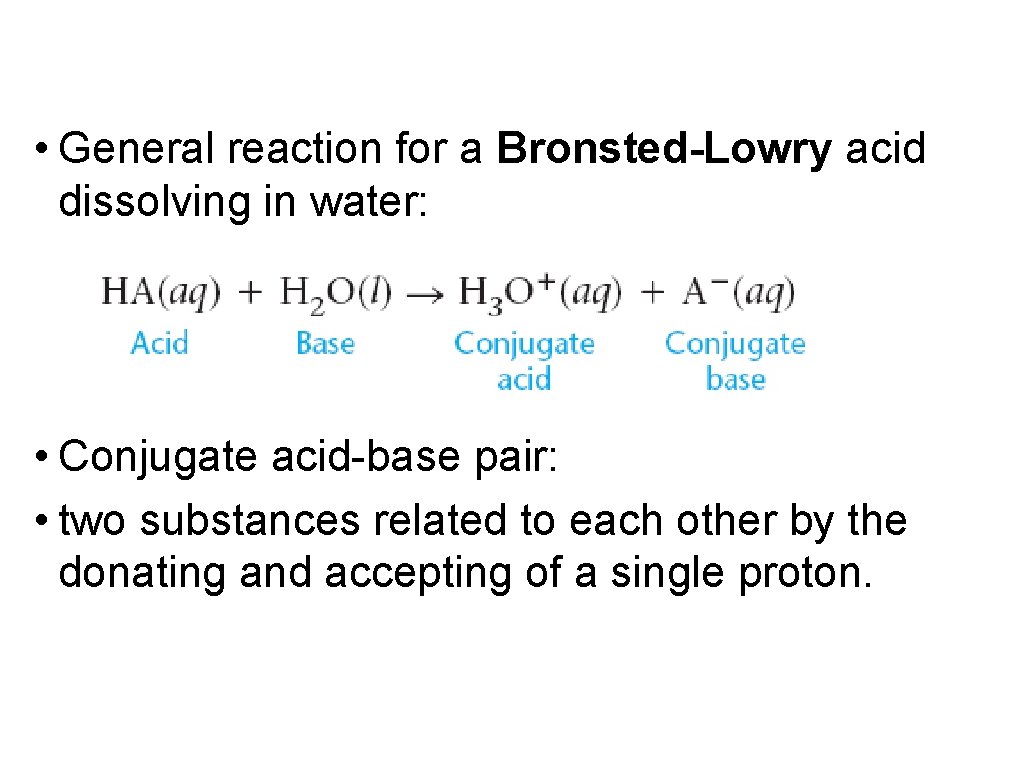
• General reaction for a Bronsted-Lowry acid dissolving in water: • Conjugate acid-base pair: • two substances related to each other by the donating and accepting of a single proton.
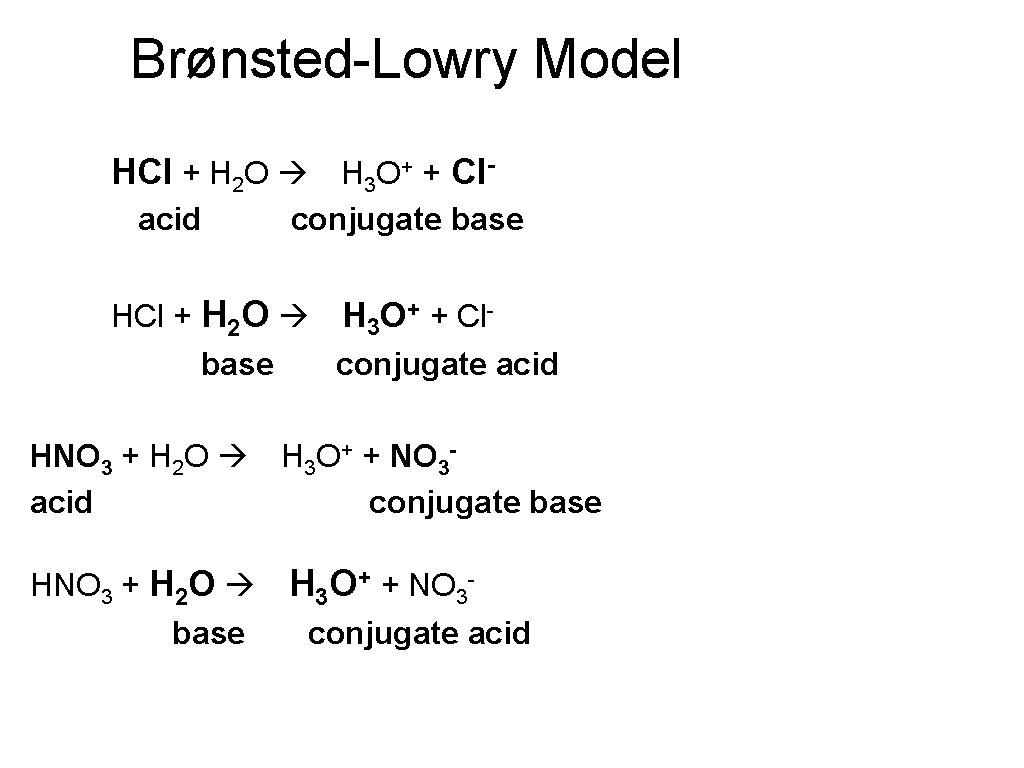
Brønsted-Lowry Model HCl + H 2 O H 3 O+ + Clacid conjugate base HCl + H 2 O H 3 O+ + Clbase conjugate acid HNO 3 + H 2 O H 3 O+ + NO 3 acid conjugate base HNO 3 + H 2 O H 3 O+ + NO 3 base conjugate acid
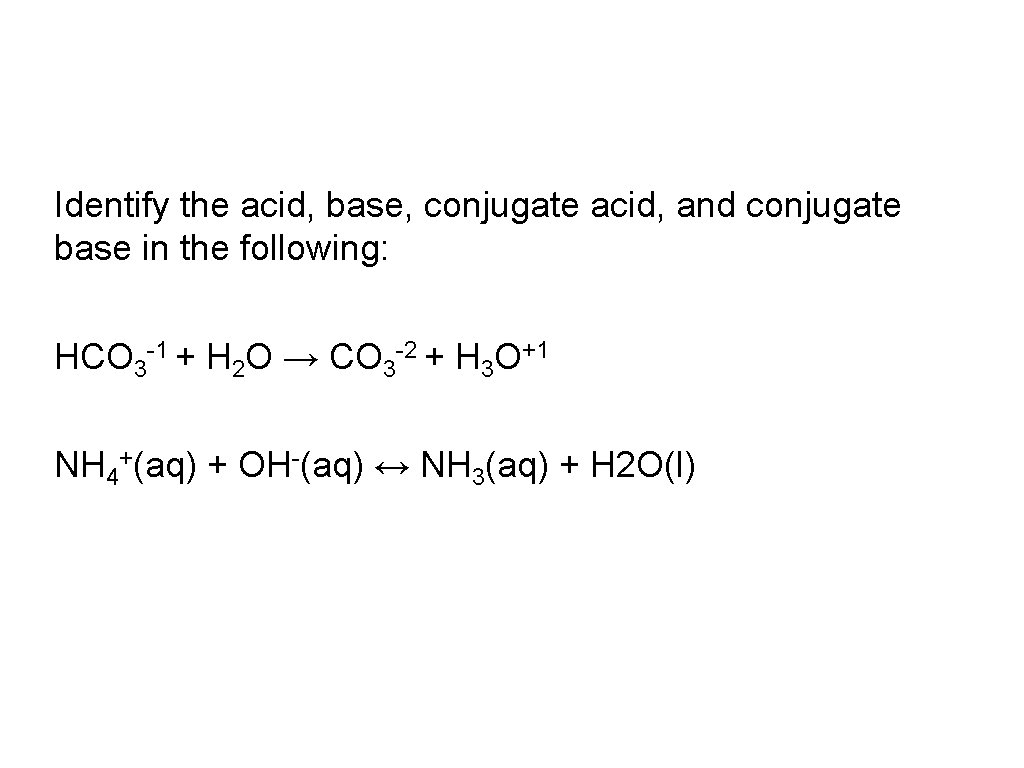
Identify the acid, base, conjugate acid, and conjugate base in the following: HCO 3 -1 + H 2 O → CO 3 -2 + H 3 O+1 NH 4+(aq) + OH-(aq) ↔ NH 3(aq) + H 2 O(l)

