Unit 13 Electrochemistry Redox Oxidation Number State identifies

Unit 13: Electrochemistry (Redox)

Oxidation Number (State) identifies how many electrons are either lost or gained by an atom or ion during a chemical reaction

Review from Unit 5: Rules for assigning oxidation numbers 1. 2. 3. 4. An element by itself has an oxidation number of zero. Ions have an oxidation number equal to the charge. Group 1 metals in compounds always have a +1 oxidation number, and Group 2 metals in compounds are always +2. Fluorine always has an oxidation number of -1 in compounds.

Rules for assigning oxidation numbers: 5. 6. 7. 8. Hydrogen is +1 in compounds unless it is combined with a metal, when it is -1. Oxygen is usually -2 in compounds, but when combined with F it is +2. Oxygen is -1 in the peroxide ion. The sum of the oxidation numbers in a neutral compound must be zero. The sum of the oxidation numbers in a polyatomic ion is equal to the overall charge.

How to Recognize a Redox Rxn Ex) Na + Cl 2 Na. Cl REDOX Ex) Mg + Ni(NO 3)3 Mg(NO 3)2 + Ni Ex) H 2 SO 4 + Na. OH Na 2 SO 4 + H 2 O REDOX NOT REDOX

How to Recognize a Redox Rxn � Redox occurs when electrons are transferred between reactants � Because of electron transfer, the oxidation numbers of certain atoms or ions change � Helpful Hints: ◦ Must be redox if an atom is by itself on one side but in a cmpd on the other ◦ Single Replacement and Synthesis: always redox ◦ Double Replacement: never redox

Oxidation • the loss of e • LEO: Lose Electrons Oxidation • If a particle is oxidized, oxidation number increases • The particle undergoing oxidation is called the reducing agent Reduction • the gain of e • GER: Gain Electrons Reduction • If a particle is reduced, oxidation number decreases • The particle undergoing reduction is called the oxidizing agent LEO says GER!!

Redox Half-Reactions �Used to show many electrons are lost (oxidation) or gained (reduction) �Half-reactions must obey BOTH the Laws of Conservation of Mass and Charge �# of electrons lost = # of electrons gained �Oxidation: e- written on the right �Reduction: e- written on the left

Writing Half-Reactions Ex) Ca + Cu. Cl 2 Cu + Ca. Cl 2 Ex) Mg + Ni(NO 3)3 Mg(NO 3)2 + Ni Ex) Na + Cl 2 Na. Cl

Spontaneous Reactions (Table J) METALS: Strong tendency to lose electrons � More reactive metal gets OXIDIZED (loses e-) � Less reactive metal gets REDUCED (gains e-) � Element higher up starts as the atom (neutral) and becomes the ion (positive charge) NONMETALS: Strong tendency to gain electrons � More reactive nonmetal gets REDUCED � Less reactive nonmetal gets OXIDIZED

Writing Half-Reactions Ex) Na + Cl 2 Na. Cl Ex) Mg + Ni(NO 3)3 Mg(NO 3)2 + Ni Ex) Al + Fe 2+ Al 3+ + Fe

Spontaneous Reactions (Table J) METALS: Strong tendency to lose electrons � More reactive metal gets OXIDIZED (loses e-) � Less reactive metal gets REDUCED (gains e-) � Element higher up starts as the atom (neutral) and becomes the ion (positive charge) NONMETALS: Strong tendency to gain electrons � More reactive nonmetal gets REDUCED � Less reactive nonmetal gets OXIDIZED
What is an electrochemical cell? Electrochemical Cell: any device that converts chemical energy into electrical energy or electrical energy into chemical energy Voltaic (Galvanic): chemical electrical Electrolytic: electrical chemical

Voltaic Cells �Chemical energy electrical energy �Electrical energy is produced by a spontaneous redox reaction ◦ Electrons flow on their own

Voltaic Cells �AN OX: oxidation at the anode ◦ Anode has a negative charge ◦ Anode decreases in mass as oxidation occurs �RED CAT: reduction at the cathode ◦ Cathode has a positive charge ◦ Cathode increases in mass as reduction occurs • Electrons always flow from anode cathode

Electrode potentials � Standard electrode potential: a half-cell potential measured relative to the potential of zero for the standard hydrogen electrode, Eo. � The more positive (less negative) Eo, the greater the tendency for the substance to be reduced. � Eo= oxidation potential + reduction potential � In manipulating potentials, only change the signs of the values, not the magnitude.

Example: � What is the cell potential (cell voltage) of an electrochemical cell made of a copper electrode in a 1. 0 M Cu(NO 3)2 solution and a chromium electrode in a 1. 0 M Cr(NO 3) 3 solution?

Electrolytic Cells � Uses electrical energy to drive a non-spontaneous redox rxn � Electrons are pushed by an outside power source (battery) � Electrons still flow from anode cathode, but anode is (+) and cathode is (-) Anode: Ag (s) Ag+ (aq) + 1 e. Cathode: Ag+ (aq) + 1 e- Ag(s) Electroplating
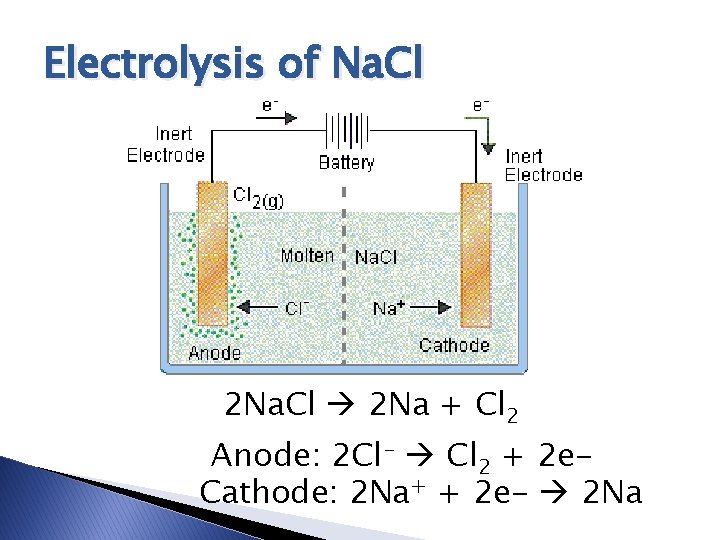
Electrolysis of Na. Cl 2 Na. Cl 2 Na + Cl 2 Anode: 2 Cl- Cl 2 + 2 e. Cathode: 2 Na+ + 2 e- 2 Na
- Slides: 19