Unit 13 Solution Chemistry What is a solution













- Slides: 17

Unit 13 Solution Chemistry

What is a solution? • A solution is a homogeneous mixture of two or more substances in a single phase. Requirements: 1. contains two or more substances 2. each substance maintains its own chemical identity 3. the substances combine in a single phase

Circle the solutions: Gatorade Chocolate Milk Ice Cream Peanut Butter Nail Polish Distilled water Vinegar HCl (aq) Jello Shaving Cream Apple juice Salad dressing Salt water H 2 O(l) ocean water Cu. SO 4(aq) pond water drinking water Soda Na. Cl(s)

Solutions: Gatorade Chocolate Milk Ice Cream Peanut Butter Nail Polish Distilled water Vinegar HCl (aq) Jello Shaving Cream Apple juice Salad dressing Salt water ocean water pond water drinking water H 2 O(l) Cu. SO 4(aq) Soda Na. Cl(s)

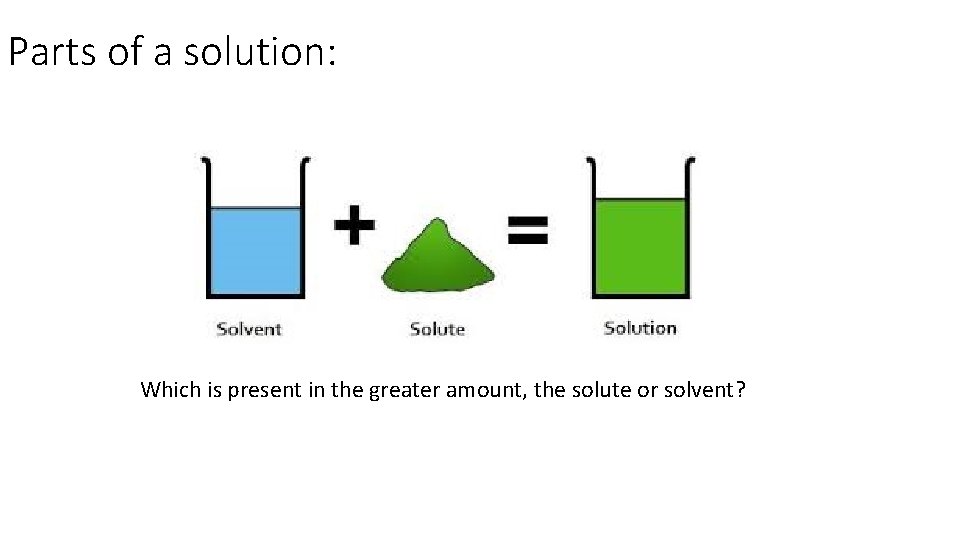
Parts of a solution: A solution is made up of two parts, the solute and the solvent. Solute = the substance being dissolved in a solutions, usually present in a smaller amount. Solvent = the substance used to dissolve the solute, usually present in a larger amount.

Parts of a solution: Which is present in the greater amount, the solute or solvent?

Concentration of a Solution: Qualitative (Non-quantitative) description of solution concentration: • Concentrated = a solution that contains a lot of solute. • Dilute = a solution that contains a small amount of solute. Some descriptions of solution concentration: 1. The solution is darker or lighter in color. 2. The solution is thick, not easy to pour.

Concentration of Solutions: • Quantitative (measured) description of solution concentration: Molarity (M) = moles solute Liter solution Dilution: M 1 V 1 = M 2 V 2

Summary solution concentration word bank: concentrated, molarity, dilute __________ solutions have a high solute: solvent ratio. __________ solutions have a low solute: solvent ration __________ gives a quantitative measurement of solution concentration

Molarity calculations: 1. Write the equation for molarity: 1. What is the molarity of a solution containing 0. 5 moles HCl in 1 L solution? 2. Sea water contains roughly 28. 0 g of Na. Cl per liter. What is the molarity of sodium chloride in sea water? 3. What is the molarity of 5. 00 g Na. OH in 1. 00 L solution? 4. How many moles of sodium carbonate (Na 2 CO 3) are there in 1. 5 L of a 2. 0 M solution?

Dilutions: M 1 V 1 = M 2 V 2 1. 100 m. L of a 5. 0 M solution is diluted to 250 m. L. Calculate the concentration (M 2) M 1= V 1 = M 2 = V 2 = 2. What volume (V 1) is needed to prepare 500. m. L of a 1. 0 M Na. Cl solutions. The starting solution has a concentration of 2. 5 M. 3. Calculation the concentration (M 2) of a solution prepared by diluting 25 m. L of a 1. 25 M solution to a new volume of 150 m. L.