What is the conjugate base of the following? HCl 3 HCO H 2 SO 4 What is the conjugate acid of the following? Br. H 2 O 2 HPO 4 20

Water as an Acid and a Base • Amphoteric substance – can behave either as an acid or as a base • Ionization of water:

Strengths of Acids and Bases • Strong acids and bases ionize completely • Weak acids and bases ionize only partially
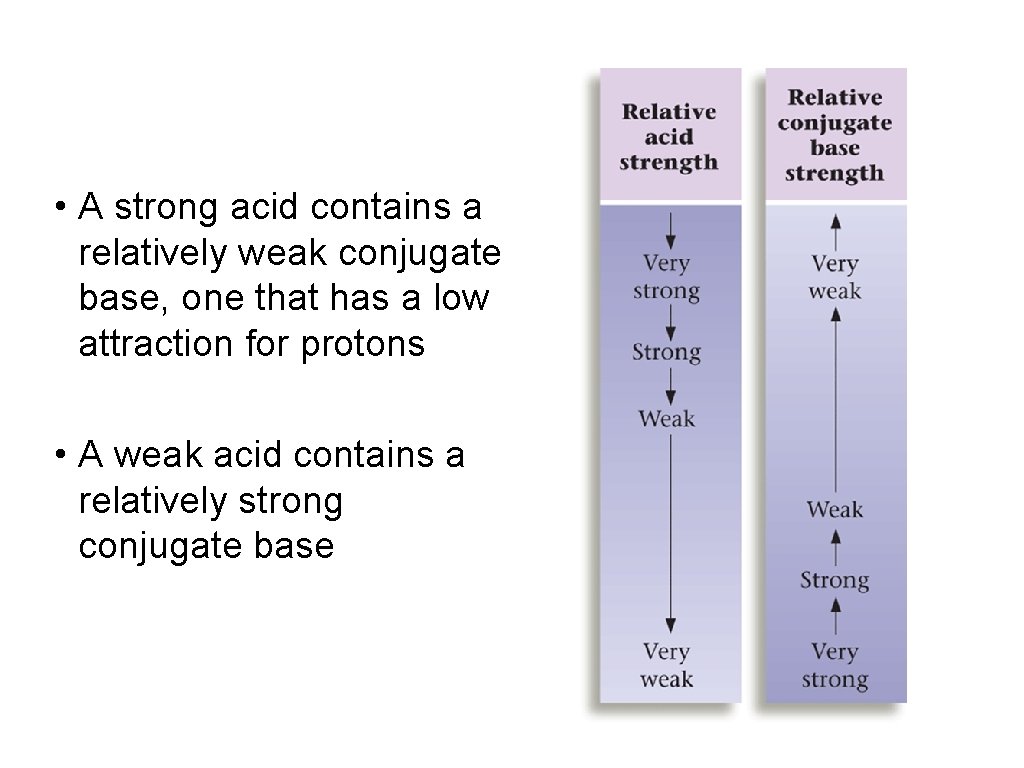
• A strong acid contains a relatively weak conjugate base, one that has a low attraction for protons • A weak acid contains a relatively strong conjugate base
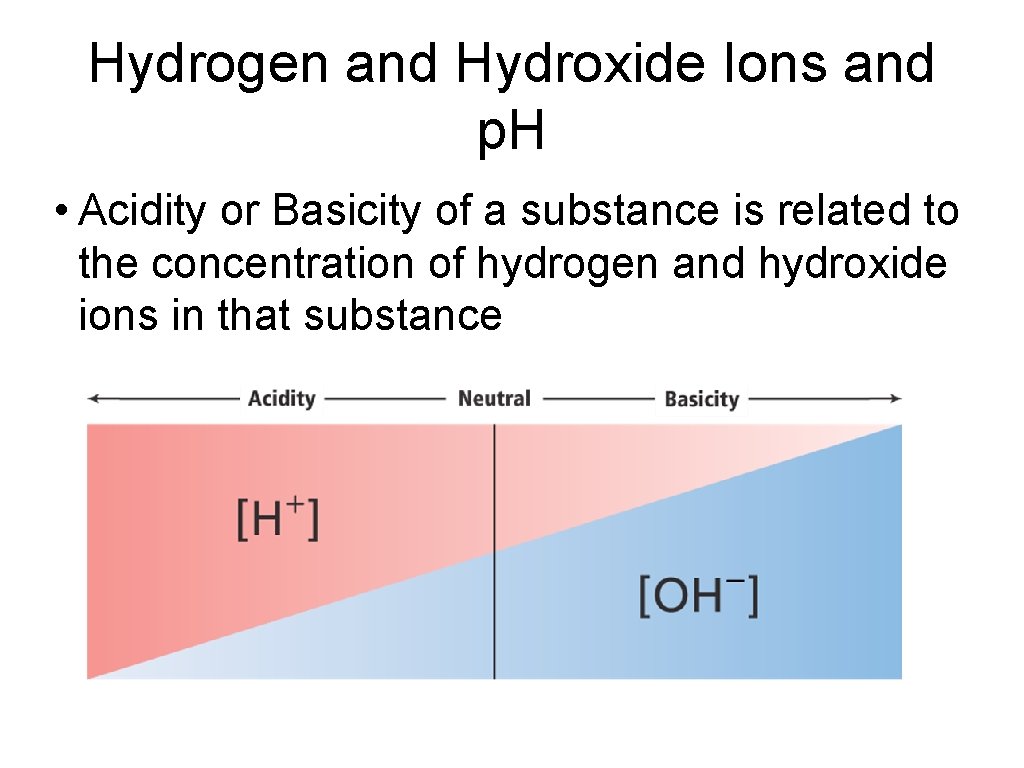
Hydrogen and Hydroxide Ions and p. H • Acidity or Basicity of a substance is related to the concentration of hydrogen and hydroxide ions in that substance
![• The product of constant + [H ] and [OH ] is always • The product of constant + [H ] and [OH ] is always](http://slidetodoc.com/presentation_image_h2/a8539b83e2fa27dc6a5a3d4a42ed4758/image-25.jpg)
• The product of constant + [H ] and [OH ] is always • Kw = ion product constant for water • No matter what the solution contains, Kw will -14 always equal 1. 0 x 10
![1. An acidic solution, where [H+] > [OH-] 2. A basic solution, where [OH 1. An acidic solution, where [H+] > [OH-] 2. A basic solution, where [OH](http://slidetodoc.com/presentation_image_h2/a8539b83e2fa27dc6a5a3d4a42ed4758/image-26.jpg)
1. An acidic solution, where [H+] > [OH-] 2. A basic solution, where [OH ] 3. A neutral solution, where + [H ] > + [H ] = [OH ] [H+] = [OH-] = 1. 0 x 10 -7 In each case, however, Kw = [H+] [OH-] = 1. 0 x 1014
![• Calculate the [H+] or [OH−] and state whether the solution is neutral, • Calculate the [H+] or [OH−] and state whether the solution is neutral,](http://slidetodoc.com/presentation_image_h2/a8539b83e2fa27dc6a5a3d4a42ed4758/image-27.jpg)
• Calculate the [H+] or [OH−] and state whether the solution is neutral, acidic, or basic – 1. 0 x 10 -5 M OH– 1. 0 x 10 -7 M OH– 10. 0 M H+

Determining the Acidity of a Solution The p. H Scale: 0 – 14 < 7 = acidic 7 = neutral > 7 = basic
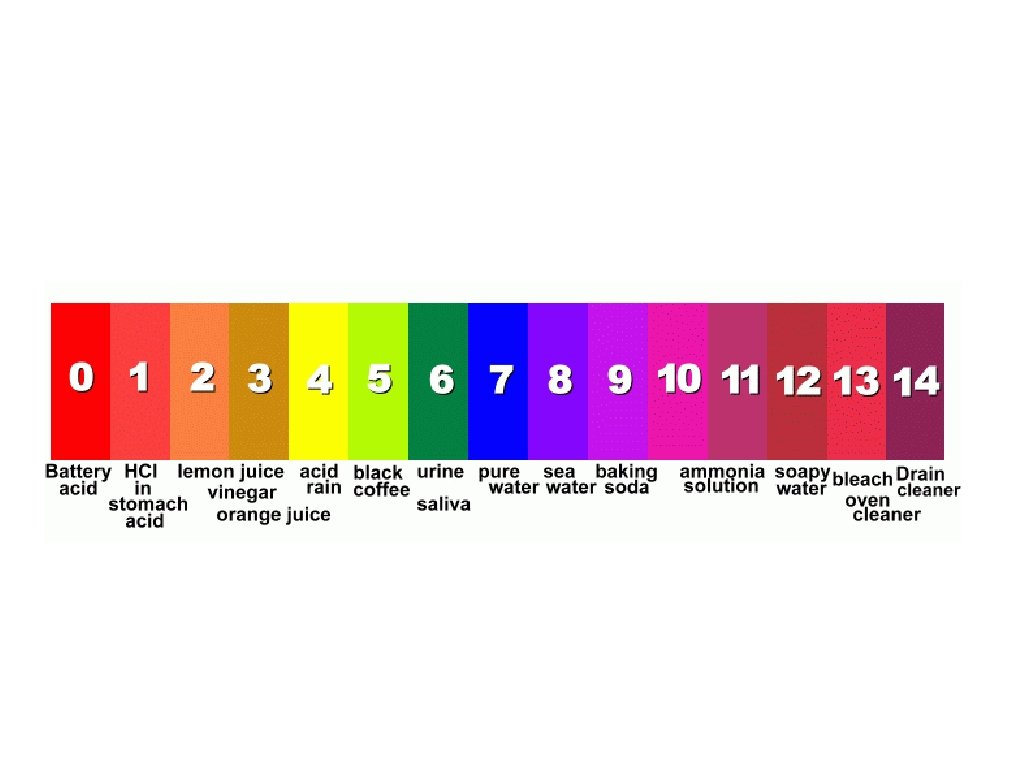