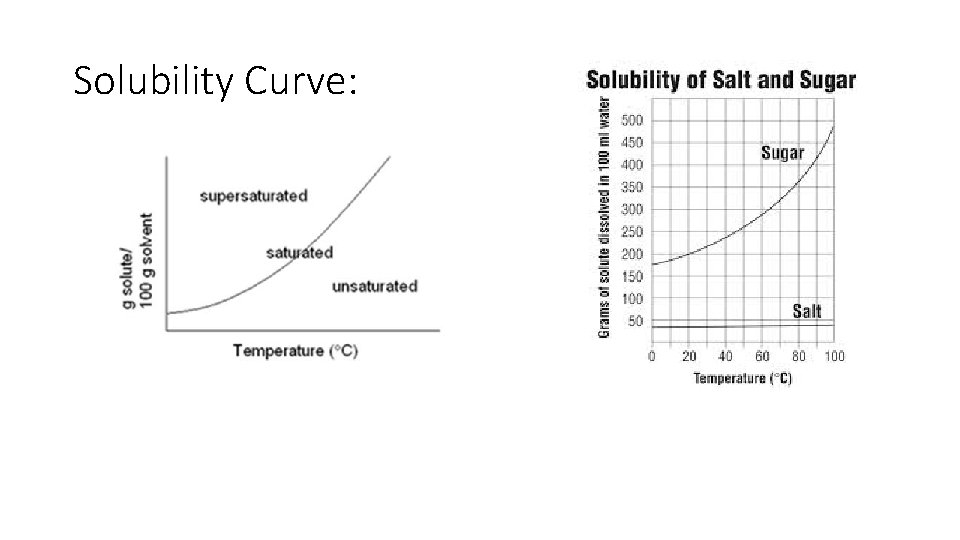
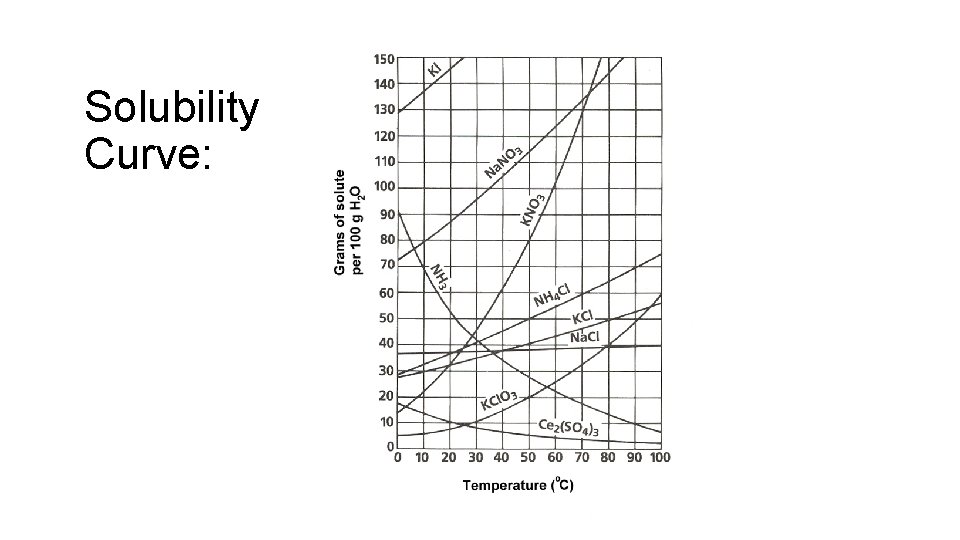
Solubility and solutions: “likes dissolve likes” Solubility = the amount of solute that can dissolve in given amount of solvent (generally water, 100 g) Unsaturated solution = less than the maximum amount of solute Saturated solution = the maximum amount of solute for a given amount of solvent Supersaturated solution = more than the maximum amount of solute is dissolved for a given amount of solvent

Solubility curve: • Describes the relationship of solubility and temperature ( Temp = Solubility) Think Ice tea and hot tea!

Solubility Curve:

Solubility Curve:

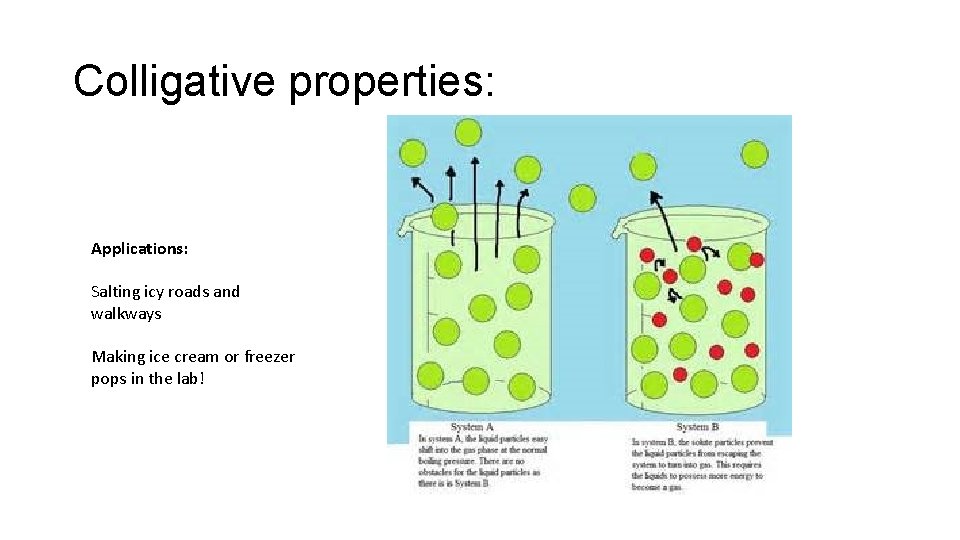
Solutions & Colligative Properties: Colligative properties are properties of solutions that depend upon the ratio of the number of solute particles to the number of solvent molecules in a solution. • Properties that are affected include: Freezing point Boiling point Vapor pressure 1. The freezing point of a solvent is lowered when a solute is added to a solvent. 2. The boiling point of a solvent is raised when a solute is added to a solvent. 3. The vapor pressure of a solvent is lowered when a solute is added to a solvent.

Colligative properties: Applications: Salting icy roads and walkways Making ice cream or freezer pops in the lab!