A mathematical scale in which the concentration of H+ ions in a solution is expressed as a number from 0 – 14 p. H = −log [H+]

• What is the p. H of solutions having the following ion concentrations? – [H+] = 1. 0 x 10 -2 M – [H+] = 3. 0 x 10 -6 M

Because the p. H scale is a log scale based on 10, the p. H changes by 1 for every + power of 10 change in the [H ]

• Log scales similar to the p. H scale are used for representing other quantities: − p. OH = −log [OH ]

• What is the p. OH of a solution having the following ion concentration? – [OH-] = 1. 0 x 10 -6 M – [OH-] = 6. 5 x 10 -4 M

p. H + p. OH = 14 What is the p. OH of a solution whose p. H is 5? What is the p. H of a solution whose M? [OH ]=4. 0 x -3 10
![Calculate the p. H and p. OH of the following solutions: [H+] = 0. Calculate the p. H and p. OH of the following solutions: [H+] = 0.](http://slidetodoc.com/presentation_image_h2/a8539b83e2fa27dc6a5a3d4a42ed4758/image-36.jpg)
Calculate the p. H and p. OH of the following solutions: [H+] = 0. 000033 M [OH-] = 0. 0095 M

• It is also possible to find the or from the p. H or p. OH by undoing the log operation + -p. H • [H ]= 10 -p. OH • [OH ] = 10 + [H ] [OH ]

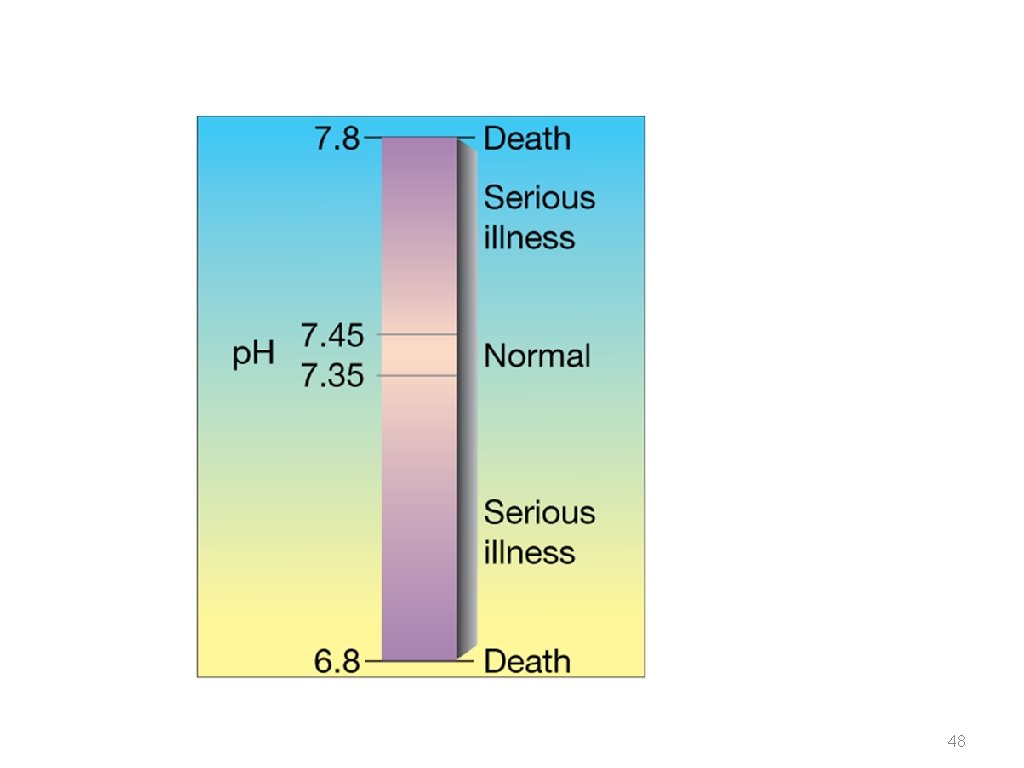
The p. H of a human blood sample was measured to be 7. 41. + What is the [H ] and [OH ]in this blood?

Neutralization Reactions • Acid + Base → salt + water • HCl + Na. OH→Na. Cl + H 2 O –Ionic: –Net ionic:

Titration • Method for determining the concentration of a solution by reacting a known volume of that solution with a solution of known concentration. –If unknown is acid, known must be base

Titration Procedure • Measured volume of acid or base of unknown concentration is placed in a flask and initial p. H is recorded • Buret is filled with the solution of known concentration (standard solution/titrant) • Standard solution added slowly until neutral p. H is reached (equivalence point) [H+] = [OH-]
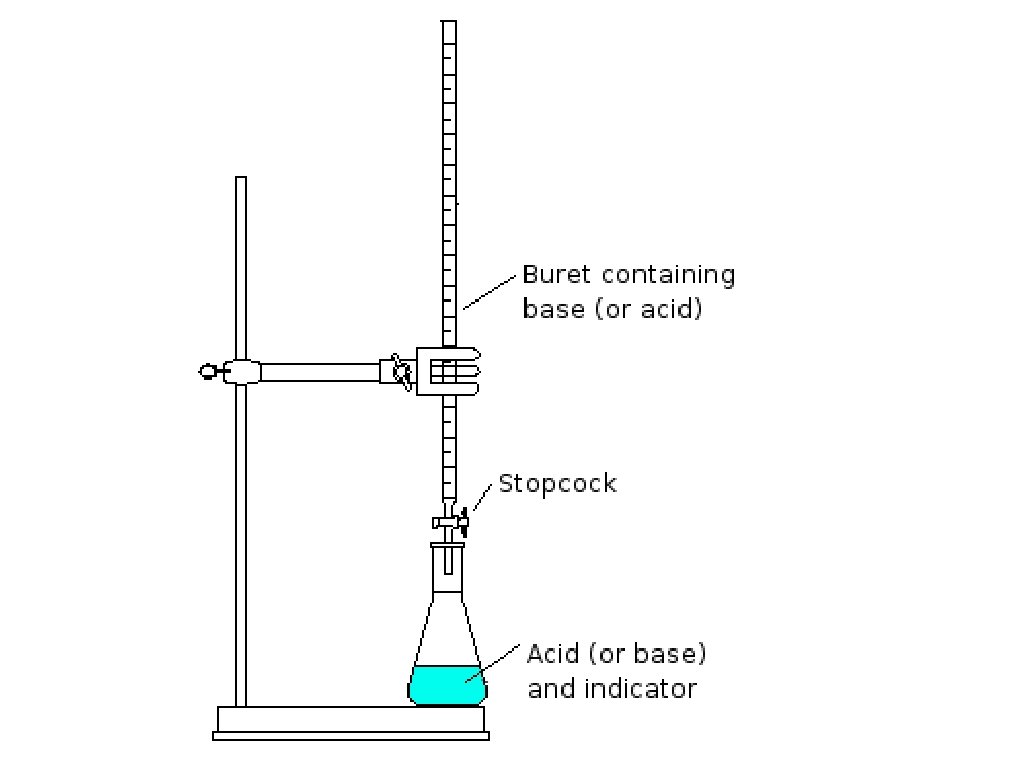

• End point of a titration can be measured using a p. H meter or an indicator –Indicator changes color at different p. H values
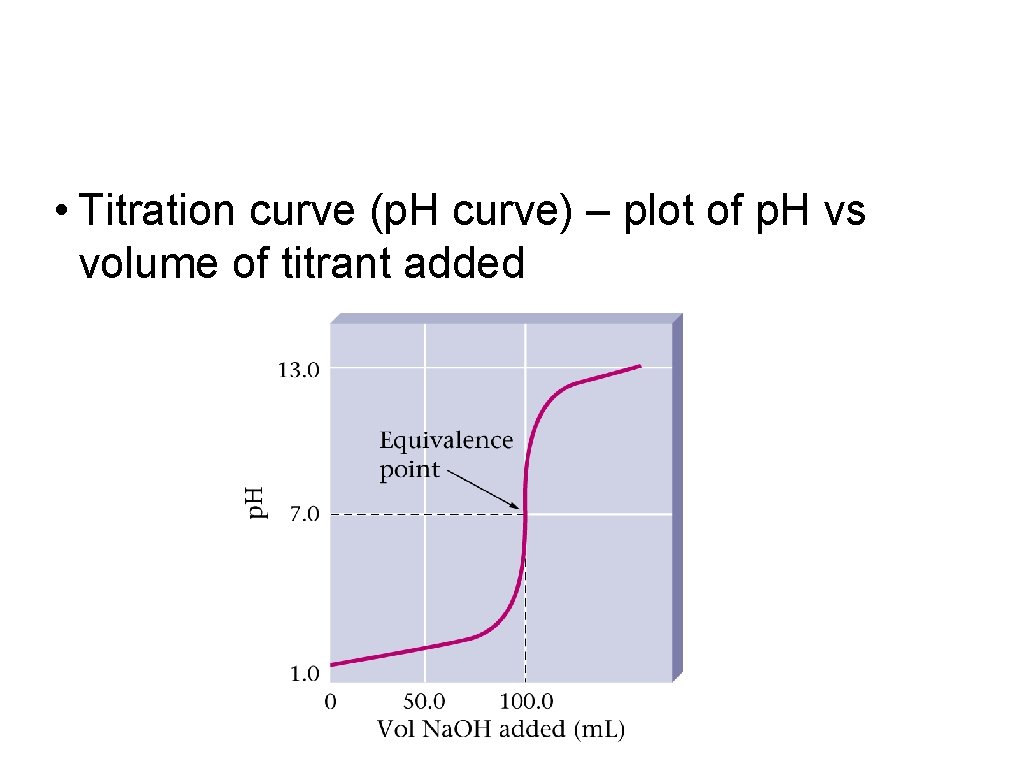
• Titration curve (p. H curve) – plot of p. H vs volume of titrant added

• A volume of 18. 28 m. L of a standard soluiton of 0. 1000 M Na. OH was required to neutralize 25. 00 m. L of a solution of nitric acid. What is the concentration of the nitric acid?

Buffered Solutions • Solutions that resist changes in p. H when acids or bases are added • Weak acid + salt containing conjugate base (strong) • Reacts with H+ and OH- ions so they don’t accumulate

HF + Na. F ↔ HF + + F + Na Add acid Add base 47
48

- Slides: 49